Fig. 34.1
Two views of the anatomy of the velopharyngeal valve. At left is an axial view from above showing what looks like a two-dimensional opening, but when the same specimen is viewed from a different angle (from the right posterolateral aspect of the pharynx), the vertical height of the velopharyngeal mechanism can be appreciated. Therefore, the velopharyngeal valve is a three-dimensional tube with width, breadth, and height
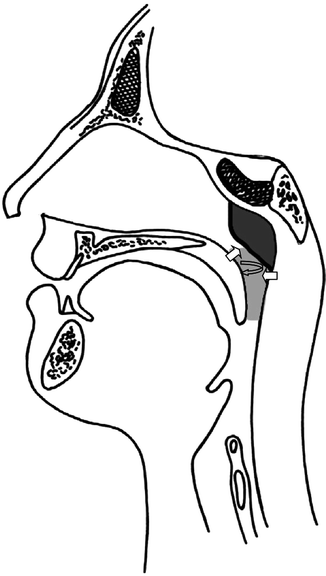
Fig. 34.2
Drawing showing the planes of movement necessary for velopharyngeal closure
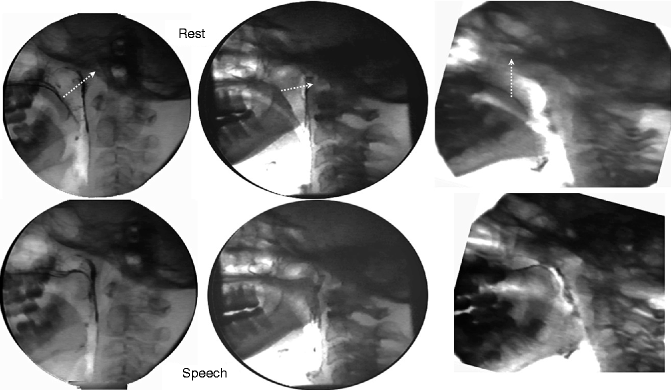
Fig. 34.3
Three lateral view videofluoroscopies showing the different types of movement of the velum that can occur depending on the orientation of the levator palatini
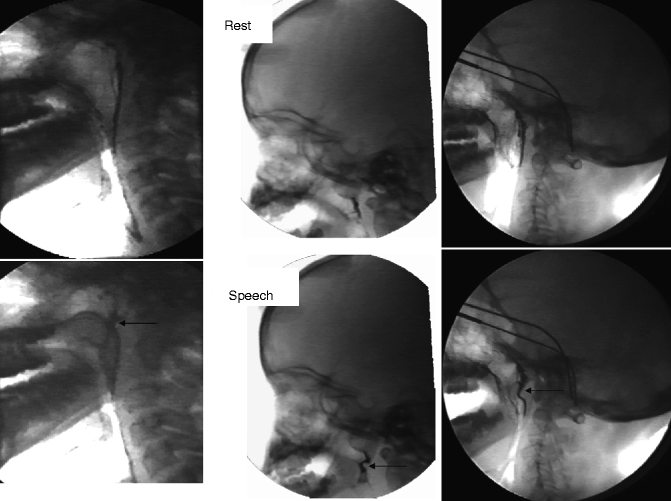
Fig. 34.4
Several different examples of Passavant’s ridge (arrow)
The variability of movement in the velopharyngeal valve from person to person was first described in a landmark article by Skolnick and colleagues (1973). They showed that some individuals have more lateral pharyngeal wall motion than others, some have Passavant’s ridge and some do not, and that this variability of movement is true for both normals and people with VPI. Subsequent research in large samples of both normals and people with VPI confirmed this finding (Croft et al. 1981b). This variability may be related in part to structural differences from person to person, but learnng is also clearly a factor. The role of learning has been demonstrated experimentally by using visual nasopharyngoscopic biofeedback to alter patterns of movement (Siegel-Sadewitz and Shprintzen 1982), therefore indicating that it can be altered by learning new muscular movements.
Therefore, velopharyngeal closure is a voluntary, CNS-mediated motor activity that is learned during early speech acquisition. The role of diagnosis needs to be aimed at determining if a failure of the velopharyngeal mechanism to close completely during normally nonnasal speech is an anatomic or physiologic limitation versus an error in learning.
34.3 Failures of Velopharyngeal Valving
There are a substantial number of reasons why velopharyngeal closure might not occur during speech that normally requires it. VPI is usually associated with palatal anomalies, but the frequency of VPI in disorders unrelated to structural malformations of the palate is also very common. There are no definitive data available that help us to determine what the most common cause of VPI might be, but suffice it to say that it is a common disorder. For the purposes of this text, we will focus primarily on clefting anomalies, but the reader should be aware that problems with the velopharyngeal valve associated with other disorders require careful scrutiny, as well.
Disorders of velopharyngeal function might best be divided into three major categories: structural, neurogenic/myopathic, and learned. These categories are not mutually exclusive. In other words, it is typical to have only one dysfunction causing the problem, but it might also be possible to have two or three of these factors in operation simultaneously.
34.4 Disorders of Structure
Structural anomalies include clefting of the palate (in all of its variations), anomalies of symmetry of the palate and/or pharynx, and anomalies of the surrounding pharynx (including abnormalities of the lymphoid tissue). Cleft palate is a broad designation for a broad spectrum of anomalies that range from complete clefts of the lip and palate to nearly an undetectable malformation known as occult submucous cleft palate. However, all of these anomalies could result in the same disorder of VPI.
34.4.1 Overt Clefts
Overt clefts of the palate are the most easily recognizable form of palatal anomaly and may range from complete bilateral clefts of the hard and soft palate to a cleft of the posterior aspect of the soft palate (Fig. 34.5). The extent of the cleft varies in both syndromic and nonsyndromic cases. The frequency of velopharyngeal insufficiency does not really vary according to cleft type, but cleft type does vary according to the primary diagnosis. In nonsyndromic clefts where there are no associated anomalies to affect velopharyngeal closure, the frequency of VPI after repair is probably primarily related to a combination of surgical variables and the amount of muscle deficiency associated with the cleft. However, in syndromic cases that account for more than half of all clefts (Shprintzen et al. 1985a, b), other syndromic features can contribute to VPI. For example, in velo-cardio-facial syndrome, besides cleft palate, there are three other clinical findings that contribute to VPI, none of which are mutually exclusive. Individuals with VCFS are known to have a high frequency of pharyngeal hypotonia (Zim et al. 2003), absent or hypoplastic adenoid tissue (Williams et al. 1987), and platybasia (Arvystas and Shprintzen 1984) that can increase pharyngeal depth. Therefore, primary cleft repair in VCFS almost always fails to resolve VPI (Shprintzen 2001).
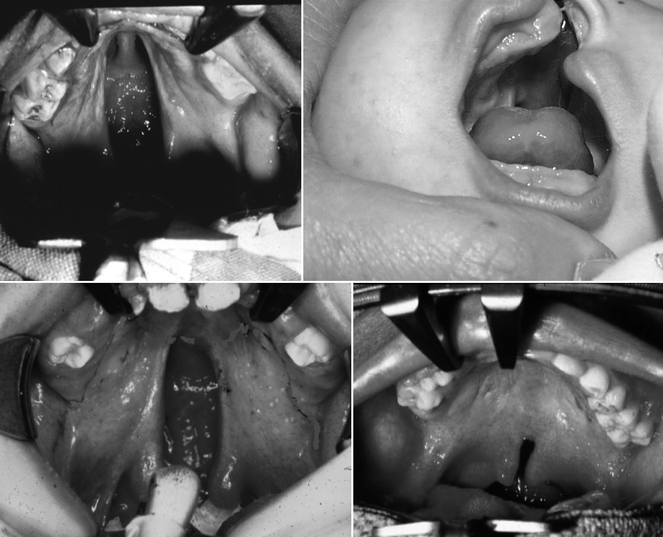

Fig. 34.5
Four different types of overt palatal clefts, including bilateral complete cleft of the hard and soft palate (top left), unilateral cleft of the hard and soft palate (top right), cleft of the entire soft palate (bottom left), and a cleft of the posterior third of the soft palate (bottom right)
Surgical repair of the palate is important for only one reason, to provide the patient with the possibility of developing normal speech. Feeding, ear infection, growth, etc.—none of these are factors of importance to consider in relation to the need for palatoplasty. Babies with clefts feed quite successfully when proper technique is used, as will be discussed briefly later. In fact, because palate repair is not usually completed until approximately 1 year of age, babies must feed over that period of time with a cleft. Middle ear disease is common in babies with clefts but is related to malformation of the Eustachian tube rather than muscular action of the velum (Shprintzen and Croft 1981). Middle ear disease can also be successfully managed medically or with myringotomy and tubes. Although feeding and hearing can be normal with an unrepaired cleft palate, speech cannot. Achieving acoustically acceptable speech is dependent on normal resonance and the ability to produce pressure consonants orally, both highly dependent on velopharyngeal closure.
Surgical repair of clefts are highly successful when they are accomplished prior to 18 months of age, and most surgeons in the USA are timing primary palatoplasty at or before the first birthday in order to intercept the early phases of speech development. It is clear that the majority of children who are born with overt clefts do not have VPI following primary palatoplasty (Salyer et al. 2006). Although surgical approaches to cleft repair have varied in relation to technique and timing, the majority of experienced surgeons who complete primary palatoplasties prior to 18 months of age achieve between 80 and 90 % positive outcomes defined as an absence of VPI after palatoplasty. Although this leaves a core of patients who will require secondary surgical procedures at a later date to resolve VPI, the overall percentage of individuals with VPI who have repaired clefts is probably quite low in most treatment centers.
The logical conclusion regarding the successful outcome of palate repair is that it is highly dependent on surgical technique. Actually, this has never really been proven, and there are many factors involved, including the age at time of surgery, the type of operation, other anatomical variations in the pharynx, muscle tone and developmental factors, and one factor that has received very little attention in the literature, the surgeon and his or her skill level. It is not within the scope of this chapter to discuss each of these issues, but it is important to show that the successful outcome of palate repair is dependent on more than one factor.
Although the palate is an important component of velopharyngeal valving, it is not the only one. The palate, and the velum in particular, is simply one component of the velopharyngeal mechanism, and there is quite a bit of variability of the size and shape of the other structures within the pharynx. The height and width of the pharynx varies from person to person just as height, weight, and head circumference do. The size of the adenoid is of particular importance because the adenoid sits in the plane of velopharyngeal closure in young children. In fact, velopharyngeal closure in children until at least 6 years of age is actually velar-adenoidal (Williams et al. 1987) (Fig. 34.6). Adenoids vary considerably in size, and there is some evidence that suggests that adenoid size may be the most important factor for successfully achieving velopharyngeal closure (Havkin et al. 2000). The larger the adenoid, the less movement is required of the velum. Although the adenoids are important to normal speech production, the tonsils are not and will be considered separately later in this chapter. It is clear that adenoidectomy should be avoided (unless there is severe upper airway obstruction in which case partial adenoidectomy may be indicated) in individuals with cleft palate or submucous cleft palate because of the potential for the development of the VPI once the adenoid tissue has been removed having the effect of increasing the volume of the nasopharynx (Croft et al. 1981a, b; Havkin et al. 2000).
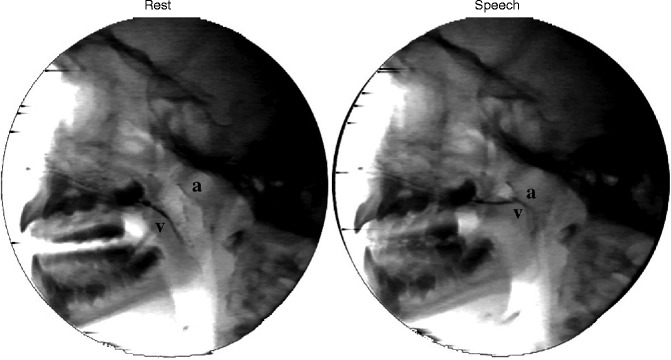

Fig. 34.6
Lateral view videofluoroscopy in a normal 4-year-old child demonstrating that the velum (v) never contacts the posterior pharyngeal wall during speech but instead approximates to the adenoid (a)
Another important factor is the volume of the nasopharynx. An increased volume of the nasopharynx can be caused by an abnormally obtuse angulation of the skull base (Shprintzen 2001). Many clinicians who use lateral view radiographs of the head and neck may interpret an increased nasopharyngeal volume as a “deep pharynx.” Alternatively, because the palate appears short within the presence of an increased pharyngeal volume, some clinicians may interpret this as a “congenitally short palate.” As will be discussed later, structural integrity of the palate cannot be assessed without a direct endoscopic view of the nasal surface of the velum. However, the depth of the nasopharynx can be assessed radiographically using the standardized measurements provided by a lateral cephalograph. Neither procedure (endoscopy or cephalometrics) is individually perfect for describing the anatomy of the pharynx. Rather, the information obtained from each is complementary.
34.4.2 Submucous Cleft Palate
Submucous cleft palate has traditionally been defined as the triad of bifid uvula, notching of the posterior border of the hard palate, and a visible diastasis of the musculature in the soft palate referred to as a zona pellucida (Fig. 34.7) (Calnan 1954). However, although all cases with these clinical findings have submucous cleft palate, not all people with submucous cleft palate have all three of these findings. Some have two of these findings, usually a bifid uvula and hard palate notch, some have only one of these findings, almost always a bifid uvula, and more interestingly, some have none of these findings. Submucous cleft palate is simply a milder or less obvious expression of cleft palate. In this case, the skin, or mucous membrane, is intact but the underlying musculature is not and some muscle may be congenitally absent (Croft et al. 1978; Lewin et al. 1980). Of interest, although people with submucous cleft palate do have velopharyngeal insufficiency and hypernasal speech, the large majority do not (Shprintzen et al. 1985a, b). This is interesting in light of the fact that many surgeons perform palatoplasty in infancy, before the onset of speech in an attempt to avoid speech problems by rearranging the muscles to a more normal configuration in the palate for individuals with submucous cleft palate even though the large majority of people with submucous clefts have normal speech without such surgery. This may indicate that other factors besides muscle integrity play a large or even larger role in normal speech production than simply the degree of palatal normality.
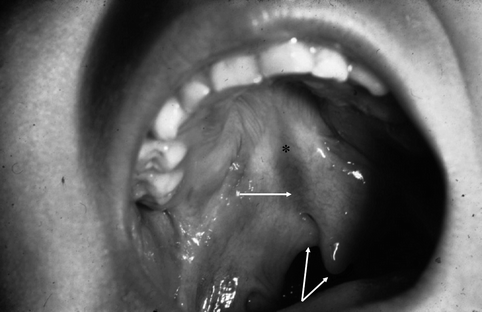

Fig. 34.7
Obvious submucous cleft palate showing a bifid uvula (dual arrow), a notch in the posterior border of the hard palate (*) and zona pellucida (single arrow)
34.4.3 Occult Submucous Cleft Palate
In 1975, Kaplan reported an abnormality of the palate he labeled the occult submucous cleft palate (Kaplan 1975). Kaplan’s report described four individuals with VPI who had normal appearing palates on oral examination, without evidence of bifid uvula, a zona pellucida, or notching in the hard palate. At surgery for correction of their VPI, he dissected the palate and studied the musculature noting that the muscle fibers were abnormal in position and orientation as might be found in a cleft palate or submucous cleft palate, hence the label occult submucous cleft palate (occult meaning mysterious). He concluded that the only way one could detect this anomaly was by surgical dissection. However, subsequent publications have demonstrated that occult submucous cleft palate can be detected by nasopharyngoscopy (Croft et al. 1978; Lewin et al. 1980; Shprintzen 1979a, b, 2000). Unfortunately, many clinicians and researchers have not applied this diagnostic approach so that many cases of occult submucous cleft go undetected and the frequency of clefting becomes significantly underestimated. It is interesting to note in retrospect that three of the four patients shown by Kaplan clearly have velo-cardio-facial syndrome, a condition known to have a high frequency of occult submucous cleft palate (Shprintzen 1982). It is not known what the frequency of hypernasal speech is in association with occult submucous cleft palate because it is very unlikely that individuals with normal speech will ever have the anomaly detected unless nasopharyngoscopy is performed for some other reason.
34.5 Normal Anatomy of the Velopharyngeal Mechanism: Developmental Changes
The structures of the palate, nasopharynx, and oropharynx (the structures that constitute the velopharyngeal mechanism) are not static. They undergo significant alteration and remodeling over time relative to their functions and age. Therefore, mechanisms of velopharyngeal function at one point of time in life may not be the same as another. This means that broad generalizations about how the palate and pharynx function should be avoided because they may be age dependent. As mentioned previously, the palate and pharynx are multifunctional. Modulating the size and shape of the upper airway, sometimes referred to as the aerodigestive tract, is necessary for proper function during speech, feeding, and airway maintenance. However, these functions vary with age. Infants have the ability to separate the oropharyngeal airway from the nasopharyngeal airway making it easier for them to breathe through their noses while they eat than is true for adults. This is because the upper airway in infants is vertically shorter and oriented in a more horizontal than vertical angle (Shprintzen 2003) (Fig. 34.8). The shorter upper airway in infants allows the epiglottis to be in close proximity to the soft palate creating a conduit for milk to flow unobstructed into the pyriform sinuses laterally while the baby breathes through the nose. Because infants’ ventilatory efforts are faster and more frequent than adults and their smaller lung capacity and blood volume makes them more prone to rapid oxygen desaturation than adults, it is important for them to breathe uninterrupted during feeding to prevent hypoxia and hypercarbia. Babies typically eat semi-reclined, a position that actually makes the upper aerodigestive tract more vertical so that feeding is facilitated by gravity and an easy flow of fluids into the hypopharynx and esophagus.
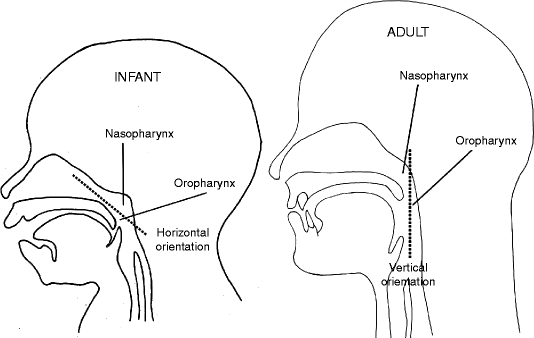

Fig. 34.8
Comparison of the upper airway in an infant (left) to that of an adult (right) showing a more vertical orientation of the pharynx in the adult compared to the more horizontal position in the infant
As children grow, growth in the maxilla and mandible is both horizontal and vertical. This expands the upper airway vertically and draws the palate and velum away from the epiglottis and larynx. The entire pharynx expands in three dimensions (width, depth, and height) as the entire palate grows in length. In fact, if the upper airway did not increase in height, the palate would hang too deeply into the pharynx resulting in upper airway obstruction. In fact, in some genetic disorders with midfacial anomalies, such as Crouzon syndrome, Apert syndrome, and Down syndrome, where the maxilla is hypoplastic in all dimensions, the phenomenon of the velum contributing to upper airway obstruction is commonplace, resulting in obstructive sleep apnea as well as obstructive daytime respiration.
As the pharynx becomes more vertical with age, the adenoids begin to involute. As reported previously, if the adenoid did not involute, the pharynx would become obstructed because of its change in shape and orientation (Shprintzen 2003). This is because the hard and soft palates actually migrate to a position relatively closer to the posterior pharyngeal wall over time. If the adenoid remained large (typically occupying about 25–50 % of the postnasal space before involution begins between 8 and 10 years of age), the palate would be flush against it, thereby compromising the nasopharyngeal airway.
The changes in the shape, volume, and position of the upper airway have been speculated to potentially result in changes in velopharyngeal function over time. Clinically, I have often heard professionals suggest that gradual adenoid involution can result in the development of velopharyngeal insufficiency. Although there are a very small number of anecdotal cases reported with a late onset of velopharyngeal insufficiency (Mason and Warren 1980), such events are very rare, and I have never encountered such a case in more than 38 years of practice with the exception of late onset muscular dystrophies, myasthenia gravis, or other neuromuscular diseases.
34.6 Other Structural Disorders
Although the adenoid is important to normal speech production, other lymphoid tissues do not play a role and may be detrimental to normal speech production. There are three other masses of lymphoid tissue within the oral and pharyngeal airway: two palatine tonsils and one lingual tonsil. The lingual tonsil sits at the base of the tongue anterior to the vallecula and plays no role whatsoever in speech production. When enlarged, the lingual tonsil can cause the sensation of globus, or a feeling that something is stuck in the bottom of the throat. If very large, a muffled resonance may also result from an enlarged lingual tonsil because it may physically damp the sound waves produced by the vocal cords just millimeters below the base of the tongue. However, the lingual tonsil does not cause hypernasality or hyponasality and is too far away from the velopharyngeal valve to interfere with it.The palatine tonsils, however, may alter nasal resonance patterns as has been documented in the medical literature on several occasions (Henningsson and Isberg 1988; MacKenzie-Stepner et al. 1987; Shprintzen et al. 1987).
The palatine tonsils are situated in the posterior oral cavity with their attachment in the faucial arch in between the anterior and posterior tonsillar pillar. Assessment of tonsillar size is often relegated to oral assessment with a flashlight and tongue blade. It is typical for clinicians to use a rating scale between 0 and 4+ based on the degree of medial excursion of the tonsils toward the midline of the posterior oral cavity with 0 implying that the tonsils are not visible, 1+ referring to tonsils fully contained within the faucial pillars, 2+ meaning the tonsils extrude medially beyond the pillars but less than halfway to midline, 3+ indicating that the tonsils extend more than half of the way to midline, and 4+ meaning they are touching in the midline (kissing tonsils). The problem with this scale is that no one has told the tonsils that if they get big that they must grow medially. In fact, in many cases, they grow back into the pharyngeal airway and may grow down into the hypopharynx or up into the nasopharynx (Fig. 34.9
Stay updated, free dental videos. Join our Telegram channel

VIDEdental - Online dental courses


