Fig. 9.1
Electron micrograph of titanium sandblasted and acid-etched surface (SLA)
Many researchers studied the SLA surface commercially produced by Institut Straumann AG, Basel, Switzerland, and found that the Sa value (the arithmetic mean of deviations in the roughness profile from mean line in 3 dimensions) is 1.79 ± 0.2 μm (evaluated at the top of the thread) [18] and the Ra value (the mean height of the roughness based on only 2 dimensions) is 2.93 ± 0.46 μm [19]. Additionally, the difference between the Sa and Ra value with regard to these 2 studies can be related to the fact that the Sa value was measured by optical profilometry [18] while the Ra value was determined by evaluating scanning electron micrographs from implants using image analysis software [19]. Such a titanium oxide surface exhibits low surface energy because of adsorbed hydrocarbons and carbonates from ambient air [2]. Taborelli et al. confirmed the water contact angle of the SLA surface about 117° ± 2.7 [20], while Buser et al. measured the dynamic contact angle (DCA) of the SLA surface and the results indicated that the SLA surface was hydrophobic (DCA = 138.3° ± 4.2) [11]. During acid-etching, the titanium oxide layer is dissolved, and small native hydrogen ions diffuse into the unprotected implant surface, which enrich the implant surface with hydrogen and precipitate into titanium hydride (TiH) [21]. X-ray diffraction (XRD) analysis of SLA-treated titanium samples showed the presence of 20–40 % of titanium hydride (d-TiH2-x) in addition to titanium [22, 23].
It has been reported that the etching process modifies the titanium surface composition of SLA-treated implants. Observation has shown that the dull SLA surface is soft [24], particularly when compared with a titanium plasma–sprayed (TPS) surface. The SLA surface consists mainly of TiO2 with some carbon containing contamination (like hydrocarbons) due to the exposure to air. X-ray photoelectron spectroscopy (XPS) analysis indicated that the SLA surface had a 44.2 ± 1.9 at% (atomic concentration) oxygen (O) concentration, an 18.4 ± 1.6 at% titanium (Ti) concentrations [11], and a 37.3 ± 3.4 at% carbon (C) concentration, which is comparable to the result of Kang et al. with 47.1 at% O, 20.1 at% Ti, and 32.0 % C [25].
Besides pure titanium, SLA surfaces can also be produced on other materials, such as titanium-zirconium alloys [13] and zirconium dioxide ceramics (Fig. 9.2). Due to the similar crystal structure of titanium and zirconium, the TiZr alloy can be sandblasted and acid-etched, exactly like commercially pure titanium (compare 3.1 in this chapter), to create a micro-rough SLA surface (Fig. 9.3). In contrast to that, with regard to zirconia ceramics, it has been shown that surface treatment procedures that create micro-rough surface topographies, like conventional sandblasting or uncontrolled machining processes, might reduce the fracture strength of zirconia dental implants and lead to implant fractures [26, 27]. Thus, the manufacturing processes of creating micro-rough surfaces on commercially pure titanium or titanium alloy implants cannot simply be transferred to zirconia but must accurately be attuned to the material properties of the zirconia ceramics. The first manufacturing process that created a micro-rough surface topography on zirconia implants that was similar to the SLA surface on titanium implants, without compromising the zirconia implant fracture strength, was a low-pressure injection molding technique, using a mold showing an SLA-like surface topography, followed by an etching procedure with hot hydrofluoric acid [28–31]. Later on, for zirconia dental implants, the manufacturing and surface treatment procedures have been further developed so that currently a micro-rough surface topography can be produced on zirconia implants by using a specially designed, mild sandblasting procedure followed by an etching procedure with hydrofluoric acid (zirconia large-grit sandblasted and acid-etched, ZLA™ surface, Straumann® PURE Ceramic Implant, Institut Straumann AG, Basel, Switzerland, Fig. 9.2), again without compromising the resistance to fracture of the ZLA-treated zirconia implants. The sandblasted and acid-etched surface on zirconia implants has a similar surface topography compared to the SLA surface on titanium implants (Figs. 9.1 and 9.2); however, with regard to quantitative surface analysis, the micro-rough zirconia implant surface shows a reduced arithmetic mean value (Sa = 0.70 μm). Calculations were performed with the use of a Gaussian filter with a cutoff wavelength of 30 μm (information provided by Institut Straumann AG, Basel, Switzerland) and compared to implants with an SLA surface (Sa = 1.15 μm, Table 9.1).




Fig. 9.3
SEM image of SLA/modSLA surface on c.p. titanium (a) and on TiZr alloy (b)
Table 9.1
Roughness parameters of SLA and modSLA surfaces [11]
|
Roughness parameter
|
SLA
|
modSLA
|
|---|---|---|
|
Sa [μm]
|
1.15 ± 0.05
|
1.16 ± 0.04
|
|
Sq [μm]
|
1.45 ± 0.06
|
1.45 ± 0.04
|
|
St [μm]
|
7.83 ± 0.47
|
7.65 ± 0.31
|
|
Sk [μm]
|
4.44 ± 2.33
|
4.51 ± 2.26
|
Preclinical In Vitro Studies
SLA: Osteoblast Activity
Bone formation on implant surfaces requires recruitment of osteoblast precursor cells, their differentiation into secretory osteoblasts, production of unmineralized extracellular matrix (osteoid), and calcification of the extracellular matrix [32], which finally lead to osseointegration. Initial interactions between implant surfaces and cells in the early stages of healing are believed to predetermine later events in the process of osseointegration of dental implants [32, 33]. Baschong et al. [34] investigated the influence of SLA surfaces on the differentiation of human mesenchymal progenitor cells (HMPC), i.e., the type of cells immigrating to colonize the implant surface upon implant insertion [35, 36], from adult bone marrow and demonstrated that SLA surfaces promotes osteogenic differentiation of HMPC, which could explain in vivo observations of enhanced bone–implant integration.
It is widely recognized that SLA surfaces increase osteoblast attachment, differentiation, and biomineralization, in comparison with smoother topographies, which promote adhesion formation, spreading, and proliferation [37]. Zinger et al.’s study showed that for hemispherical cavities, the minimal width for bone cell response is around 30 μm, and interestingly, cells cultured on the SLA surfaces preferentially occupied the shallow hollows existing in the SLA rough topography, which measured around 20–30 μm [38]. Osteoblasts also appear to be sensitive to surface roughness and exhibit greater initial attachment to rough titanium surfaces [39]. Sammons et al. demonstrated that rat calvarial osteoblasts attached and spread on the SLA surfaces more rapidly than on smoothed, anodized, or acid-etched surfaces [19]. It has also been shown that osteoblasts exhibit a more differentiated phenotype when grown on titanium substrates with micron-scale roughness than when grown on smooth titanium substrates or on tissue culture polystyrene [40]. Schwartz et al. found that osteoblasts exhibited a decrease in cell number and increase in osteocalcin when grown on SLA surfaces. A number of in vitro laboratory studies have shown that SLA surfaces could increase the response of osteoblast-like cells to systemic hormones [41] and promote osteoblast and chondrocyte differentiation [15]. Interactions with the SLA surface may facilitate mechanical interlocking of cells with the surface, allowing the ingress of vascular tissue and favoring osseointegration.
The osteophilic properties of the SLA surface were confirmed in a series of in vitro studies examining various titanium surfaces in tissue culture with osteoblast-like cells [15, 41–43]. In vitro studies have shown that SLA surfaces also influence a number of events in the process of osteoblast differentiation including spreading and proliferation; the production of alkaline phosphatase, collagen, proteoglycans, and osteocalcin; and synthesis of cytokines and growth factors (TGF-β1 and PGE2). Osteoblasts grown on SLA surfaces enhance osteointegration by producing local factors that regulate bone formation as well as bone remodeling, including the RANK ligand decoy receptor osteoprotegerin (OPG). In addition to producing a collagen-rich extracellular matrix [44], increased alkaline phosphatase activity, and elevated levels of osteocalcin [42], osteoblasts produce increased levels of local factors on these surfaces, including prostaglandins E1 and E2 (PGE2) [41] and transforming growth factor beta-1 (TGF-β1) [45]. Levels of latent and active TGF-β1 were increased in the conditioned media of cultures grown on the SLA surfaces, which then increase the levels of OPG [2]. The SLA surface induced an accelerated gene expression of the bone matrix molecules osteopontin and osteonectin, along with an upregulation of bone sialoprotein, collagen type III, and integrins in the initial healing stages up to 1 week [46]. These studies demonstrated that, in vitro, the SLA surface could accelerate the bone formation that occurs at the cell-implant surface.
SLA: Osteoclast Activity
Osteoclasts are also important in the process of osseointegration. Osteoclasts and osteoblasts show a close interplay, called coupling, regulated through multiple signaling pathways. Lossdorfer et al. showed that the SLA surface can enhance the phenotypic maturation of MG63 osteoblast-like cells towards a more differentiated osteoblast cell type expressing local factors that inhibit osteoclastogenesis [17]. In addition, Brinkmann et al. found that osteoclast differentiation on SLA surfaces seems to be comparable with differentiation on native bone, which will attract osteoblasts and then mineralize the bone around the implant [47].
Preclinical In Vivo Studies
SLA: Characteristics
The key events that lead to osseointegration of an implant occur largely at the bone-implant surface. Various publications have demonstrated that bone contact differs when different titanium implant surfaces are used. In several in vivo studies, SLA surfaces were found to produce better bone fixation than smooth surfaces [3, 48–50], and the SLA surfaced implants also showed a more than 5 % higher interfacial stiffness than machine surfaced implants [51]. A histometric study by Buser et al. [3] evaluated 5 different titanium surfaces in miniature pigs and found that the SLA surfaced implant revealed the best bone apposition to the implant surface among the titanium surfaces, with 52 % and 58 % of bone-to-implant contact (BIC) after 3 and 6 weeks of healing, respectively. This surface also was evaluated in a separate animal system in the oral cavity under loaded and non-loaded conditions and compared to implants with a titanium plasma-sprayed surface. The SLA implant surface produced favorable results in this study as well, with the BIC level of about 72.33 % after 12 weeks of healing [50]. Perrin et al. acquired 82.12 ± 6.1 % BIC by using SLA surfaced implants in Landrace pigs in a submerged way after 10 weeks [23]. Data from these in vivo animal studies suggest that implants with SLA surfaces produce a more rapid bone response and/or more bone-to-implant contact than ones with smooth or turned surfaces.
Besides animal studies, a human study was performed by Lang et al. [52], during which SLA surfaced implants were placed in the volunteers’ mandibular retromolar area and retrieved with the surrounding tissues after healing periods of 7, 14, 28, and 42 days. The histological analysis showed that osseointegration took place with an increasing BIC from 7 to 42 days at which time it reached 62 % of the implant surface exposed to the parent bone. The authors pointed out that osseointegration appeared to be slower in humans when compared with animals, which is consistent with bone formation rates. Nevertheless, with a BIC of 62 % after 4 weeks of healing, the osseointegration may have developed to a sufficient degree to fully carry functional load [53]. The results of these tests show that the SLA surface has a high potential to form bone-to-implant contact in humans.
A significantly higher removal torque value has been demonstrated for SLA implants compared to smooth implant surfaces [14]. A removal torque study by Buser et al. in the maxillae of miniature pigs compared SLA surfaces with the machined and the TPS surface and confirmed that the machined surface had 8 to 10 times lower removal torque values when compared with the SLA surface and that the SLA surface showed slightly higher removal torque values than the TPS surfaces [14]. A biomechanical study by Wilke et al. tested removal torque values (RTVs) of unloaded titanium implants with various surface characteristics in the tibia of sheep. This study demonstrated that the RTVs for the SLA surface clearly exceed the mean RTVs of polished or fine-textured implant surfaces during the course of the study period. Further studies [4, 14, 54] using functional assays demonstrated that this same SLA surface resulted in significantly higher removal torque values than did smoother surfaces (e.g., machined surface). Thus, it appears that implants with an SLA surface might be able to be restored after shorter healing times than those associated with implants with a machined surface.
Less loss of bone height at the preload evaluation, as well as after a loading period, has been demonstrated for SLA implants. Cochran and coworkers [16, 50] found significantly less coronal bone loss in arches in which SLA surfaced implants had been placed, and this may be the result of the higher osteoconductive properties of the SLA surface. In a study conducted by Cochran et al. [16], an SLA implant was compared radiographically to a TPS implant under unloaded and loaded conditions in the canine mandible for up to 15 months. Radiographic assessment of the bone response to the implants was carried out by measuring the distance between the implant shoulder and the most coronal bone-to-implant contact (DIB) and by evaluation of bone density changes using computer-assisted densitometric image analysis (CADIA). DIB measurements revealed that SLA implants had significantly less bone height loss (0.52 mm) than TPS implants (0.69 mm) at the preload evaluation (p = 0.0142) as well as at 3 months of loading (0.73 mm/1.06 mm; p = 0.0337). The same trend was also evident for CADIA measurements with SLA implants showing higher crestal bone density values when comparing preload to baseline data (p = 0.0890) and 3 months to baseline data (p = 0.0912). Arlin also found that the frequency of crestal bone loss was lower for SLA implants than for TPS implants [55]. Moreover, they also found that cumulative survival rates were equally good for SLA and TPS implants; additionally, the failure rate for SLA implants was lower than that for TPS implants. In an additional study, Perrin et al. found that surface composition did not play a significant role in the bone response to the SLA surface, and they concluded that the osteophilic properties of the SLA surface are due to its surface topography and not to its specific surface composition [23].
SLA: Bone Growth
It has been demonstrated that SLA surfaces allowed for “contact osteogenesis” to take place [56]. Cochran et al. demonstrated that remodeling processes were “fully occurring” already after 3 months of healing of a 200-mm-wide gap that occurred between the bone wall and the screw threads of an implant with an SLA surface [50]. Botticelli et al. [57–60] have published the results from 4 experimental studies in the dog. In these studies, they created defects lateral to the implants to simulate implants placed in extraction sockets. The histological analysis indicated that defects lateral to implants with an SLA surface heal with adequate osseointegration and demonstrated that the healing of a wide marginal defect around an SLA implant is characterized by appositional bone growth from the lateral and apical bone walls of the defect. In another one of Botticelli’s publications [61], a model was described which allowed the study of bone formation adjacent to endosseous implants. In this model, prior to implant placement, a large defect was prepared in the marginal bone compartment of the recipient site. In biopsies obtained after 4 months of healing, it was observed that the defects adjacent to the implants had been filled with newly formed bone. In addition, the degree of bone-to-implant contact that had been established between the newly formed bone tissue and the SLA surfaced implant was high and not different from those characterizing similar implants placed in a recipient site without defects of the alveolar bone crest. The SLA surface appeared to have the ability to close the marrow spaces with new bone when inserted in rather spongious bone, leading to a quasi-continuous layer of bone running along the implant surface. This property could explain the higher BIC level recorded at SLA surfaces when compared to TPS surfaces [3]. This SLA surface appeared to express its osteophilic properties more readily in spongious bone rather than in cortical bone.
SLA: Bone Graft Materials
Guided bone regeneration (GBR) has been successfully applied to treat bone defects associated with implants, and bone grafting materials have often been placed around dental implant surfaces to simultaneously reduce treatment time. Freilich et al. [62] tested the vertical bone-forming capacity of demineralized bone matrix (DBM) around SLA surfaced implants in membranous bone in a rabbit model and demonstrated that the DBM, in combination with the implant and scaffold retention screw which kept the scaffold in a stable, non-compressed position during healing, was successful at inducing new bone formation around the SLA roughened surface. The mean supracrestal bone height determined via histomorphometric analysis was 2.4 ± 0.6 mm and that by micro-CT was 2.1 ± 0.9 mm. They also pointed out that the high bone contact value obtained (58.1 ± 14 %) within the new supracrestal bone indicates successful implant placement into alveolar bone by clinical standards, and these data illustrate the potential for vertical bone-guiding capacity of sandblasted and acid-etched implants. Carmagnola et al. [63] demonstrated that the SLA implants placed in rabbit tibiae previously grafted with 3 different biomaterials, i.e., Bio-Oss®, Ostims-Pastes®, and PerioGlas®, obtained a larger extent of osseointegration, although not statistically significant, than implants placed in non-grafted bone. De Vicene et al. [64] evaluated the efficacy of collagen membranes (Bio-Gide®, Geistlich Pharma), either alone or combined with a human demineralized freeze-dried bone allograft (DFDBA) or natural bovine bone graft, in bone defects around dental implants with an SLA surface, and it was observed that the mean bone-to-implant contact was 35.3 %, and was even higher, 75.6 %, when the defects were filled with inorganic bovine bone. They pointed out that although no statistically significant differences were found in this study between the membrane and non-membrane groups, bone defects augmented with anorganic bovine bone and membranes showed the most promising results from a histological and histomorphometric perspective. Retzepi et al. [65] demonstrated that de novo alveolar bone formation can be achieved around SLA surfaced implants via application of the GBR principle in experimentally induced diabetic rats; however, there was higher outcome variability and an increased rate of infectious complications in these diabetic animals. These studies demonstrate the value of this surface to help promote vertical bone growth by providing an osteoconductive surface to support osteoblast cell adhesion and growth [66]. Therefore, the SLA surface has osteoconductive characteristics, which have the capacity to guide bone growth when placed in conjunction with bone grafting materials.
SLA-like Surface on Zirconia
As described above, recently a micro-rough surface topography has been produced on zirconia implants by using a specially designed sandblasting and acid-etching procedure (Fig. 9.2). However, the first generation of these micro-rough zirconia implants was produced by a low-pressure injection molding technique, using a mold showing an SLA-like surface topography, followed by an acid-etching procedure [28–31], creating a similar surface topography compared to the current sandblasting and acid-etching procedure (Fig. 9.2). At present, only experimental data from the first generation of these micro-roughened zirconia implants are available [28–31]. Histological and biomechanical animal studies in miniature pigs investigated the bone tissue response to these first-generation micro-roughened zirconia implants and compared it to titanium implants with an SLA surface [28–31]. The authors demonstrated comparable bone-to-implant contact ratio and bone density values for zirconia implants (BIC range between 67.7 ± 21.1 % and 70 ± 14.5 %; bone density range between 60.4 ± 9.9 % and 63.3 ± 21.5 %) and titanium SLA implants (BIC range between 64.7 ± 9.4 % and 83.7 ± 10.3 %, bone density range between 61.1 ± 6.2 % and 68.2 ± 5.8 %) after 4, 8, and 12 weeks of unloaded healing. No statistically significant differences could be detected at any investigated time point [30]. With regard to biomechanical testing, mean torque-out values ranged between 97.4 ± 29.3 and 139.6 ± 56.63 Ncm for zirconia and between 131.6 ± 35.6 and 177.6 ± 51.6 Ncm for titanium SLA at corresponding time intervals. After 4 and 12 weeks of healing, no significant differences could be evaluated between both materials [31]. Thus, it can be suggested that the micro-roughened zirconia implants with an SLA-like surface topography have a comparable osseointegrative capacity as titanium implants with an SLA surface topography.
Clinical Studies
SLA: Bone Formation
The concept of enhanced bone formation around the SLA implant surface in humans was demonstrated by a clinical human study [67], during which the SLA surfaced implants were placed in extraction sockets. This occurred both in the maxilla and the mandible and all implants were not loaded. Four months later, surgical re-entry procedures were performed, and it could be observed that the majority of the extraction sockets were almost completely filled by bone. Based on these results, the authors concluded that marginal gaps following placement of the SLA surfaced implants into extraction sockets may predictably heal with bone formation and defect resolution. Barewal et al. measured the resonance frequency of SLA implant stability in vivo and found that there was no significant difference in the pattern of stability changes among different bone types (type I–IV) after 5 weeks of healing; however, the dip in stability which occurs around 2–3 weeks in patients is lower in more cancellous bone types [68]. The results of another evaluation show that the SLA surfaced implants have a significantly higher survival rate than machine-surfaced implants in autogenous grafted maxillary bone [69]. Stricker et al. [70] demonstrated that placement of SLA surfaced implants, in combination with autografts for sinus floor elevation, is a safe and predictable procedure for the reconstruction of the severely resorbed posterior aspect of the maxilla.
SLA: Reducing Time to Loading
A formal prospective, multicenter clinical trial [71] was initiated to determine whether the advantages of increased bone formation and osseointegration demonstrated for SLA surfaced implants seen in experimental in vitro and animal studies could be translated into benefits for patients. It was reasoned that if greater bone contact and a stronger bond to the bone occurred at earlier times around implants with an SLA surface, then patients should be able to have their implants restored after shorter healing periods than those considered conventional (3–6 months) which were established in studies with smooth machined surfaced implants. In this clinical study, SLA implants were loaded after 6 weeks when placed in class I, II, or III bone or after 12 weeks if in class IV bone. The results demonstrated that, under these defined conditions, the SLA surfaced implants can be restored after approximately 6 weeks of healing with a high predictability of success, defined by abutment placement at 35 Ncm without counter torque [71]. This represented a significant advancement in the restoration of missing teeth with implants by directly providing the patient benefit of reduced treatment time. However, one possible shortcoming of this trial was that the patients were selected carefully and monitored closely. As such, their treatment represented ideal conditions in which to evaluate the reduced healing time. However, the successful abutment placement that has been demonstrated in this clinical trial, and the subsequent implant success, provides further evidence that the SLA surface has significant clinical advantages such as shorter restoration times than those used in earlier studies on implant restoration [71]. At both 1- and 2-year follow-up, 99 % of the implants were successful. An identical success rate (i.e., 99 %) was also reported at 3 years [72], which indicated that no so-called “late failures” occurred in the 3-year time frame.
The possibility of reduced healing times was also examined in another larger multicenter, prospective human clinical study, in which the patients were not as carefully selected and monitored. The success of early abutment placement was again assessed without counter torque, based on the documented advantages of the SLA titanium surface, which has both high percentages of bone-to-implant contact in descriptive histomorphometric studies [50] and high removal torque values in functional studies based on the strength of the bone-to-implant contact [4, 73]. The implants in this less-controlled environment could be placed and restored successfully with reduced healing times (approximately half the conventional healing time) without a consequent increase in failure or complication. The results of this multicenter clinical trial clearly established the capacity of implants characterized by the SLA surface to withstand abutment torquing and consequent loading within the experimental time frame. Therefore, SLA surfaced implants have a benefit for patient care which is a shorter healing time than that historically recommended. By reducing the time required for osseointegration and loading, patient acceptance and rehabilitation options were improved.
As shown above, early loading of SLA implants have demonstrated clinical outcomes equivalent to conventional loading [71, 74]. A healing time of 6 weeks for implants placed in good-quality bone has been recommended for SLA surfaced implants [71, 75]. Bornstein et al. used an early loading protocol with 6 weeks of healing to demonstrate that titanium implants with the SLA surface can achieve and maintain successful tissue integration with high predictability for at least 5 years of follow-up in selected patients and sites [76]. Salvi et al. pointed out that loading of titanium implants with an SLA surface as early as 2 weeks did not appear to jeopardize the osseointegration healing process in the posterior mandible [77]. Quinlan used 48 SLA surfaced implants in a canine model and indicated that no statistically significant differences existed between conventionally loaded, early loaded, and immediately loaded SLA implants by clinical, radiographic, and histologic data [78]. Immediate or early loading of SLA implants in single-tooth replacement [79], splinted crowns and fixed prostheses [80–82], full-arch prostheses [83], and overdentures [84] have demonstrated success and survival rates comparable with those obtained using older standard conventional loading protocols (6–9 months).
Due to the 2 levels of micro-roughness of the SLA surface, osseointegration of SLA implants has been improved, and time to loading has been reduced to at least 50 % of conventional healing times. The biologic reasons for the early loading capacity of the SLA implants are based on the osteoconductive nature of the surface and the physical characteristics of the titanium surface resulting from the subtractive techniques of producing the SLA implant surface. Electron micrographs show bone cells with their cell bodies located over the 20- to 40 μm pits created by the sandblasting procedure, whereas the cell extensions reach out to the 1- to 2 μm roughness of the acid-etching procedure. Apparently, osteoid is laid down under the cell bodies, calcifies, and acts as retentive areas for the implant to resist forces applied to the implant [54, 85]. These mechanisms may explain why the implants can resist 35 Ncm of force without counter torque at 6–8 weeks and allow for restoration of the implant at reduced healing times. This suggests that the early bone-to-implant contact that is formed is sufficiently strong to resist occlusal forces on the restorations.
SLA: High Survival and Success Rate
A meta-analysis of longitudinal descriptive implant experiences in patients has suggested that, in certain indications, the advantages of rough titanium surfaces can also be reflected in better success rates for patients [86]. A prospective, multicenter, human clinical observational study was conducted by Cochran et al. [87] which was performed in a prospective fashion involving 92 practitioners in 16 countries. Most implants were placed in posterior locations and were placed in intermediate-quality (type II and III) bone. The implants were to be placed and restored in predominantly private practice settings around the world. Ninety-two practitioners in 16 countries agreed to participate, and 86 followed the study design. Very high survival and success rates were documented in this field trial. The cumulative survival rate was 99.56 % at 3 years and 99.26 % at 5 years. The overall success rate was 99.12 % at 3 years and 97.38 % after 5 years. Typical of many studies, the majority of lost implants occurred as early failures, with only 1 implant lost as a late failure. Additionally, the implant complications were typical and included inflammation, infection, and discomfort of a few implants on yearly recall. Thus, although many variables were uncontrolled, the predictability of the procedure with the SLA surfaced implant was not affected compared to rates in a carefully controlled, prospective, multicenter human clinical trial [71] using the same implant.
A retrospective study conducted by Buser et al. [5] assessed the 10-year outcomes of 511 titanium implants with the SLA surface in 303 partially edentulous patients. Over the 10-year period, no implant fracture was noted, whereas 6 implants (1.2 %) were lost. Two implants (0.4 %) showed signs of suppuration at the 10-year examination, whereas 7 implants had a history of peri-implantitis (1.4 %) during the 10-year period but presented healthy peri-implant soft tissues at the final examination. This retrospective analysis resulted in a 10-year implant survival rate of 98.8 % and a success rate of 97.0 %. In addition, the prevalence of peri-implantitis in this large cohort of orally healthy patients was low with 1.8 % during the 10-year period.
As periodontitis is one of the main reasons for tooth loss, Roccuzzo et al. [88] compared the long-term outcomes of SLA implants in patients previously treated for periodontitis and in periodontally healthy patients (PHP). After clinical measurement of the pocket depths at 4 sites per tooth, patients with an initial diagnosis of periodontitis were classified according to a special score, which was calculated according to a defined formula: S = number of pockets (5–7 mm) +2× (number of pockets (≥8 mm)). Patients with a score >25 were considered as severe periodontally compromised patients (PCP). The results showed that at the 10-year follow-up, the implant survival rate was 100 % for PHP and 97.1 % even for severe PCP. They pointed out that SLA implants, placed under a strict periodontal control, offer predictable long-term results and patients should be informed, from the beginning, of the value of the supportive periodontal therapy (SPT) in enhancing long-term outcomes of implant therapy, particularly those affected by periodontitis.
Besides partially edentulous patients, Fischer et al. [89] also evaluated the totally edentulous maxilla of 24 patients with SLA surfaced implants and found that after a 10-year follow-up, the implant survival rate was 95.1 %, with a 1.07 mm mean value of bone loss. This confirmed the prospective study above that evaluated implants that had been restored in half the conventional healing time over a period of 5 years, and the life table analyses demonstrated a 99.1 % survival rate and a 98.8 % success rate for the SLA surfaced implants in a formal, carefully and externally monitored, multicenter, multinational clinical study [90]. A further clinical study conducted by Dam [91] evaluated the crestal bone loss (CBL) around dental implants by measuring radiographs after 5–6 years and found that SLA surfaced implants had less CBL than TPS implants. Lethaus et al. [92] conducted a prospective study to examine the long-term outcome of early loading of SLA implants in the edentulous mandible and reported a 5-year cumulative success rate of 96.7 %, with a 0.77 mm mean loss of crestal bone height. In a clinical follow-up report, Luongo et al. [81] presented favorable 3-year results of a prospective multicenter study with healing times between 0 and 11 days. In particular, Baker and coworkers [93] suggested that the early integration strength of SLA implants could be particularly advantageous in single-stage surgical protocols.
SLA: Summary of Clinical Studies
Taken together, these studies demonstrated that SLA surfaced titanium implants have a high survival and success rate and a very small number of implant failures occur during follow-up. Implant failures can be divided into early failures and late failures. Early failures are those occurring prior to and at the time of restoration, while late failures are those occurring after restoration [55]. Many studies have shown that the majority of implant failures occur as early failures during the healing period, and very few implant losses are observed after the onset of loading or during the follow-up period [71, 87, 92, 94]. Implant failure is to be expected in patients exhibiting risk factors such as systemic diseases causing wound-healing problems, heavy smoking, increased periodontal susceptibility, and anatomic factors such as poor bone density or extreme atrophy [95]. In spite of these many variables during the healing time and loading phase time, the predictability of the SLA surfaced implants in the listed studies was not affected. This suggests that the preclinical findings of large amounts of bone-to-implant contact and high interfacial strength with SLA implants have long-term advantages in patients.
In conclusion, these clinical studies of early loaded SLA implants reinforce that, under well-controlled clinical conditions and in less well-controlled private practice conditions, using a variety of indications, SLA implants can achieve very high success rates when abutments are torqued to 35 Ncm without counter torque at a healing time of 6 weeks. Furthermore, the osseointegration also allows the SLA surfaced implants to be highly successful in the longer term, with 10-year follow-up in some studies with very few late-term failures and very few implants with peri-implantitis.
ModSLA Implant Surfaces
Physical and Chemical Properties
As detailed above, titanium implants, intended for placement in osseous host tissue, can have different physical surface properties that are defined by the manufacturing process of the implants. Thus, titanium implants with different surface topographies, like relatively smooth surfaces created by the machining process or turning of the metal rod or a moderately rough surface topography [96] after sandblasting and acid-etching, can be produced [3]. The type of surface topography directly influences bone apposition, since it has been demonstrated that roughening of the implant’s surface up to a certain degree induced significantly increased osseous integration [3, 4]. Besides different physical properties, titanium surfaces can also show different chemical properties, defined by the atoms and molecules that are attached to the implant surface (chemical composition). Thus, the chemical properties of the titanium implant-bone complex are not defined only by the material itself but also by the dioxide layer that is automatically and immediately formed on the titanium implant surface when exposed to air if the surface atmosphere is not controlled and/or protected [97]. It has been stated that “It is thus a combination of the microarchitecture and the chemical composition of the surface that is expected to determine the overall, mutual interaction between implant and biological system” [98]. In this context, it is important to know that the chemical properties of the implant surface directly influence surface wettability and surface free energy [99] and that an increased surface free energy can further promote the interaction between the implant surface and the aqueous peri-implant biologic environment [100, 101]. Moreover, it has been shown that an increased surface free energy was correlated with increased surface hydrophilicity [12]. Pure titanium dioxide surfaces show a high initial hydrophilicity, since the oxide layer that is formed on the surface (as noted above) is intrinsically hydrophilic and immediately hydroxylated under room temperature and when aqueous solutions or its vapors have contact to the surface. Those binding OH groups are amphoteric in character as they can react as an acid or a base [102]. Unfortunately, when exposed to air, this clean and initial hydrophilic TiO2 surface is contaminated with hydrocarbons and carbons from the atmosphere and air, leading to a hydrophobic character and a reduced surface wettability and surface free energy [20].
Many experimental studies have demonstrated that the sandblasted, large-grit, and acid-etched titanium (commercially pure titanium (c.p. Ti)) implant surface (SLA®, Institut Straumann AG, Basel, Switzerland) is highly osteoconductive [16, 50], but as a possibly restrictive feature, it has a hydrophobic character [12]. As described above, native titanium dioxide surfaces are initially hydrophilic and chemically reactive. One goal for further development of the c.p. Ti-SLA surface was to fabricate a surface that has a hydrophilic character like in its native, uncontaminated state. In this context, a modified SLA surface with a high hydrophilicity and surface free energy has been developed and established on the market (SLActive®, Institut Straumann AG, Basel, Switzerland). The modified SLA surface is similarly produced like SLA, but after the same sandblasting and acid-etching procedure, the titanium implants (or experimental disks) are rinsed under N2 protection and directly stored in isotonic NaCl solution at pH 4–6, again protected by N2 filling [2, 11, 12]. Qualitative examinations using scanning electron microscopy (SEM) and quantitative evaluations by confocal white-light microscopy to calculate three-dimensional roughness parameters [2, 11] showed no differences between both types of surfaces concerning surface topography and surface roughness parameters (Fig. 9.3a, Table 9.1). However, more detailed SEM investigations of both surfaces clearly identified nanostructures only on the modSLA surface, leading to a significantly increased surface development on modSLA compared to SLA surfaces [103].
When investigating surface wettability and surface free energy, significant differences have been found between modSLA and SLA surfaces. Rupp et al. investigated the surface hydrophilicity and the surface wettability from both surfaces by dynamic contact angle analysis (DCA) and determined the surface free energy [12]. The main outcome of these evaluations was that modSLA showed significantly increased initial hydrophilicity and a complete wettability documented by water contact angles of 0°, compared to SLA that was rather hydrophobic with initial contact angles of 139.9° (Fig. 9.4). Furthermore, the authors demonstrated that an “increased initial hydrophilicity of modSLA could be clearly correlated with increased surface free energy” [12]. Analogous results could also be demonstrated from different authors using similar methods [2, 11, 103].


Fig. 9.4
Drop of water on hydrophobic SLA surface (a) and on hydrophilic modSLA surface (b). Increased hydrophilicity on modSLA surface indicated by complete dispersion of waterdrop (b, no measurable contact angle between water drop and surface); however, no dispersion of waterdrop on hydrophobic SLA surface (a, obvious measurable contact angle between waterdrop and surface)
The increased surface hydrophilicity can also induce an accelerated covering of the implant surface with blood from the peri-implant soft and bone tissue during placement of the implant (Fig. 9.5), compared to hydrophobic SLA surfaces, thus accelerating the initial implant healing period.


Fig. 9.5
Accelerated covering of the implant surface with blood on implant with hydrophilic modSLA surface (b) compared to implant with hydrophobic SLA surface (a)
Additionally, by using x-ray photoelectron spectroscopy (XPS) to investigate the chemical composition of both surfaces, it could be evaluated that modSLA showed increased oxygen and titanium concentrations and reduced carbon concentrations compared to SLA (Table 9.2). Before analysis, the modSLA surfaces were taken from the containment ampule, rinsed with ultrapure water, and dried with N2 to remove electrostatically bound ions from the surface. The results indicated an increased concentration of hydroxylated groups bound to the modSLA surface in comparison to SLA and a decreased adsorption of potential contaminants (carbons, hydrocarbons) from the atmosphere [11, 12, 103].
Table 9.2
Chemical surface composition [12]
|
Chemical composition [%]
|
||
|---|---|---|
|
Element
|
SLA
|
modSLA
|
|
O
|
49.2 ± 2.1
|
61.1 ± 0.9
|
|
Ti
|
14.3 ± 1.3
|
22.5 ± 0.9
|
|
N
|
1.3 ± 0.4
|
0.7 ± 0.3
|
|
C
|
35.2 ± 2.2
|
14.2 ± 1.2
|
Another study evaluated the chemical composition of the modSLA surface as received from the manufacturer without rinsing the implant surface with ultrapure water prior to XPS analysis. The results showed similar values for C (16.8 %) and N (1.1 %) but slightly different values for Ti (11.6 %) and O (28.6 %). Furthermore and expectedly, they could detect Na (25.2 %) and Cl (16.1 %) ions indicating a surface coating with NaCl crystals due to the storage of the implant in NaCl solution [104]. After drying up of the solution, the water evaporated but the NaCl that was in the solution remained on the implant surface.
Besides commercially pure titanium, also titanium alloys were used for the production of dental implants with a sandblasted and acid-etched surface with or without high surface free energy. However, due to the chemical composition of the implants, not every alloy can be sandblasted and acid-etched like c.p. Ti. For example, it has been shown that acid-etching of the alloys Ti-6Al-4V and Ti-6Al-7Nb resulted in different surface microstructures than acid-etching of c.p. titanium [105]. These differences were related to the different crystal structures of c.p. titanium and these 2 alloys. At room temperature and pressure, c.p. titanium shows a hexagonal closed-packed (α) crystal structure, while at high temperatures, it adopts a body-centered cubic (β) structure [106]. The addition of Al stabilizes the α structure, while addition of Nb or V stabilizes the β structure [105]. In the case of these 2 alloys, the etching induced a selective dissolution of the α-phase and an enrichment of the β-phase, resulting in different surface microstructures compared to c.p. titanium [105]. Recently, a new titanium-zirconium (TiZr) alloy with a modSLA surface has been established on the market (Roxolid® SLActive®, Institut Straumann AG, Basel, Switzerland) that consists of titanium and 13–17 % zirconium [13, 107]. In comparison to c.p. titanium, it has been found that titanium-zirconium alloys show superior biomechanical properties, identified by increased hardness and tensile strength [108]. Since titanium, zirconium, as well as the TiZr alloy show the α-phase crystal structure at room temperature and pressure [106], the manufacturing procedure (sandblasting and acid-etching, rinsing under N2 protection, storage in isotonic NaCl solution) for creating the modSLA surface on c.p titanium can be transferred to this particular TiZr alloy [107]. Thus, one specific advantage of this TiZr alloy is its increased biomechanical strength compared to c.p. titanium combined with the ability to create a modSLA surface topography.
SEM investigations on both materials show a similar micro-rough surface topography (Fig. 9.3). When investigating surface roughness parameters by performing quantitative surface analysis using confocal white-light microscopy (Table 9.3, [109]) and blue light laser and interferometer [107], significant differences have been observed for the number of samples measured. However, the c.p. Ti and the TiZr-modSLA surface both have similar characteristic roughness parameters between 1 and 2 μm (e.g., both surfaces are moderately rough [96]).
Table 9.3
Roughness parameters for modSLA surfaces [109]
|
Roughness parameter
|
c.p. titanium
|
TiZi
|
|---|---|---|
|
Sa [μm]
|
1.00 ± 0.02
|
1.30 ± 0.09
|
|
St [μm]
|
6.73 ± 0.18
|
8.90 ± 0.36
|
|
SSk [μm]
|
0.14 ± 0.03
|
0.21 ± 0.06
|
|
Sdr [%]
|
29.6 ± 1.2
|
39.0 ± 3.3
|
When investigating the chemical composition of the modSLA surfaces of both materials, no significant differences were observed. On the modSLA surface of TiZr, additional amounts of aluminum and zirconium were detected, and besides TiO2 molecules, also ZrO2 molecules were found (Table 9.4) [107]. The presence of Al can be explained by Al2O3 residuals due to the sandblasting process [103].
Table 9.4
Chemical surface composition of modSLA surfaces [107]
|
Chemical composition [%]
|
||
|---|---|---|
|
Element
|
c.p. titanium
|
TiZr
|
|
O
|
52.20 ± 1.7
|
50.87 ± 2.7
|
|
Ti
|
21.11 ± 1.2
|
17.94 ± 1.4
|
|
N
|
1.21 ± 0.4
|
0.80 ± 0.1
|
|
C
|
24.79 ± 3.1
|
24.63 ± 4.5
|
|
F
|
0.83 ± 0.6
|
0.61 ± 0.2
|
|
Al
|
–
|
2.56 ± 0.0
|
|
Zr
|
–
|
2.79 ± 0.2
|
These results suggest that manufacturing a sandblasted and acid-etched surface with high surface free energy is not specific for one certain material, as the manufacturing process can be applied on commercially pure titanium as well as on this particular titanium-zirconium alloy, creating a similar moderately rough surface topography with an analogue chemical surface composition and similar surface roughness parameters.
ModSLA: Summary of Physical and Chemical Properties
In summary, due to the same sandblasting and acid-etching procedure, there were no differences in surface topography or surface roughness parameters between SLA and modSLA surfaces; however, nanostructures could only be found on the modSLA surface. Due to the different rinsing and storage procedures, differences were found in the chemical surface composition of the surfaces. In comparison to SLA, after rinsing with ultrapure water, the modSLA surfaces showed decreased adsorption of contaminants from the atmosphere and an increased concentration of hydroxylated groups on the surface, thus resulting in an increased surface hydrophilicity, surface wettability, and surface free energy. This is a result of the shared chemical bonding of the oxygen molecules from the native dioxide surface. Furthermore, the modSLA surface can be created on c.p. Ti as well as on a specific TiZr alloy, resulting in similar moderately rough microsurface topographies, with an analogue chemical surface composition and similar surface roughness parameters.
In Vitro Studies
In vitro investigations on cell-implant interactions provide valuable and detailed information about single cells and factors that affect the initial osseointegration process at the implant-bone interface. Therefore, it has been concluded that an “in vitro approach provides a powerful tool to elucidate the detailed biological events that take place at such an interface and has permitted the compositional and structural characteristics of the interface to be defined” [110]. Previous in vitro investigations have demonstrated that an increased surface roughness decreased cell proliferation (identified by a decreased number of cells on the surface) and increased cell attachment and differentiation of osteoblast-like cells that were cultured on titanium disks [15, 39, 41]. Due to the increased amount of hydroxylated groups bound to the dioxide layer (see above), the modified SLA surface is chemically more reactive than the SLA surface. Thus, it could be hypothesized that not only surface roughness but also surface free energy can directly influence cellular behavior and growth factor or cytokine production of cells, which are involved in the osseointegration process. One part of the goal of this review was to present current in vitro studies that investigated single cells and factors, which are involved in the implant healing process. Most findings on the modSLA surface were directly compared to findings on the SLA surface so that only differences due to the change in surface chemistry could be evaluated.
ModSLA: Bone Tissue Healing and Remodeling
The first part of implant healing consists of the recruitment and the migration of osteogenetic cells to the implant surface through the fibrin network of the peri-implant blood clot. This process is mainly initiated by platelet activation (release of cytokines, growth factors) within the first 3 days of healing [32, 35]. In this context, it was demonstrated that whole blood collected from human patients in direct contact with modSLA surfaces revealed significantly increased degrees of platelet binding, platelet activation, and intrinsic coagulation system activation, when compared to SLA surfaces [111]. The activated platelets not only stimulate factors that enhance osteogenetic cells but also release factors that activate leukocytes and macrophages (between 24 and 48 h after implant placement), which are involved in the demolition of the peri-implant blood clot [35], thus probably delaying the healing process. An attenuated pro-inflammatory response of the activated macrophages that might indicate a faster and improved wound healing was identified on modSLA compared to SLA. Whereas there were no significant differences in cellular macrophage attachment or proliferation on both surfaces, differences were found concerning the regulation of pro-inflammatory genes for cytokines and chemokines. Surface roughness induced activation of pro-inflammatory genes on both surfaces, but in contrast to SLA, on modSLA the expression of many genes was also significantly downregulated after 24 h and 3 days of investigation [112, 113].
Following recruitment and migration, osteogenetic cells attach to an implant surface and begin to proliferate [32, 35]. When investigating the attachment of MG63 osteoblast-like cells by seeding a defined number of cells on modSLA and SLA surfaces and evaluating the number of attached cells after 4 h of incubation, no differences in cell attachment levels could be evaluated between both surfaces [114]. The same results were found after cell attachment analysis of human mesenchymal stem cells and human mesenchymal stromal cells after 3 h of incubation [115, 116] and for human periodontal ligament cells after 5 h of incubation [117]. In contrast to that, when evaluating the number of attached human osteoblast cells, the modSLA surface demonstrated significantly increased cell attachment rates after 1 and 3 h of seeding in comparison to SLA [118]. Furthermore, integrin β1 and αV gene expression analysis of human osteoblast indicated a higher initial cell adhesion rate, and a strongly enhanced gene expression after 24 and 48 h was demonstrated on modSLA surfaces in comparison to SLA surfaces [119]. Thus, the increased attachment effect on modSLA implant surfaces appears to be cell specific for human osteoblasts.
With regard to cell proliferation, previous studies have shown that an increased surface roughness decreased the proliferation of MG63 osteoblast-like cells [15, 41]. When investigating these bone-producing cells, a higher surface free energy and wettability induced a further significant decrease of cell proliferation on modSLA in comparison to SLA [2, 114, 120–123]. Moreover, it was shown that the addition of 10−9 M calcitriol (1α,25(OH)2D3) further significantly decreased the cell number on SLA, whereas no effects were noticed on modSLA [2]. Cell proliferation was also investigated using human osteoblast cells. The results again demonstrated a significantly reduced cell proliferation on modSLA compared to SLA surfaces [123, 124]. In general, cell number is decreased when cells begin to differentiate, indicating a change from a mainly proliferative to a more differentiated cell state [125, 126]. Thus, with regard to new bone formation, cell differentiation can be considered as a more decisive factor than initial cell proliferation.
Initial cell morphology has additionally been investigated on modSLA and SLA surfaces. It was shown that there were no significant differences between single MG63 osteoblast-like cells grown on modSLA or SLA surfaces. The shape of the cells was described as polygonal, elongated with many thin filopodia, attached to the surface [2, 118, 120]. By using time-lapse microscopy, Qu et al. demonstrated that MG63 osteoblast-like cells on modSLA surfaces began to form clusters as a precursor for noduli formation after 2 days of incubation, whereas cells on SLA still spread homogenously [114]. After 4 days of incubation, cells also began to form clusters on SLA, and after 9 days, modSLA still showed larger cell clusters compared to SLA [114]. When investigating human osteoblasts, minor morphological differences were found. After 6 h of seeding, human osteoblasts on modSLA showed increased spreading behavior by forming better-defined actin stress fibers in comparison to SLA [118]. After 24 h incubation, the cells on SLA had a more elongated structure, whereas osteoblasts on modSLA were smaller and rather roundish [119]. These studies suggest that bone-forming cells are in general stimulated to differentiate earlier on modSLA surfaces compared to the hydrophobic SLA surface and that this effect is cell type dependent.
Within the implant healing process, after recruitment, attachment, and proliferation, the cells undergo osteoblastic differentiation by producing osteoid, including matrix vesicles and growth factors (3–6 days). After that, the cells begin to calcify their matrix, indicated by increased alkaline phosphatase and phospholipase A2 activity (6–14 days) [32, 35]. In this context, more differentiated and more active MG63-, MC3T3-E1 osteoblast-like cells, human osteoblast cells, human mesenchymal stem cells, and human periodontal ligament cells on modSLA in comparison to SLA have been reported, indicated by significant increases of factors that stimulate early and late osteogenic differentiation, like cell layer alkaline phosphatase (ALP), osteoprotegerin (OPG), type I collagen, osteopontin (OPN), and osteocalcin (OC) [2, 114, 117, 120, 121, 123, 125, 127, 128]. In addition to that, by producing significantly higher levels of local factors like prostaglandin E2 (PGE2), transforming growth factor-β1 (TGF-β1), phospholipase D, and protein kinase C (PKC), the cells seeded on modSLA surfaces also created a more osteogenic microenvironment when compared to cells seeded on SLA surfaces [2, 120, 121, 123]. In contrast to cell number, the addition of calcitriol (1α,25(OH)2D3) caused further remarkable increases in the amount of ALP, OC, PGE2, TGF-β1, and vascular endothelial growth factor-A (VEGF-A) production [2, 123]. Additionally, it has been shown that the differentiation of human mesenchymal stem cells into an osteoblastic phenotype was not only sensitive to surface microstructure but also to surface hydrophilicity [125]. Again, these results clearly indicate that the bone-forming cells begin to differentiate earlier and even more distinctively on modSLA compared to the SLA surface. This particular effect seems to be independent from the cell type.
After a cell differentiation period around inserted implants, the newly formed woven bone begins to be remodeled (21 days after initial implant placement), and this involves recruitment of osteoclasts that resorb the newly formed bone tissue [32, 35]. Osteoclasts are activated by a protein that is called receptor activator of nuclear factor kappa-B ligand (RANKL). This protein is produced by fibroblasts, T lymphocytes, B lymphocytes, and also osteoblasts and binds to the RANK receptor on the surface of mature osteoclasts or osteoclast precursors [129, 130]. Another protein called OPG is also produced by osteoblasts. It inhibits the differentiation of osteoclasts by blocking RANKL binding to its cellular receptor RANK [129]. Thus, bone formation occurs when the RANKL/OPG ratio decreases, whereas bone resorption occurs when the RANKL/OPG ratio increases [130]. It has been shown that the modSLA surface could directly influence the RANKL-RANK-OPG axis. An in vitro study investigated the mRNA gene expression for OPG and RANKL of human periodontal ligament cells that were seeded on modSLA and SLA surfaces [117]. After 5, 24 h, 3, and 5 days of investigation, no differences could be detected between both types of surfaces, indicating that bone cells are selectively activated on implant surfaces. Though, after 7 days of seeding, the authors demonstrated a significantly increased OPG gene expression and a significantly decreased RANKL gene expression on modSLA surfaces in comparison to SLA surfaces [117]. In addition to that, mouse bone marrow-derived macrophages showed a significantly reduced gene expression of osteoclastogenesis-related genes (TRAP, NFATc1, OSCAR, c-Fos) on modSLA surfaces compared to SLA surfaces [127]. Mature dendritic cells can also initiate T-cell activation and therefore influence the differentiation of osteoclasts [131]. It has been demonstrated that dendritic cells seeded on SLA surfaces promoted a more mature phenotype (based on surface marker expression, cytokine profiles, and cell morphology), whereas dendritic cells that were cultivated on modSLA surfaces promoted an inactive, immature phenotype that might indicate a noninflammatory biological peri-implant environment enhancing peri-implant bone formation [131]. These results suggest that the differentiation of osteoclasts is suppressed on modSLA surfaces, thus leading to decreased bone resorption and a further increased bone formation on modSLA implant surfaces in comparison to SLA surfaces within the bone remodeling processes.
ModSLA: Angiogenesis and Neovascularization
Besides osteoblastic and osteoclastic differentiation, further important factors that influence a successful osseointegration are peri-implant angiogenesis and neovascularization processes, predominantly initiated by endothelial cells but also involving other cell types [35, 132]. MG63 osteoblast-like cells seeded on modSLA implant surfaces demonstrated significantly increased production of growth factors that serve to initiate and control angiogenesis (VEGF-A, basic fibroblast growth factor (FGF-2), epidermal growth factor (EGF)) compared to cells seeded on SLA surfaces. Furthermore, these growth factors induced a significantly increased differentiation of human aortic endothelial cells (HAEC, indicated by endothelial tube formation and number of branch points) on modSLA surfaces compared to SLA surfaces. When investigating human osteoblasts, only significantly increased amounts of VEGF-A on modSLA in comparison to SLA surfaces could be demonstrated, whereas the increased amounts of other growth factors (see above) could not be detected [124]. Rausch-Fan et al. confirmed the significantly increased VEGF-A production of MG63 osteoblast-like cells and human osteoblasts on modSLA compared to SLA surfaces [123]. When investigating endothelial progenitor cells from goats, a remarkable increase of VEGF-A production was detected on modSLA compared to SLA surfaces; however, the results were not statistically significant [133]. In addition to that, human umbilical vascular endothelial cells (HUVECs) grown on modSLA demonstrated a significantly increased gene expression of angiogenesis-related factors (von Willebrand factor, thrombomodulin, endothelial cell protein C receptor) when compared to cells grown on SLA surfaces [134]. These results indicate that bone-forming cells as well as endothelial cells might provoke an earlier angiogenesis and an increased neovascularization on modSLA compared to SLA surfaces, thus probably enhancing the early peri-implant healing process around these hydrophilic implant surfaces.
ModSLA: Soft Tissue Healing
Not only bone tissue is a decisive factor for a successful implant healing process but also the peri-implant soft tissues [135] are important. In contrast to MG63 osteoblast-like cells and human osteoblasts, when investigating epithelial cells (oral squamous carcinoma cell line), an increase in initial cell attachment, proliferation rate, and cell spreading could be demonstrated on modSLA compared to SLA surfaces, but no differences in gene expression of functional factors that influence cytokine secretion were detected [136]. In addition to that, also human periodontal ligament cells seeded on modSLA surfaces demonstrated significantly increased proliferation rates after 24 h, 5, and 7 of days of incubation compared to cells seeded on SLA surfaces [117]. Thus, this increase in cell proliferation rate appears to be specific for non-bone-producing cells and may stimulate a more rapid soft tissue healing around implants with a modSLA surface.
ModSLA: Summary of In Vitro Studies
In summary, the results of in vitro studies demonstrate that modSLA surfaces in comparison to SLA surfaces increase the recruitment and migration of osteogenic cells to the implant surface in the very early healing period, indicated by increased platelet activation and an attenuated pro-inflammatory response of activated macrophages. After migration, only human osteoblasts showed a significantly increased cell attachment rate on modSLA compared to SLA surfaces within the first 48 h of investigation demonstrating a preferential selectioning of cell types for modSLA. Not only surface roughness but also an increased surface free energy induced a further significant decrease in the number of proliferated MG63 osteoblast-like cells and human osteoblasts on modSLA compared to SLA surfaces indicating that cells stop proliferation and rather increase differentiation, thus preparing for new bone. Only the proliferation of soft tissue cells (human periodontal ligament fibroblasts) was significantly increased on modSLA compared to SLA surfaces. Concerning cell morphology, no differences were observed between both types of surfaces; however, MG63 osteoblast-like and human osteoblast cells on modSLA surfaces showed a faster initial cell spreading behavior compared to cells seeded on SLA surfaces. After attachment and proliferation, all investigated types of osteogenic-related cells were significantly more differentiated and more active and created a more osteogenic environment on the modSLA surfaces compared to cells that were incubated on the SLA surfaces. In addition to that, modSLA surfaces promoted bone formation within the bone remodeling process by decreasing the RANKL/OPG ratio, thus inhibiting the differentiation of osteoclasts. Furthermore, in comparison to SLA, MG63 osteoblast-like cells and human osteoblasts seeded on modSLA surfaces also demonstrated significantly increased productions of growth factors that initiate and control angiogenesis and enhance the differentiation of endothelial cells that initiate neovascularization (Fig. 9.6). Besides osteogenic-related cells, increased surface free energy also directly influenced other soft tissue cells (epithelial cells) by increasing epithelial attachment, proliferation, and cell spreading on modSLA compared to SLA surfaces. Thus, the chemically active hydrophilic modSLA surface compared to the hydrophobic SLA surface promotes bone cell attachment and differentiation, blood vessel formation, and soft tissue proliferation.
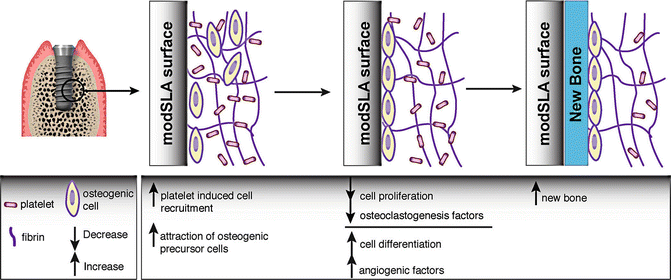

Fig. 9.6
Schematic diagram of enhanced peri-implant bone healing around modSLA implant surface
In Vivo Studies
As osseointegration is a very complex process that involves a wide variety of different types of cells and in vitro protocols only allow investigating interactions of single cell types (e.g., osteoblasts), controlled in vivo models are clinically most relevant to study the osseous integration and culmination of cellular interactions of dental implants [52]. In addition, as implant restorations in the oral cavity always have to penetrate the soft tissues, the implant restoration-gingival tissues should provide an effective barrier function similar to dentogingival tissues to ensure the integrity of the integument. Based on this fact, it has been demonstrated that nonsubmerged dental implants must successfully integrate with bone tissue as well as with connective tissue and epithelium at the time of implant placement [135, 137]. The following section presents preclinical in vivo studies investigating peri-implant bone integration and defect regeneration, as well as soft tissue integration around c.p. titanium and TiZr alloy implants with a modSLA surface that were performed using different animal models and different examination methods.
ModSLA: Bone Tissue Integration
Concerning bone tissue integration, it has been reported that implants with a modSLA surface showed a faster osseous healing within the initial osseointegration process in comparison to SLA surfaces [11, 52, 66, 132, 138–141]. Qualitative histological findings of preclinical studies performed in canines [132, 138
Stay updated, free dental videos. Join our Telegram channel

VIDEdental - Online dental courses


