6. Describe the bacterial elements of the biofilm community that contribute to dental caries.
S. mutans and Lactobacillus are the dominant species.
• Aciduric—survive in an acid environment
• Acidogenic—produce higher amounts of acid from sugars
• Produce extracellular polysaccharides to thicken plaque and aid in adherence within the biofilm
• Use high levels of adenosine triphosphate (ATP) to maintain an intracellular neutral pH and pump out acids
7. What is generally accepted as a new standard of dental care for preventing dental disease and managing a patient’s risk?
According to the American Dental Society and American Academy of Pediatric Dentistry, an individual caries risk assessment analysis should be part of a complete dental examination. Disease indicators and risk factors will determine the probability that a specific patient will become susceptible to dental caries, and early intervention therapies may be implemented.
8. What does the acronym CAMBRA stand for, and what is its value in caries prevention?
CAMBRA stands for caries management by risk assessment. Using a written form or oral interview, specific elements that could lead to a bacterial imbalance of dental pathogens can be assessed. Dental treatments and restoration recommendations are formulated from this data.
9. List the elements of CAMBRA.
• Current decay levels (number of decayed teeth)
• Current bacterial challenge (measured by testing levels)
• Decay history (e.g., decay-missing-filled [DMF] index, missing filled teeth)
• Dietary habits (food types, degree of snacking)
• Current medications
• Saliva status (e.g., amount, buffering capacity)
• Medical conditions
• Oral appliances present (e.g., braces)
• Oral hygiene habits
10. How does ATP testing determine if someone is at risk for dental caries?
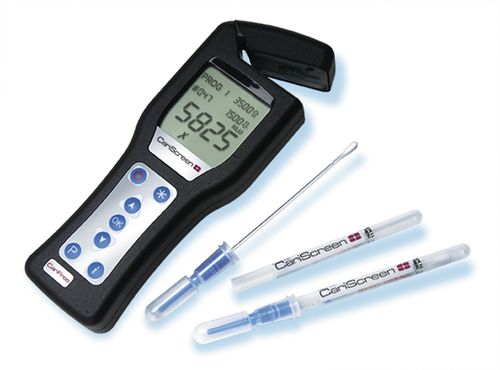
ATP is the basic energy molecule in all living cells. The caries-linked bacteria S. mutans and lactobacilli produce large quantities of acid during sugar metabolism and use ATP to pump this metabolite out of their intracellular space. By assaying the amount of ATP, a correlation can be made to the quantity of bacteria present. By mixing rather simple reagents, a bioluminescence assay for light produced can be correlated with quantities of microorganisms.
Taking an oral swab and placing it in an ampule containing luciferin and luciferase (the firefly enzyme), the following reaction occurs:
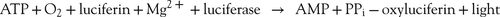
The ampule is placed into a sensitive light meter, and the numeric value obtained correlates with the bacterial population, indicating at-risk levels. See www.carifree.com and the CariScreen (Fig. 8-2).
11. What is the cariogram?
Developed in Sweden, the cariogram is a simple software program that generates a pie graph representation of the elements of a risk assessment survey (Fig. 8-3).
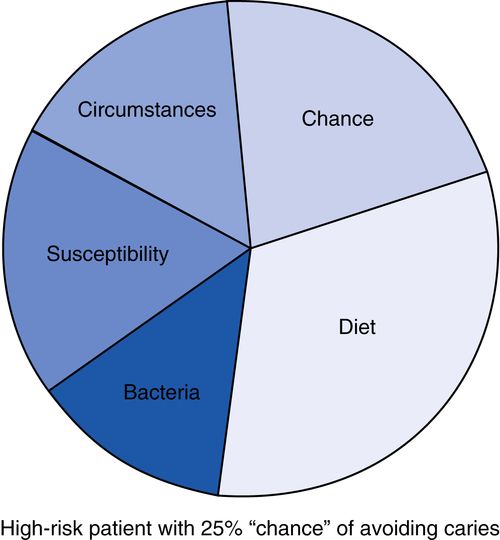
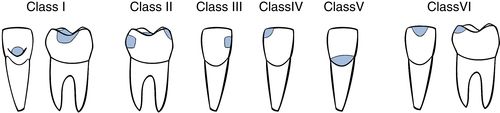
Smooth surface: interproximal: class II posterior, class III anterior
Cervical and root surface: class V
2. Rate of progression—incipient (early enamel surface or white spot lesion)
Acute or rampant (rapid progression and widespread)
Chronic or arrested (demineralization has ceased)
Recurrent (new decay at sites of previous lesions or restorations)
3. Hard tissue involved—enamel, dentin, cementum
4. Cause—radiation caries, early childhood caries (ECC), baby bottle caries, xerostomia
5. Caries (reduced or absent saliva)
The ICDAS (International Caries and Detection System) has standardized the classification system (www.icdas.org). The GV Black classification system is illustrated in Figure 8-4.
13. Outline the caries susceptibility of teeth in the dental arches.
Maxillary > mandibular arch
Tooth type: first molars (upper and lower) > second molars (upper and lower) > second bicuspids (upper) > first bicuspids (upper) and second bicuspids (lower) > central and lateral incisors (upper) > canine (upper) and first bicuspids (lower) > lower anteriors
Tooth surface: occlusal >> mesial > distal > buccal > lingual
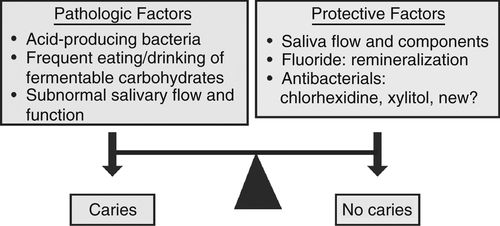
14. List the key risk factors associated with dental caries.
• Susceptible tooth surface that can maintain plaque and bacteria
• Presence of acidogenic bacteria
• Steady supply of dietary fermentable carbohydrates
• Inadequate salivary flow or buffering capacity
• Low exposure to topical or dietary fluoride
15. What organisms are responsible for caries formation?
S. mutans and lactobacilli are the most cariogenic, with contributions from Streptococcus sanguis and Streptococcus salivarius. These organisms metabolize sucrose to form acidic byproducts destructive to enamel surfaces. Root surface caries are initiated by Actinomyces viscus on accumulated plaque deposits.
16. What are the properties of cariogenic bacteria?
Specifically unique are their ability to survive at low pH and metabolize simple sugars to form acid byproducts. During the process, extracellular polysaccharides are produced, which aid in adhesion within the plaque biofilms.
17. What is the role of saliva in caries susceptibility?
• Adequate flow reduces plaque accumulation on tooth surfaces and rate of clearance of carbohydrates.
• Diffusion of the salivary components calcium, phosphate, hydroxyl, and fluoride ions into plaque can reduce the solubility of enamel and promote remineralization of early carious lesions.
• The bicarbonate-buffering ability of saliva can reduce or limit the fall in pH when bacteria metabolize sugars.
• Salivary proteins form the protective acquired pellicle, which retards the flow of ions out of enamel.
• The salivary components of secretory immunoglobulin A (IgA), lysosomes, lactoperoxidase, and lactoferrin have antibacterial activity.
18. Describe the role of fluoride in preventing dental caries.
• The anticaries effects of fluoride are primarily topical for children and adults.
• Fluoride inhibits demineralization at enamel crystal surfaces inside teeth.
• Fluoride enhances the remineralization of the enamel crystal surface after demineralization and increases acid resistance.
• The systemic benefits of fluoride are minimal.
19. What is the effect of fluoride on bacteria?
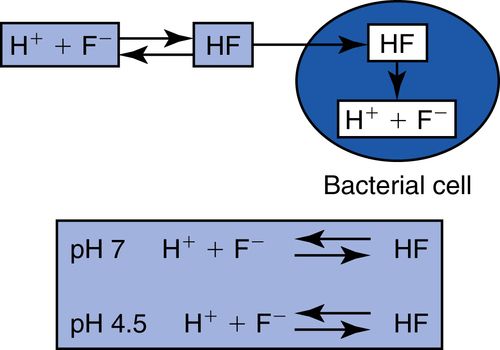
Fluoride can enter the bacterial cell only as hydrogen fluoride (HF), where it can dissociate and inhibit bacterial enzymes (enolases). As acid production proceeds, some H+ and F− ions form HF, which can enter the bacterial cell. At neutral pH, within the bacterial cell, it dissociates, and the fluoride ion is free to act on bacterial enzymes as an inhibitor (Fig. 8-5).
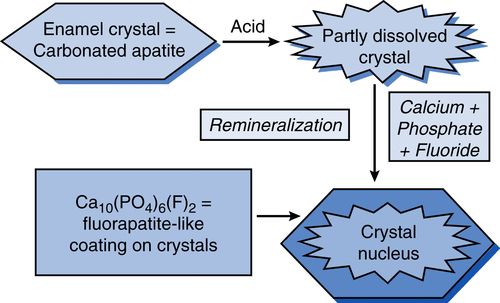
20. How dose fluoride aide in remineralization of tooth structure?
Fluoride present in saliva or plaque fluid will become incorporated into a new crystal surface during remineralization. It competitively displaces OH− ions and then acts topically, from the surface inward, exerting its effects against acid dissolution of the crystal surface. Present in plaque fluid, it will travel into the subsurface enamel with the acids, adsorb to the crystal, and protect it from dissolution. In summary, fluoride present in solution from topical sources enhances remineralization by speeding up the growth of a new surface on the partially demineralized subsurface crystals of the carious lesion. The new crystal surface is fluorapatite-like, with much lower solubility than the original carbonated apatite tooth mineral (Fig. 8-6).
21. Express the caries risk factors in a functional relationship.
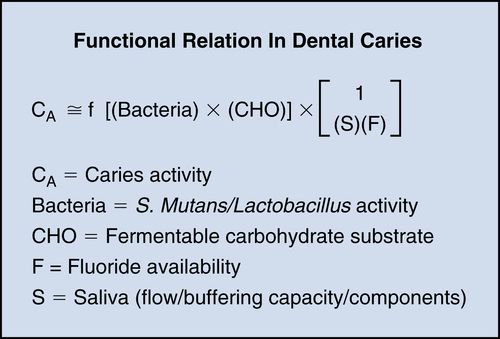
Dental caries occurs when the process of demineralization is faster than the process of remineralization and there is a net loss of tooth mineral into the environment. If the acid production is reduced by removing plaque accumulation or reducing dietary sugar substrates, tooth mineral dissociation will cease (decrease in caries activity [CA]). The presence of topical fluoride increases enamel resistance to dissolution (forming fluorapatite) and enhances remineralization. It also inhibits bacterial metabolism. Thus, increases in fluoride relate inversely to CA. Finally, a high salivary buffering capacity decreases caries activity, whereas low flow rates tend to increase CA.
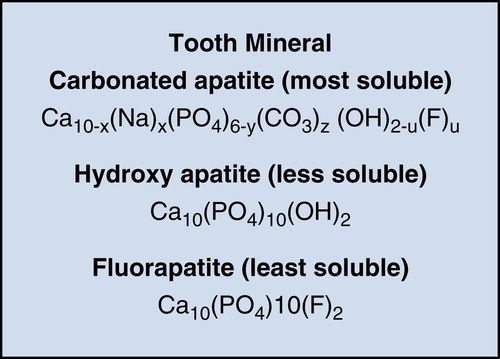
22. What are the three main tooth mineral complexes and their relative solubilities?
The enamel and dentin of a tooth are composed of tiny crystals embedded in a protein/lipid matrix. The mineral formed during tooth germination is a highly substituted carbonated apatite. It is related to hydroxyapatite but is more acid soluble, as well as calcium-deficient (replaced by sodium, magnesium, and zinc) and contains 3–6% carbonate replacing phosphate ions in the crystal lattice. During demineralization, carbonate is preferentially lost, and during remineralization, it is excluded and replaced by OH − or F− ions, thereby decreasing the acid solubility. This is the maturation cycle. Mature enamel is mostly hydroxy or fluorapatite.
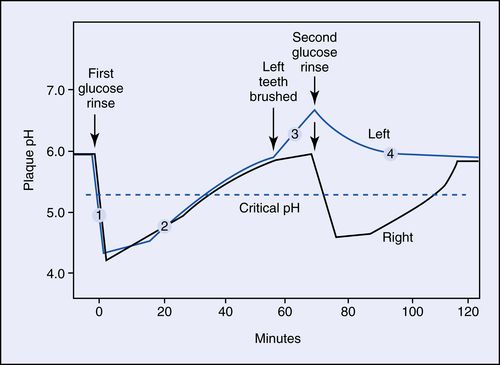
23. What is a Stephan plot?
The classic experimental measurement of pH changes on tooth enamel surfaces during exposure to fermentable carbohydrates in the presence of acidogenic bacteria (in plaque) over time is called a Stephan plot. It demonstrates the acid production of bacteria (pH decrease) with a glucose swallow, and the gradual rise caused by salivary buffering. At position #3, after left side brushing, the unbrushed side pH drops, whereas the left brushed side #4 remains above the critical pH (Fig. 8-7).
24. What is the critical pH for enamel dissociation?
The critical pH for enamel (hydroxyapatite) is 5.3 to 5.5; for fluorapatite, it is 4.5. Carbonated beverages (e.g., Coke, Pepsi) have a pH of about 3.5.
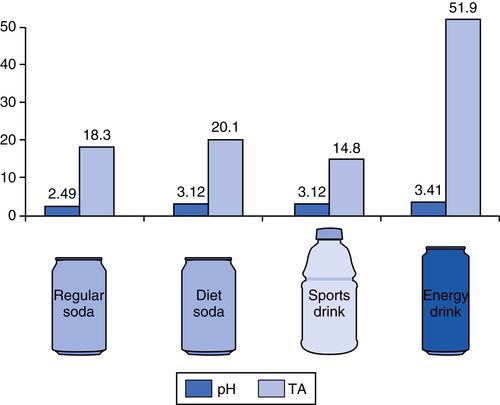
25. What are the pH values of some common beverages?
Knowing that the critical pH of enamel demineralization is 5.5, carbonated beverages can be very destructive (Fig. 8-8). The TA (titratable acidity) is the equivalent to titrate to neutral pH. Energy drinks (e.g., Red Bull) and sports drinks (e.g., Gatorade) showed the greatest degree of enamel surface dissolution. Possible explanations for the higher degree of enamel dissolution by sports and energy drinks is the addition of high concentrations of refined carbohydrates (e.g., sucrose, glucose) by the manufacturer, which promotes greater degrees of acid production and, in turn, higher buffering capacities. In addition, beverages containing citric acids have the ability to irreversibly chelate (bind) calcium at higher pH values, with the net effect of an accelerated loss of calcium from tooth structure; this maintains the pH of the beverage below the threshold level (pH = 5.5) for enamel erosion to occur. Red Bull contains sodium citrate (sodium salt of citric acid), a buffering agent that might help in maintaining the pH levels in soft drinks (Fig. 8-9).
26. What is the term for the earliest observable enamel caries lesion?
The term white spot lesion is applied to the earliest visually observable or macroscopic lesion in enamel. It represents dissolution of the surface structure, with increased porosity, and takes on a dull appearance when air-dried because of the differences in light scattering (refractive index) from the surrounding enamel. These lesions may maintain their surface integrity, become stained and arrested, or progress to frank cavitation lesions.
27. What are the differences in the mechanisms of enamel and dentinal caries?
Enamel caries is primarily an acidogenic or physiochemical progression of tooth mineral dissolution, whereas dentinal caries involves acid decalcification followed by proteolytic or enzymatic degeneration of the organic matrix.
28. What is the current thinking about treating early enamel caries or the incipient lesion?
Most early enamel lesions are capable of remineralization, or arresting, if the risk factors are reduced and there is adequate fluoride content in the microenvironment. Risk factor assessment (including diet, bacterial, and salivary analysis), followed by fluoride supplements from topical sources, is thus the first line of therapy.
29. What clinical tests are available to determine caries susceptibility?
Proprietary kits from Ivoclar Vivadent and Viadent provide the essential components to culture and grade levels of S. mutans and lactobacilli in saliva and measure salivary pH, flow rates, and buffering capacity. A rapid test by CariFree (www.carifree.com) is a simple, 1-minute chairside bacterial test for assessing patients’ caries risk. CariScreen uses ATP bioluminescence to identify oral bacterial load and has been proven to correlate with patients’ risk for decay. The test is quick and painless. A swab sample of the plaque is taken from the patient’s teeth, which, when combined with special bioluminescence reagents within the swab, creates a reaction that is measured with a meter. The CariScreen will give a score between 0 and 9999 relative light units (RLUs); a score under 1500 RLUs is considering relatively healthy, whereas a score above that shows considerable risk for decay.
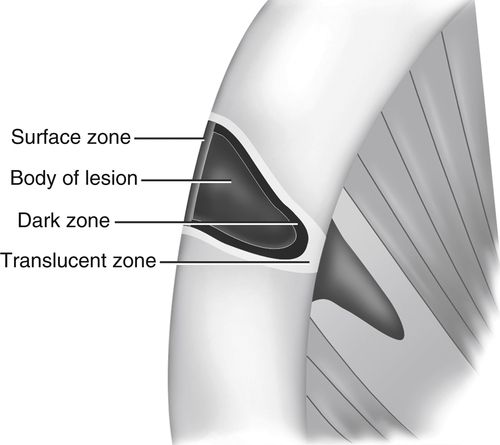
30. What are the histologic zones of enamel caries?
Four zones of alternating levels of demineralization illustrate the dynamic nature of caries. The surface zone blocks passage of calcium ions into the body of the lesion. It is well mineralized by replacement ions from plaque and saliva. The body of the lesion is poorly mineralized. The dark zone has some remineralization, whereas the translucent zone has high demineralization (Fig. 8-10).
31. What are the histologic zones of cavitated dentinal caries?
There are two distinct layers.
1. Outer layer—dentin heavily infected with bacteria; organic matrix and mineral are lost and cannot be repaired; liquefaction foci and clefts form.
2. Inner layer—dentin affected by plaque acids and demineralized; few bacteria, damage reversible.
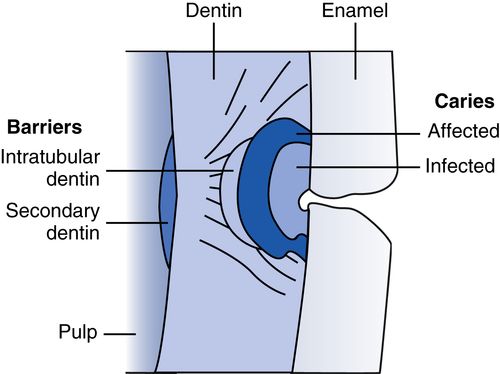
A barrier layer of sclerotic (translucent), well-mineralized dentin may be formed by odontoblast deposits into intratubular spaces. Finally, a layer of secondary reactionary-reparative dentin forms at the dentin-pulp junction. These preceding two layers are dependent on a vital pulp’s reaction to noxious stimuli (Fig. 8-11).
32. Describe the progression of caries in dentin.
Because dentin is a vital tissue, it shows reactivity with bacterial invasion. Caries generally spreads laterally along the dentoenamel junction (DEJ), involving dentinal tubules. At the infected layer of dentin, bacteria enter the dentinal tubules and decalcify the matrix by acid and proteolytic dissociation (microscopic liquefaction foci and clefts are present). The inner affected layer is demineralized, but contains few bacteria. The damage here is reversible if the bacterial metabolism is halted by excavation of the infected layer. Under the affected layer is the sclerotic or microscopic translucent layer, followed by a layer of reparative or reactionary dentin produced by odontoblasts at the dentopulpal boundary.
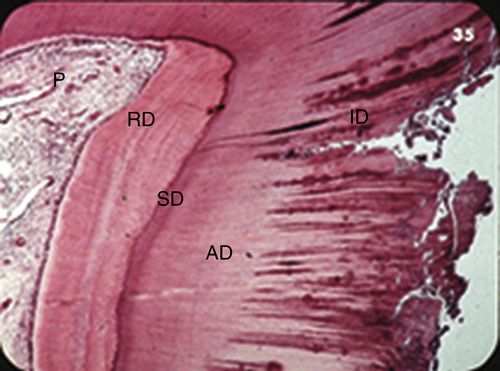
33. Identify the labeled structures in this cavitated carious lesion shown in Figure 8-12.
Carious dentin fills most of the slide, on the right. At the far left is the pulp; the faint blue dots are lymphocytes (chronic inflammation). To its right is a band of reparative dentin that protected the pulp from infection for a time. Next, progressing right, is a thin band of sclerotic dentin, followed by a layer of affected dentin, and at the far right is infected dentin.
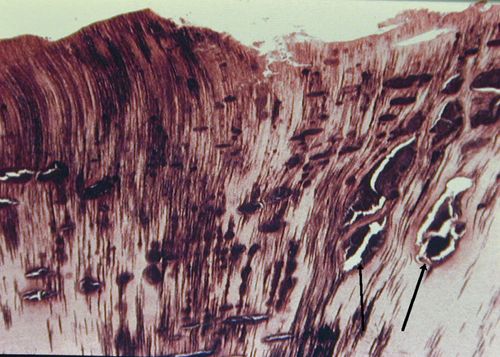
34. Identify structures shown in Figure 8-13.
Note that zones of destruction coalesce to form clefts and liquefaction foci. These are filled with necrotic debris. These advancing bacteria seem to have acidogenic and proteolytic properties, resulting in the enzymatic destruction of the organic dentin matrix. Horizontal clefting is typical of dentinal caries. The process of beading, coalescence, and clefting typifies progression of dental caries.
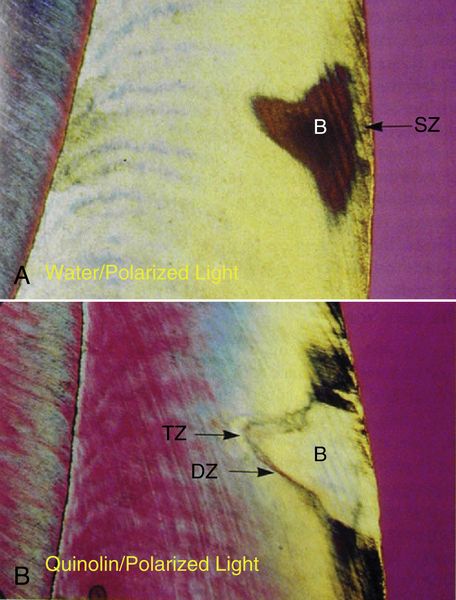
35. Describe the elements shown in Figure 8-14.
Figure 8-14A—small, smooth-surfaced enamel caries in water with polarized light; lesion is cone- shaped; body appears dark beneath intact surface layer.
Figure 8-14B—same section in quinolin (same refractive index as enamel) with polarized light
1. Translucent zone visible as porous, less mineralized leading zone in enamel
2. Dark zone thought to be area of redeposition of mineral
3. Body is demineralized, with striae of Retzius evident
4. Surface zone (SZ) intact because of constant demineralization (destruction) and remineralization (repair) if fluoride present
36. Clinically, how are the different types of carious dentin treated?
All infected dentin must be removed for successful tooth viability. Because affected dentin has undergone only early demineralization, removal may not be necessary. Topical bonded dentin sealants containing fluoride affect a barrier under restorations.
37. Explain the differences between smooth and pit and fissure caries.
Because smooth surface caries (interproximal or cervical) have a wide enamel surface pattern that converges with the anatomic form of the enamel rods toward the DEJ, fewer dentinal tubule are affected, enamel undermining is generally less, and the rate of progression is slow. These lesions have a high success of remineralization with adequate fluoride exposure and reduction in risk factors.
Pit and fissure caries are narrow at the enamel surface and spread widely as the caries progresses to the DEJ, involving many dentin tubules and creating extensive undermining. The progression is often more rapid than that of smooth surface caries. Because of the anatomic sheltering of bacterial plaque, remineralization is not easy, and fissurotomy followed by sealants is necessary for early lesions.
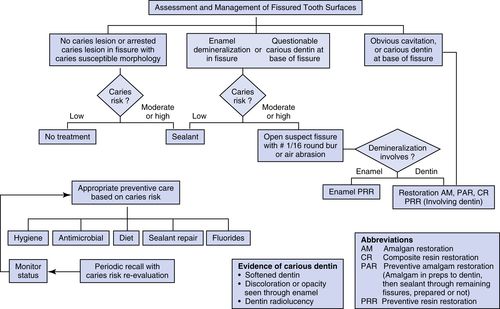
38. Develop a decision tree for the management of pit and fissure tooth surfaces.
See Figure 8-15 for an example of a decision tree for the management of pit and fissure tooth surfaces.
39. How may caries be diagnosed?
Caries may be detected by a combination of techniques. The most commonly accepted criteria for identifying infected tooth structure are the following: (1) discolored, softened tooth structure; (2) frank cavitation; and (3) areas of radiolucency on radiographs. Direct visual inspection of pits and fissures, root surfaces, and interfaces of restorations and tooth with a sharp explorer and air-drying with the use of magnification are the first steps of the examination. This procedure is supplemented by evaluating properly angulated bitewing and periapical radiographs. Finally, the use of transillumination from a visible light curing wand can reveal shadowing and discoloration on occlusal and interproximal tooth surfaces.
40. Describe two high-tech methods of diagnosing dental caries.
1. Quantitative light-induced fluorescence (QFL) measures the scattering of light induced by the degree of enamel demineralization. KaVo’s DIAGNOdent (www.kavousa.com), Midwest’s Caries I.D. (www.mwdental.com), and AirTechniques’ Spectra probe (www.airtechniques.com) use red laser light to assess pit and fissure and smooth-surfaced lesions. A newly developed system, the Canary System, uses frequency domain photothermal radiometry and modulated luminescence (FD-PTR and LUM) to detect carious tooth structures (www.thecanarysystem.com).
2. Digital imaging fiberoptic transillumination (DIFOTI; www.difoti.com) provides a highly sensitive method of diagnosing occlusal and interproximal carious lesions. Images of transilluminated visible light are captured by a digital charge-coupled device (CCD) camera and sent to a computer where analysis of the decreased densities caused by demineralization are shown as images. This method is claimed to be twice as sensitive for detecting interproximal caries and three times more sensitive for occlusal caries than currently used x-rays.
41. What are the objectives of operative treatments in carious teeth?
1. To remove bacterially infected enamel and dentin
2. To protect the dental pulp
3. To preserve healthy tooth structure and restore missing structure
4. To remove the sources of cariogenic bacteria by facilitating plaque control
5. To provide minimal fluoride concentrations in the microenvironment
42. List logical steps to stabilize dentition with active and multiple caries.
1. Carry out a thorough medical and dental history and examination to assess caries risk factors.
2. Initiate appropriate preventive measures.
3. Plan extraction of nonsalvageable teeth.
Note: This step may be resequenced in the apprehensive patient until full trust and compliance are achieved because extractions may be too traumatic as an initial treatment phase.
4. Evaluate of caries in vital teeth and protect with pulp-capping agents such as Ca(OH)2 sealed with bonded and flowable resins or resin-reinforced glass ionomers. Use glass ionomer build-up materials for the temporary filling.
5. In frank pulpal exposures, removal of the pulpal tissues is advisable to prevent potential pain, followed by temporization with a suitable glass ionomer material and endodontic treatment.
6. In very deep carious lesions, whether symptomatic or not, if pulpal exposure is to be expected, it is probably best to go directly to endodontic treatment rather than try an intermediate step of excavation and temporary stabilization.
7. Finalize a treatment plan with permanent restorations for the existing teeth and make suitable provisions for replacement of missing teeth.
43. What are caries detector solutions? How are they used?
Caries detector solutions are usually a colored dye (red, green, blue) in a propylene glycol base; they help distinguish between infected and affected dentin. The dye bonds to the denatured collagen in the infected dentin that is part of the decay process. The affected dentin, which may be slightly softer than sound dentin, is not infused with bacteria and is not stained, but still may show a dye-stained haze (pink haze with red dye). This dentin should not be removed. The caries detector solution is applied for 10 seconds and then rinsed off. Any deeply stained tooth structure is then removed. The materials also help identify cracks in tooth structure (e.g., Seek [Ultradent], Snoop [Pulpdent]).
Note: Some products may decrease the bond strength to dentin and careful consideration of their use is necessary when high bonding requirements are needed.
44. Are cavity disinfectants useful?
Some current thought reflects the goal of cleaning a preparation before bonding or placing a restoration with the addition of a bactericidal agent to reduce sensitivity and bacterial growth under a restoration. It is thought that bacteria reaching the pulp may contribute to sensitivity. Current products contain benzalkonium chloride and EDTA or 2% chlorhexidine gluconate.
45. What supplemental sources of topical fluoride may be used for caries prevention?
• Public water supplies: 1 ppm sodium fluoride (NaF)
• Prescription: PreviDent 5000 Plus, 1.1% NaF
• Mouth rinses: Act, FluoriGuard, Prevident rinse, 0.2% to 0.5% NaF
• Brush-on gels, fluoride trays: Prevident, 1.1% NaF, neutral pH
46. What is a contraindication to the use of acidulated or stannous fluoride preparations?
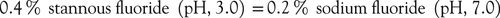
Acidulated fluoride (APF) solutions and topical 0.4% stannous gels (Gel-Kam, Colgate) remove the glaze from porcelain, glass ionomer, and composite restorations. It is best to use neutral pH supplements if these restorations are present. Always check the product specifications.
47. Describe a medical model of caries treatment.
The medical model attempts to shift the caries balance toward no caries by using medical and behavioral components. The key elements are:
1. Control bacteria.
a. Surgical antimicrobial treatment: fill or temporize cavitated lesions.
b. Chemotherapeutic antimicrobial treatment: use chlorhexidine, cetylpyridinium,
fluoride varnish, xylitol chewing gum.
2. Reduce risk level of at-risk patients.
a. Diet
b. Oral hygiene
c. Sealants
3. Reverse active sites (remineralization) with fluoride supplements via gels, rinses, or toothpastes.
4. Carry out long-term follow-up and maintenance (Fig. 8-16).
48. What is Pro-t-action, and how is it used to prevent tooth decay?
BASF, in collaboration with the Berlin-based OrganoBalance GmbH, will commercialize Pro-t-action, made from probiotic microorganisms (Lactobacillus spp.). The active ingredient in Pro-t-action binds to and eliminates caries-causing bacteria (S. mutans) from the mouth. A particular strain of L. paracasei has been identified, and BASF developed a proprietary production process so that the active ingredient can easily be integrated into common consumer products used for daily oral care such as toothpaste, mouthwash, candies, lozenges, and chewing gums. By swishing and swallowing or rinsing, the clumped pathogens are easily expectorated from the mouth. This is the first pathogen-specific clearance rinse. See http://www.basf.com/group/corporate /en_GB/innovations/research/innovation/innovative-solutions/pro-t-action.
49. What is CAMBRA?
As noted earlier, in questions 8 and 9, CAMBRA is a modern caries treatment concept that attempts to identify pathologic and risk factors and implements protective strategies that can prevent cavitation or progression of carious lesions. Clinicians identify the patient’s caries risk by evaluating disease indicators, risk factors, and preventive efforts. After evaluation, a caries risk classification can be determined, and preventive chemical measures and lifestyle and habit changes can be suggested and implemented by the clinician.
50. How may caries be diagnosed?
Caries may be detected by a combination of techniques. The most commonly accepted criteria for identifying infected tooth structure are the following: (1) discolored softened tooth structure; (2) frank cavitation; and (3) areas of radiolucency on radiographs. Direct visual inspection of pits and fissures, root surfaces, and interfaces of restorations and tooth, along with additional tactile information from a sharp explorer and air-drying with the use of magnification, are the first steps of examination. This procedure is supplemented by evaluating properly angulated radiographs. Finally, laser fluorescence and fiberoptic devices can be used in diagnosing carious lesions.
51. Describe light-based methods of diagnosing dental caries.
1. Fiberoptic transillumination (FOTI) uses light transmission through the tooth to measure surface scattering on the outside of the lesion. Digitized fiberoptic transillumination (DIFOTI, Electro-Optical Sciences; now licensed with KaVo Dental GmbH as DIAGNOcam) is a light-based tool that captures images of transilluminated visible light on a digital CCD camera and sends them to a computer, where analyses of the decreased densities because of demineralization are shown as images.
2. Optical coherence tomography (OCT) can create cross-sectional images of biologic tissues using nonionizing imaging techniques. When a carious tooth is illuminated with infrared light, OCT provides two- or three-dimensional (3D) images and a quantitative image of the subsurface lesion to the depth of the enamel.
3. Quantitative light-induced fluorescence (QFL) measures the scattering of light produced by the degree of enamel demineralization. KaVo’s DIAGNOdent probe uses red laser light to assess pit and fissure lesions (www.kavousa.com).
52. What about caries vaccines?
Studies have focused on anticaries vaccines for S. mutans. These vaccines have antibodies against bacteria surface receptors Ag I and II, preventing microorganism adhesion to tooth structure. Newer investigations have targeted intranasally administered anticaries DNA vaccines to produce mucosal and systemic responses and protection against caries.
53. What are some indications for fluoride gel applications using a custom tray?
Patients who exhibit high caries incidence, root caries, or cervical caries and who might fit into one or more of the following groups:
• High consumption of carbonated beverages (pH, 3.2-3.5) or citric fruits (e.g., lemons, limes)
• Bulimic patients (10% of female adolescents)
• Older patients and nursing home patients
• Gastric reflux patients
• Chemotherapy and head and neck radiation–treated patients
54. What is tooth attrition?
Attrition is the physiologic wear of tooth structure resulting from normal tooth to tooth contact over time.
55. What is erosion? What are its possible causes?
Erosion is the loss of tooth structure by a chemical process that does not involve bacterial action. It is generally caused by the consumption of foods that contain phosphoric or citric acid, such as fruits, fruit juices, and carbonated or acidic beverages. Excessive exposure to gastric acids because of vomiting is also a contributing factor.
56. What is the theory of tooth abfraction?
Abfraction is defined as the pathologic loss of tooth substance caused by biomechanical loading forces. The loss of structure is usually seen as wedge-shaped cervical lesions at the cementoenamel junction (CEJ) that may not be carious. This theory is used as an alternative explanation for areas that historically were attributed to toothbrush abrasion. They are also referred to as noncarious cervical lesions (NCCLs).
57. List generally accepted principles for cavity preparation.
1. Cavity preparations should be governed by tooth anatomy, tooth position in the dental arch, extent of the carious lesion, and physical properties of the filling material.
2. Gingival margins should be ended on enamel whenever possible.
3. Cavity preparations margins should be supragingival whenever possible.
4. Margins of posterior cavity preparations should not end directly in occlusal contact areas. Contact areas should be composed of one material to allow even wear. Uneven wear results if two materials meet at the contact area, thereby producing open margins.
5. Weakened and unsupported tooth structures should be removed.
6. Maintaining a dry work field with the use of a rubber dam always enhances the consistent quality of restorations.
58. Describe the principles of cavity preparation for composite resins and amalgam alloy.
The classic cavity preparations, according to Black’s principles, are generally not needed for contemporary bonded retained composite and amalgam restorations. Dovetails, retention grooves, and extension into uninvolved occlusal grooves are generally not needed. Maximizing tooth structure dominates design, with sealants replacing groove extensions.
59. How do the basic principles of operative microdentistry differ from those of traditional operative dentistry, as advocated by Black?
The Black approach to operative dentistry tends toward the destruction of healthy tooth structure to remove smaller amounts of unhealthy tooth structure (extension for prevention). The concept of microdentistry seeks to diagnose unsound tooth structure that is a threat to the tooth and remove that threat, with minimal encroachment on the surrounding healthy tooth structure.
60. What is a fissurotomy procedure? What are its applications?
A fissurotomy involves the conservative preparation of occlusal pits and fissures using air abrasion or special burs designed for this purpose; the fissurotomy kit (Ivoclar Vivadent) is one example. This procedure is used to treat pits and fissures with incipient decay. Preparations are typically narrow, long, and irregularly deep, so they are often restored with flowable composites. Hybrid composites, although stronger and more wear-resistant than flowables, may be clinically more difficult to place into such preparations without the incorporation of voids.
61. What are the advantages of the fissurotomy procedure over pit and fissure sealants?
It is often difficult to determine caries activity in pits and fissures, particularly with conventional means. Concerns arise about the placement of sealants over undiagnosed caries. Questionable occlusal grooves covered with a sealant, particularly in fluoridated communities, may mask more extensive subsurface caries activity. The fissurotomy offers better access and is a conservative technique for maximizing the retention of healthy tooth structure while ensuring certain removal of all decay.
62. Describe the so-called tunnel preparation.
The tunnel preparation is a conservative approach to restoring class II caries in teeth with relatively small interproximal lesions. It conserves the proximal marginal enamel by using only occlusal, buccal, or lingual access and then angulating mesially or distally until the external tooth enamel is perforated. Usually, application of a matrix band beforehand protects the adjacent tooth wall. The tooth cavity is then packed from the access dimension.
63. What is a slot preparation?
Any narrow access to reach interproximal caries can be called a slot preparation. The access may be buccal or lingual, as in a class III lesion, or from the occlusal aspect. The ideal is to conserve tooth structure by removing only caries and a minimal amount of tooth structure.
64. Define micro air abrasion. When is it used?
This technique uses pressurized delivery of an abrasive powder (e.g., aluminum oxide) to prepare teeth for restoration. The claimed advantages are less trauma and a less invasive, heatless procedure, often not requiring local anesthesia. It is ideally suited for pit and fissure sealant preparations and conservative class I and V preparations using flowable composites. Disadvantages include the need for special high- speed evacuation equipment and high cost of the units.
65. Describe the principle behind air abrasion for cavity preparation.
Air abrasion is based on kinetic energy. Abrasive particles, typically 27- to 50-μm aluminum oxide, are propelled at high velocity to remove tooth structure. By varying the speed of the particles, the length of time to remove tooth structure and often the level of sensitivity can be controlled.
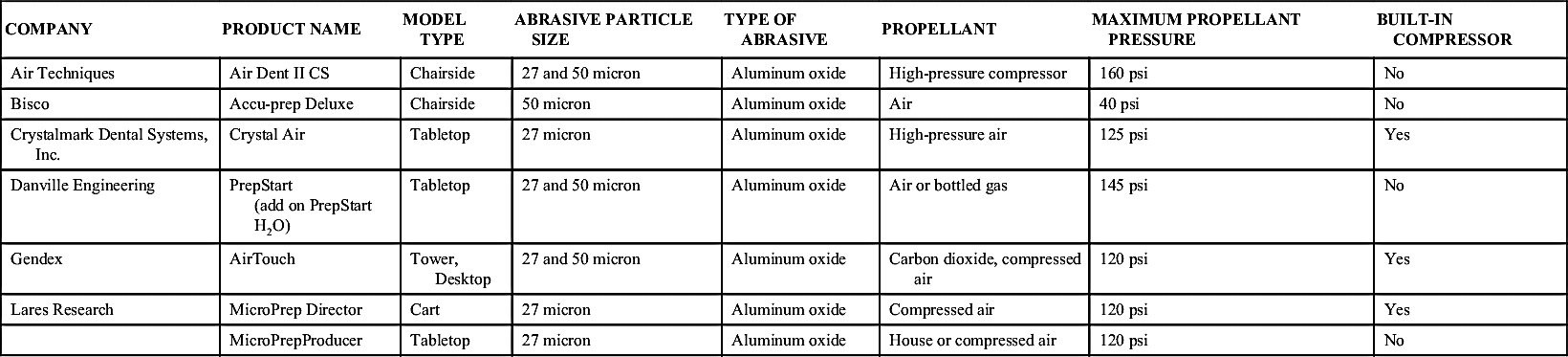
66. What are the various sources of propellant for air abrasion units?
Depending on the type of unit and manufacturer, the propellant can range from compressed air or nitrogen to operatory-compressed air lines or units with built-in air compressors.
67. What dental procedures are well suited for the use of air abrasion? Are any procedures contraindicated?
All classes of cavity preparation may be performed with air abrasion. Some operators may use a high-speed hand piece to gain initial access to deep grooves, pits, marginal ridges, and hard-to-reach areas. Although air abrasion units have the ability to remove dental amalgam, some questions remain about the amount of mercury released by air abrasion versus removal with conventional, high-speed hand pieces.
68. Why is air abrasion considered well suited to the application of microdentistry?
Treatment goals of microdentistry include preservation of sound occlusal enamel with minimally invasive occlusal preparations and the use of tunnel or slot preparations to treat primary interproximal caries. Air abrasion is much more selective in the structure it cuts, thereby removing very little sound tooth structure relative to the use of high-speed hand pieces. Additionally, the vibration from high-speed hand pieces can cause enamel fractures that air abrasion cannot (Table 8-1).
Dental Adhesives
69. What are dental adhesives?
Dental adhesives are products that allow the predictable adhesion of restorative material to dentin and enamel. They are based on using a number of agents to prepare the surface, bond to the tooth surface, and act as a substrate to which the restorative material bonds. In general, they are technique-sensitive and constantly evolving in the marketplace.
70. What are the goals of dental bonding?
• Eliminate or minimize the contraction gap of composite polymerization.
• Sustain thermal expansion and contraction cycles.
• Create 20- to 30-MPa bond strengths to enamel and dentin.
• Eliminate microleakage.
71. What are signs of microleakage?
They are stains, sensitivity and pulpal symptoms, recurrent caries, and bond failures.
72. What are the components of adhesive systems?
Most current systems are combinations of components. Some are multicomponent, depending on whether they are self-cure, light-cure, or both, whereas others have a single component. Unlike early-generation systems, all the new bonding agents are hydrophilic to allow compatibility to dentin bonding. The basic components are an acid etchant solution, hydrophilic primer, and resin.
73. What types of adhesive systems are available?
Type 1. Etchant applied, washed off to remove smear layer; primer and adhesive resin applied separately as two solutions. Type 1 systems are all-purpose types. They generally bond to light-, dual-, and self-cured composites.
Table 8-2
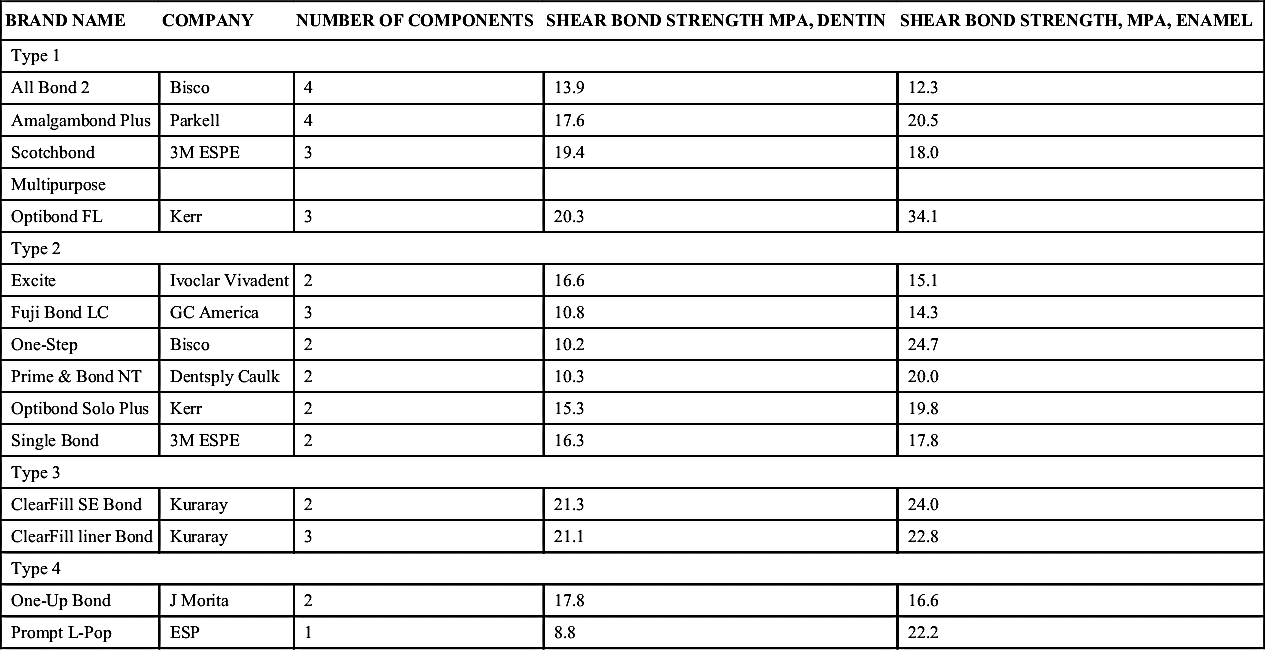
From Clinical Research Associates Newsletter, 24(11):1, 2000. A publication of CR Foundation, www.CliniciansReport.org.
Type 3. Self-etching primer (SEP) is applied to dissolve smear layer and not washed off; adhesive is applied separately.
Type 4. Self-etching primer and adhesive are applied as a single solution to dissolve and treat the smear layer simultaneously (Table 8-2).
74. What types of adhesive systems are currently used in clinical practice?
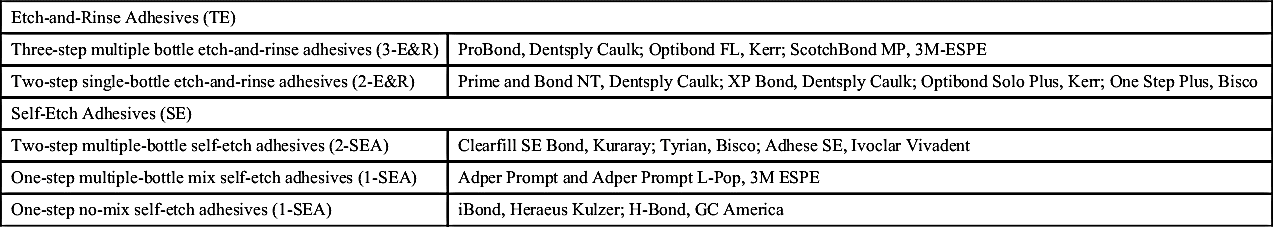
Many variables, including timing, rinsing, drying, rewetting dentin, and maintaining a controlled operative field are important in the placement of dental adhesives. Over time, manufacturers focused research efforts on the development of simplifying these systems to reduce the potential for inconsistencies in treatment by reducing the number of steps required for application. Classification systems became confusing as generational models advanced. Bonding systems that required phosphoric acid etching with rinsing were referred to as total etch whereas those that did not require the additional step were called self-etch. The sixth- and seventh-generation systems were self-etching models. In current practice, adhesives can be classified as total etch (TE) and self-etch (SE; Table 8-3).
75. What is the hybrid layer?
The hybrid layer is a multilayered zone of composite resin, and dentin and collagen. After removing the organic and inorganic debris of the smear layer by etching and reducing some hydroxyapatite from the intertubular dentin down to 2 to 5 μm, a plate of moist collagen remains on the dentin floor. Priming agents penetrate this moist collagen substrate and migrate into the tubules, lateral canals, and all areas of peritubular dentin. This becomes the hybridization process as the dentin, both collagen and hydroxyapatite crystals, become totally impregnated with bonding resin. The resin further penetrates into the dentin tubules. Light curing produces a mechanically and chemically bonded surface that can polymerize to composite restoratives.
76. What is essential for successful hybrid layer formation?
Supersaturating of the dentin substrate with primer or wetting agent is essential. If the etchant time is 15 seconds, the wash should be at least as long. The water is then dispersed to leave the dentin moist. Multiple coats of priming agent are applied to achieve a glossy surface on air dispersion. Resin is then applied and cured.
77. How is enamel bonding achieved?
Bonding to enamel is micromechanical; a low-viscosity resin penetrates the microporosity created by acid etching on the enamel surface. Once the resin is cured, it strongly adheres to the enamel and forms the suitable substrate for composite bonding. Although previously done under dry conditions, contemporary adhesive systems use a wet bond to enamel. If one is bonding only to enamel, it is necessary to use only an unfilled resin without the primer application.
78. How is bonding to dentin achieved?
Dentin is largely composed of organic materials, mostly collagen and water. (Enamel is 86% mineralized, whereas dentin is 45%.) Bonding to dentin can require removal of preparation debris (smear layer) and demineralization of the dentin surface by acid etchant. This leaves a lattice of unsupported collagen as long as the surface stays moist. Hydrophilic primers and resins (applied in solvents of acetone or alcohol) can then penetrate this matrix, infusing a micromechanical lock similar to enamel. When cured, this resin-reinforced dentin complex forms the hybrid layer, and is a suitable substrate for composite bonding. With newer self-etch primer adhesives (SEPs), the etchant and primer are applied and not washed off; the smear layer remains. An adhesive is applied separately. The dentin with these latter systems is never denuded, and there may be less technique sensitivity because the process of leaving dentin to damp-dry is eliminated.
79. How long should you etch?
Etching dentin demineralizes its surface to a depth of 1 to 10 μm. If you etch too long, you may create a depth of demineralized collage that is too deep for adhesives to penetrate, thus weakening the bond or denaturing the remaining collagen. The total etch of a tooth preparation with 32% to 40% phosphoric acid gels should be a maximum of 15 seconds, which also works for enamel.
80. Why must the dentin surface be kept wet?
When using types 1 and 2 adhesive systems, the etching step leaves the dentin collagen lattice largely unsupported. If this layer dehydrates, it will collapse and the applied adhesives will not be able to infuse through the collage. The surface should be left moist by only the slightest application of air to eliminate puddles of water—or, even better, blotted with a sponge or gauze. Some adhesive systems rehydrate the demineralized zone, even if it is overdried.
81. What potential problem may cause an incomplete seal of dentin tubules?
Incomplete placement of the bonding reagents may result in an increase in postoperative pulpal sensitivity. There may be incomplete wetting on application of the primer agent or incomplete curing of the bonding agent. One must be sure to place incremental layers of wetting agent until a glossy appearance is observed on gentle air dispersion, and a well-calibrated curing light must be used for sufficient exposure times.
82. What factors contribute to increased pulpal sensitivity, even with proper technique?
If the dentin is dried too completely, air emboli may enter the dentin tubules and the dentin bonding layer may overseal the layer of air. There is thus a potential for mechanical masticatory stresses and a resultant sensitivity on biting on the tooth restoration unit. To avoid this problem, leave the dentin moist by gentle air dispersion; do not use drying. Then the hydrophilic primers will follow fluid down the tubules and fill intertubular dentin and tubules with resin.
83. How long should you apply the adhesive?
The adhesive must penetrate through the demineralized dentin to form the hybrid layer for maximal bond strength. Some systems are faster than others; thus, following the manufacturer’s instructions is important. In general, after applying the adhesive, 15 to 20 seconds should be allowed for penetration. Air evaporation of the solvent (acetone or alcohol) is followed by curing (generally, visible light-cured). This should leave a shiny dentin surface. If this goal is not achieved, reapplication of the adhesive should be performed until a shiny layer appears.
84. Describe the composition of contemporary primers in adhesive systems.
Primers are bifunctional molecules. One end is hydrophobic to bind to the adhesive, and the other end is hydrophilic. The hydrophilic end permeates conditioned dentin and chases the water of the moist surface, assisted by solvents (acetone or alcohol). After this penetration, the solvents need to be evaporated by air drying. Examples of primers are HEMA (2-hydroxyethyl methacrylate), 4-META (4-methacryloyloxyethy trimellitate anhydride), and PENTA (dipentaerythritol penta-acrylate monophosphate). Generally, they do not have any light-curing capabilities.
85. Describe the bonding resin adhesive.
Bonding resins are unfilled bis-GMA (bisphenol A glycidyl methacrylate) or UDMA (urethane dimethacrylate). They may be visible light-cured (VLC), autocured, or dual-cured. The later generation of adhesive systems consolidated the primer and adhesive into premixed applications, which leads to a time savings. There has been a trend to add fillers to the adhesive bonding agents to enhance their physical properties.
86. What enhancement do fillers contribute to newer adhesive bonding resins?
• Increase the bond strength at the hybrid layer
• Improve stress absorption at the tooth restoration interface, enabling better retention
• Have a lower modulus of elasticity to impart added flexibility and thus relieve contraction stress caused by polymerization shrinkage; adhesive absorbs within itself some of the contraction stress
• Help adhesive cover the dentin in one application rather than multiple applications
87. What are the seventh-generation adhesive systems?
The latest systems combine applications of conditioning, priming, and adhesive resin all in one bottle. They are referred to as “single-component, one-step, self-etch adhesives” or “all-in-one adhesives.” Examples include Clearfil S3 Bond (Kuraray Dental), G-Bond (GC America), iBond (Heraeus Kulzer), and Xeno IV (Dentsply Caulk).
88. Can you use any adhesive with any composite?
Generally, any light-cured composite should bond to any light-cured adhesive. However, self-cured composites such as core pastes are not compatible with most single-component adhesives. Dual-component or self-cure adhesives must be used with self-cure composites.
89. Outline the adhesive procedures for bonding composites and amalgam to tooth structure.
1. For composite materials
To enamel: Clean surface with pumice, wash, etch 15 seconds, wash, air-dry, and apply unfilled VLC resin only.
To dentin and enamel: Clean surface, etch 15 seconds, wash, leave moist, use VLC adhesive components in layers before composite, and consider filled adhesives.
2. For amalgam (bonding to dentin only): Clean surface, etch 15 seconds, wash, use VLC primer to seal tubules, self-cure resin adhesive (two-component system), and pack amalgam before resin sets.
Composites
90. What are the components of composite resins?
• Monomers: Bis-GMA, UDMA
• Diluent monomers: TEGDMA (tetraethyleneglycol dimethacrylate), MMA (methyl methacrylate)
• Inorganic fillers: quartz, glass, zirconium
• Organic fillers: silica
• Coupling agents: silane
• Initiators: tertiary amines, camphorquinone (CMP), phenyl propanedione (PPD), benzoyl peroxide
• Inhibitors: ether of hydroquinone
• Ultraviolet absorbers: benzophenone
91. Describe the function of each monomer component.
Principal monomers are high-molecular-weight compounds that can undergo free radical addition polymerization to create rigid, cross-linked polymers. The most common monomer is Bis-GMA (an aromatic dimethacrylate that is the addition product of bisphenol A and glycidal methacrylate [GMA]). An alternative monomer is urethane dimethacrylate.
Diluent monomers are low-molecular-weight compounds used to reduce the viscosity of the unpolymerized resins to improve physical properties and handling. There are two types, monofunctional (MMA) and difunctional (ethylene glycol dimethacrylate or triethylene glycol). The latter are used most often because they form harder and stronger cross-linked composite structures because of a lower coefficient of thermal expansion. They also have less polymerization shrinkage, are less volatile, and have less water absorption.
92. What are filler particles?
Inorganic filler particles used in composite resins include quartz, glass, and colloidal silica, along with additions of lithium, barium, or strontium to enhance optical properties. These fillers are coated with a silane coupling agent (organosilane) to bond adhesively to the organic resin matrix. Silane bonds to the quartz, glass, and silica particles, whereas the organic end bonds to the resin matrix.
93. Describe the mechanism of silane coupling.
During free radical polymerization of the organic Bis-GMA, covalent bonds are formed between this polymer matrix and the silane coupling agent, commonly gamma-methacryloxypropyltrimethoxy silane. The coupling agent that coats the filler particles at the silane end thus holds the inorganic and organic phases together, which prevents further water absorption.
Stay updated, free dental videos. Join our Telegram channel

VIDEdental - Online dental courses