Indications and Patient Selection for Radiotherapy
Patient Information and Consent
Radiotherapy Treatment Technique for Electrons
Radiotherapy Treatment Technique for Photons
Surgery Followed by Postoperative Radiotherapy
Toxicities using Alternative Radiation Techniques
Introduction
Neoplasms of the salivary glands account for 2%–4% of tumors that arise in the head and neck region. Benign salivary tumors include Warthin tumors, myoepithelioma, and subtypes of adenomas, papillomas, and lymphadenomas but pleomorphic salivary adenoma (PSA) is the predominant subtype (70%–80%).1 A PSA is composed of myoepithelial cells, epithelial cells, neoplastic ducts, and stroma contained within a false capsule. The peak incidence is at a median age of 46 years (range 30–60 years) and there is a female preponderance (approx. 1.5: 1).
PSAs arise most frequently in the parotid gland. In most patients, 80%–85% of the parotid gland forms the superficial or lateral lobe, lying lateral to the facial nerve, and 90% of PSAs therefore arise in the lateral lobe of the parotid. No etiological factors have been identified, and the sublingual and submandibular glands, and minor salivary glands such as those in the palate, buccal mucosa, and lip, may also be affected.
Radiotherapy is an effective and accepted treatment for malignant tumors of the head and neck; however, there are a few indications for radiotherapy for benign tumors of this anatomic site. PSA is the main benign salivary gland neoplasm for which there are recognized indications for radiotherapy and will form the focus of this chapter.2
Clinical Presentation
Patients with a PSA of the parotid gland typically present with a painless, slow-growing preauricular mass. In contrast, malignant tumors present more commonly in older patients with a rapidly growing mass, involvement of the ipsilateral facial nerve, or palpable cervical lymphadenopathy. Lymphoma is in the differential diagnosis of a rapidly enlarging mass at this site, especially if associated with systemic lymphadenopathy. Although PSA is benign, it will slowly progress and may result in significant morbidity and, rarely, mortality.
According to the World Health Organization (WHO) definition, PSA may transform to carcinoma when there is evidence of “anaplasia, abnormal mitoses, a progressive course, infiltrative growth alongside evidence of pleomorphic adenoma.”1 For further discussion of malignant transformation of PSA, see also Chapters 22 and 24. The diagnosis of “carcinoma in pleomorphic adenoma” includes metastasizing PSA, which may spread to bone, cervical lymph nodes, lung, and abdominal viscera without evidence of invasion in the metastases.3 Carcinoma ex pleomorphic adenoma may develop from a PSA in less than 5% of cases, and in situ or intracapsular carcinoma and carcinosarcoma are other variants within this group which, together, represents 3.6%–6.5% of malignant salivary gland tumors.4 A correlation between the incidence of malignant transformation and time from diagnosis has been reported (1.6% 0–4 years, 5.9% 10–14 years, 9.4% > 15 years).5,6
Treatment
Surgery is the preferred option for the primary management of PSA and, if negative margins can be achieved, may be curative (99% local control at a median of 6–10 years).7–9 Resection of the tumor also allows verification of the histology, to exclude adenoid cystic and mucoepidermoid carcinoma in particular. Historically, some surgeons elected to enucleate a PSA, but the technique of enucleation was associated with very high recurrence rates. A lateral parotidectomy is suitable for a PSA arising in the lateral lobe. In contrast, a total parotidectomy with preservation of the facial nerve may be necessary for the minority of patients in whom the tumor arises from the deep lobe. However, some centers have suggested a total parotidectomy, sparing the facial nerve, for all patients with a PSA of the parotid gland.10,11
 For primary pleomorphic adenoma located in the parotid gland, surgical resection—i.e., parotidectomy—is the standard therapy.
For primary pleomorphic adenoma located in the parotid gland, surgical resection—i.e., parotidectomy—is the standard therapy.
The tendency of PSAs to abut the trunk and branches of the seventh cranial nerve, the facial nerve, may make complete resection difficult and increases the probability of tumor recurrence. PSAs may recur as solitary (uninodular) lesions or multifocally within the gland (multinodular). In patients with recurrent PSAs, approximately half of the lesions are unifocal and half are multinodular. A solitary recurrence may arise from a bud of tumor breaking through the tumor pseudocapsule, and multinodular recurrences are thought to arise from tumor spillage during resection. The average age of patients with recurrent PSA tends to be younger (32 years) than patients at first presentation (47 years).12,13 There is controversy on whether the apparently more aggressive nature of PSAs in young patients is due to differences in biology or clinical management.14,15
The facial nerve courses through the tissue of the parotid gland, but the literature suggests that there is minimal risk to the facial nerve from an initial resection performed by an experienced surgeon. In a modern series of 377 new cases of PSA of the deep lobe of the parotid treated at a tertiary referral center, facial nerve function was preserved in 92% of the patients.16
If the primary surgery was limited—for example, enucleation only—then the recurrence may be salvaged by a superficial or total parotidectomy with preservation of the facial nerve. If a superficial or total parotidectomy has already been undertaken, then the second operation carries a much greater risk of damage or injury to the facial nerve (see Chapter 24). Historically, a repeated surgical procedure for recurrent disease employing an aggressive surgical approach resulted in damage to the facial nerve in 30%–60% of cases.17,18 Sacrifice of the facial nerve should be considered an elective decision for patients who present with poor nerve function preoperatively. Nerve-preserving surgery, with or without adjuvant radiotherapy, reduces this risk to 3%–30%.19–24 In the light of the cosmetic and functional deficit to the patient from a seventh nerve palsy, decisions regarding the optimal management of a PSA usually center on how best to preserve the function of the facial nerve. An additional reason to aim for definitive treatment of a PSA at first presentation is to avoid the small risk of associated malignancy if the tumor recurs.
 Because of the increased risk of facial palsy in revision surgery for PSA, it may be an option in individual cases with recurrent nodules adherent to the facial nerve to perform nerve-preserving surgery in combination with postoperative radiotherapy, to decrease the risk of facial palsy.
Because of the increased risk of facial palsy in revision surgery for PSA, it may be an option in individual cases with recurrent nodules adherent to the facial nerve to perform nerve-preserving surgery in combination with postoperative radiotherapy, to decrease the risk of facial palsy.
The predicted natural history of the PSA in the context of the patient’ s age and comorbidities should also be considered when formulating management recommendations. It is believed that there is an association between the number of recurrences of a PSA and the risk of malignant transformation, possibly related to the multiple interventions.9 In a series of 114 patients with recurrent PSA, three patients with malignant transformation had had three or more recurrences and all had received irradiation.25
Indications and Patient Selection for Radiotherapy
In the rare circumstance that a PSA of a minor salivary gland is not resectable, radiotherapy may be considered as a primary treatment.2 However, a greater extent of residual PSA is predictive of poorer tumor control.10 Radiotherapy is therefore almost invariably an adjuvant treatment for benign salivary gland tumors.
Factors have been identified that increase the risk of recurrent disease following surgery. As tumor recurrence will require a second resection, with an associated risk of damage to the facial nerve, postoperative (adjuvant) radiotherapy may be considered to reduce the risk of subsequent local recurrence. Radiotherapy has potential long-term side effects, and the risk–benefit ratio has to be carefully considered, particularly in benign conditions. For this reason, there is debate as to whether it is better to recommend postoperative therapy, with a risk of morbidity, or to defer treatment until recurrence.26
Despite this dilemma, adjuvant radiotherapy can be considered a standard treatment after a first operation for a tumor that extends to the facial nerve in which clear margins cannot be achieved. Tumor spillage at the time of resection and equivocal surgical margins may be further indications for radiotherapy. Surgical spillage is still the most controversial indication, and the strength of the recommendation for radiotherapy tends to vary in accordance with surgical outcome and be center-dependent. At institutions in which greater than 90% local control has been reported despite tumor spillage at surgery, radiotherapy is no longer used routinely.27,28 For patients with recurrent PSA, radiotherapy is usually recommended.
Patient Information and Consent
Patients who are to undergo radiotherapy to the parotid region should provide written informed consent for the treatment. Acute side effects include dryness of the irradiated skin, radiation dermatitis, and mucositis of the cheek mucosa. Longer-term potential complications have to be explained to the patient and include slowing of beard growth in men, and abnormalities of taste and dryness of the mouth that can last for several months after treatment. The risk of osteoradionecrosis of the mandible or radiation-induced second malignancy is probably 0%–2%. Some series have reported effects on hearing as a consequence of treatment. Otitis externa is common, but middle ear and cochlea damage may also occur if they are within the high-dose region.
Radiotherapy Treatment Technique for Electrons
PSAs arise most commonly in the superficial lobe of the parotid. This is suitable for treatment of the superficial tissues with electrons. Excellent dosimetry—coverage of the planning target volume (PTV) by the 95% isodose—in the superficial lobe may be achieved with a single, directly applied beam of electrons. The main advantage of electrons is that the radiation dose falls off rapidly, sparing normal tissues that lie deeper to the parotid, in particular the oral cavity and the contralateral parotid gland.
Radiotherapy Treatment Technique for Photons
The lack of dose deposition at depth by electrons means that tumors in the deep lobe cannot be adequately covered and that the longer range provided by photons is required. The patient must be positioned with the neck extended, to displace the orbits superiorly to avoid irradiation of the contralateral lens. The patient is then immobilized in the treatment position using a five-point Perspex or thermo-plastic shell. This allows accurate patient positioning and a reproducible set-up to minimize the risk of geographical miss and to reduce the safety margin between the clinical target volume (CTV) and the planning target volume (PTV) (www.icru.org).
Computed tomography (CT) planning has superseded two-dimensional simulation and planning in most radiotherapy centers. The patient is CT–scanned in the immobilization device and the recurrent/residual tumor or tumor bed (CTV) is localized on relevant CT images using a radiotherapy planning system. Preoperative imaging such as magnetic resonance imaging may be available, and the contralateral parotid gland can be used as an anatomic guide. An isotropic margin (3–5 mm) is then added to expand the CTV to the PTV to account for daily variations in patient set-up.
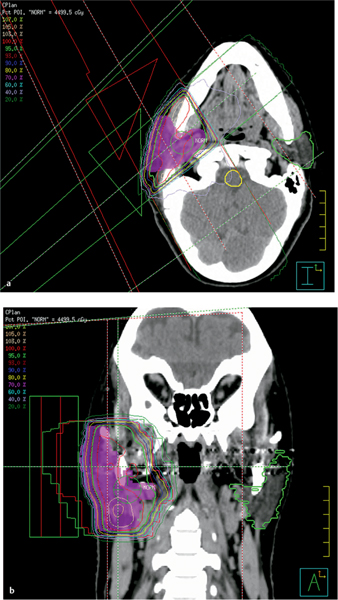
The standard photon beam arrangement for a parotid tumor is a pair of anterior and posterior oblique wedged megavoltage photon beams. This beam arrangement will spare the contralateral parotid gland (Fig. 23.1). The high-energy photons will also spare the skin, and when there is evidence of tumor seeding into the skin or a recurrence in the scar, wax bolus should be applied to increase the dose deposition in the skin. The potential disadvantages of photons are a significant exit dose through the oral cavity, brain stem, and spinal cord, and a high dose to the cochlea.
A third option that has been described in the literature but is rarely used in clinical practice is a mixed photon/electron beam. The rationale is that the lower exit dose from an electron beam helps to spare the contralateral parotid gland. In a planning study, trials of combined 18 MV photons with 20 MeV electrons weighted 2: 3 and 1: 1 were found to deliver a high dose of irradiation to the contralateral parotid gland and were therefore not recommended in practice.29 A combination of lower-energy beams (6 MV photons and 12 MeV electrons or 6 MV photons and 16 MeV electrons, both with a 1: 4 beam weighting) was found to deliver a homogeneous dose distribution to the planning target volume and low doses to the organs at risk.29
For recurrent lesions in the submandibular gland or minor salivary glands of the upper aerodigestive tract, the radiotherapy technique is individualized to the clinical scenario. Irradiation of the submandibular gland usually utilizes a lateralized wedged pair technique similar to that of parotid gland irradiation, described above. For recurrences arising from minor salivary glands in the mucosa of the upper aerodigestive tract, a variety of photon techniques can be applied, depending on the site, and concentrating on the primary tumor site with appropriate margins.
Fig. 23.1 a, b Axial (a) and coronal (b) sections of the radiotherapy treatment plan for a patient with an expleomorphic salivary adenoma of the deep lobe of the right parotid. Anterior and posterior oblique wedged photon beams were used to achieve the isodose distribution shown (the 95% isodose is represented in green). The planning target volume is shaded in pink. The contralateral left parotid (green) and spinal cord (yellow) have been contoured.
Radiotherapy Dose
A range of dose/fractionation schedules are described in the literature. For microscopic residual disease, 45 Gy in 20 fractions or equivalent is the recommended treatment schedule at the Royal Marsden Hospital and many centers in the United Kingdom. In contrast, 66 Gy in 33 daily fractions or 69.2–74.4 Gy in 1.2-Gy fractions twice daily—prescriptions equivalent to a radical dose for head and neck cancers—may be used in the United States.2
Outcome and Prognosis
 Surgery Alone
Surgery Alone
At First Presentation
In a classic surgical series of 346 patients with a new PSA who did not receive radiotherapy, it was observed that six of 26 of patients (8%) in whom tumor spillage was noted at surgery (11%) experienced a local recurrence after a follow-up period of 11–25 years. When the tumor dissection was close to the capsule, one of 87 patients (1.1%) had recurrent disease and three of 121 patients (2.5%) in whom the capsule was not seen or exposed developed a PSA recurrence.29 Interestingly, however, the status of microscopic disease at the resection margin did not impact on PSA recurrence rates in this series. There was a 2% rate of failure to achieve local control following primary resection in all three categories: (1) positive/close margins, (2) tumor cells growing through the capsule, and (3) negative margins.28 Comparable results have also been reported from another surgical series of 213 patients. There was a 7% PSA recurrence rate in the 12% of patients with capsular rupture at surgery. In the remaining 185 patients, recurrences were seen in 4%. In five of nine of these recurrences, tumor buds were seen to extend through the pseudocapsule.30 These data may be interpreted to support tumor spillage as an indication for adjuvant radiotherapy, given PSA recurrence in 7%–8% of patients, which is at least twice the incidence in comparison with “uncomplicated” surgery. Conversely, it can also be argued that tumor spillage is only associated with recurrence in 50% of cases and that recurrences also develop in the absence of spillage.
In a series of 254 patients, 252 (99%) with PSA of the parotid achieved local control following primary resection. This series included an unspecified number of patients with recurrent disease, and the two patients with recurrences had already had a previous recurrence.7 Similarly, local control rates for de novo parotid gland PSA of 99% (follow-up 6–39 years) have been reported9 and 99.6% local control was achieved in 100 patients (all with parotid lesions) following total parotidectomy (follow-up 10 years).8 Among 54 patients with a new PSA of the submandibular gland, 53 (98%) achieved local control at a median follow-up of 76 months. The patient who developed recurrent disease received salvage treatment with a second surgical procedure.31 Optimal surgery for an uncomplicated PSA at first presentation may be sufficient treatment, and complete resection should therefore be the aim.
Recurrent PSA
Unless the threat to the facial nerve is deemed too great, repeat surgery is advocated for localized, recurrent PSA. Surgical outcome data for a limited number of patients with recurrent PSA are available from the literature. In the series of 60 patients with benign tumors of the submandibular glands, four had a resection for recurrent PSA and achieved local control at a median follow-up of 76 months.31 Among 25 patients with recurrent PSA of the parotid who were followed up for a median of 12 years, 40% had local control after one operation and local control was finally achieved in 72% after between one and four further surgical procedures.9 In a series of 126 patients with recurrent PSA of the parotid and an average follow-up of 14 years, the incidence of tumor recurrence was 32.5% after one resection, 7.1% after two, and 1.6% after three. The morbidity associated with this aggressive surgical approach was significant: partial facial nerve paralysis in 13.5% of cases and total paralysis in 5.5%. In the absence of radiation, malignant disease was detected in 7.1%.22 A report on a series of 31 patients with recurrent PSA emphasized that successful surgery for recurrent disease is more difficult to achieve after a formal parotidectomy (62% local control at 7 years) than following limited initial surgery (100% local control at 7 years).32 If the function of most branches of the facial nerve can be preserved, repeat excision should be attempted for recurrent PSA. It is accepted that it may not be possible to remove all recurrent tumor, particularly if it is multinodular.
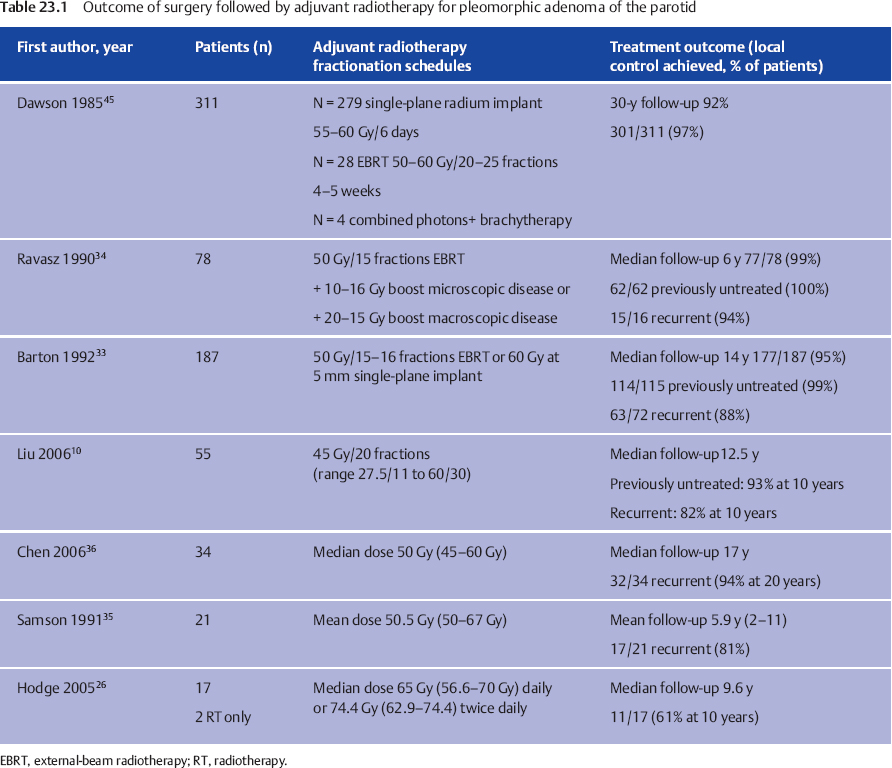
 Surgery Followed by Postoperative Radiotherapy (Table 23.1)
Surgery Followed by Postoperative Radiotherapy (Table 23.1)
PSA at First Presentation
In the series reported by Barton et al., only one of 115 cases of PSA recurred when radiotherapy was given immediately postoperatively.33 The initial surgery may have been suboptimal, and the patient received salvage treatment with a further resection.33 Among 62 patients with a first presentation of PSA, the combination of surgery followed by external-beam radiotherapy (EBRT) (50 Gy in 25 fractions), with a boost up to 16 Gy for microscopic disease and 25 Gy for macroscopic tumor, achieved 100% local control at a median follow-up of 11 years.34 These limited data suggest that judicious use of radiotherapy following surgery may yield local control rates approximating 100%. There are consistent reports that local control is more difficult to achieve both at presentation and for recurrent disease if radiotherapy is given for macroscopic residual PSA (62%–76%) than for microscopic tumor (93–110%),10,35 which again highlights the need for optimal surgery.
Incidentally, adjuvant radiotherapy also appears to protect against the surgical complications of gustatory sweating (Frey syndrome; 35% vs. 12%) and amputation neuroma developing from the stump of the great auricular nerve (16% vs. 0%).13
Recurrent PSA
In the series of 72 patients treated with surgery and postoperative radiotherapy for recurrent PSA, nine (12.5%) failed to achieve definitive local control.33 A second series of 114 patients who had resection of a first PSA recurrence yielded an overall second recurrence rate of 15% (17 of 114), which did not vary relative to the nature of the surgical procedure at the first relapse.21 Subgroup analysis revealed that adjuvant radiotherapy reduced the rate of second recurrences from 43% to 4% at 15 years (P = 0.008) in the multinodular group, but had little impact on the solitary relapse group (15% vs. 13%) at the same time point (P = 0.9). It is likely that this reflects the lower probability of eradicating multifocal disease through surgery alone, and this is supported by a second smaller series.32 A small series of 16 patients with recurrent PSA reported that, following surgery and radiotherapy, 15 patients had not had a further relapse at a median follow-up of 11 years.34 Eighty-two percent of patients with recurrent PSA treated with a median dose of 45 Gy in 20 fractions with EBRT achieved local control at 10 years. In a further series of 24 patients with recurrent PSA who were followed up for 20 years after surgical resection followed by EBRT to a median dose of 50 Gy, the local control rate was 94%.36 Eighty-one percent of 21 patients with recurrent PSA of the parotid who underwent facial nerve preserving surgery and adjuvant radiotherapy (median 50.5 Gy) achieved local control at a median of 6 years of follow-up.35 Despite variations in the surgical procedure and radiotherapy dose, these data consistently report tumor control rates of 80%–95% in patients with recurrent PSA treated with surgical resection and irradiation over the past 30 years.
Complications and Limitations
As patients with benign tumors are likely to be long-term survivors, it is critical to consider the late effects of radiotherapy and to seek to reduce these where possible.
 Audiovestibular Problems
Audiovestibular Problems
There is extensive experience of using postoperative radiotherapy for the commoner malignant parotid tumors. The proximity of the auditory apparatus to the parotid bed is associated with a significant incidence of hearing loss after radiotherapy to the parotid bed in this context.37 While a higher dose of radiation (equivalent to 60–65 Gy in 30 fractions) is prescribed for malignant tumors, long-term auditory impairment may be a consequence of 45 Gy in 20 fractions for the benign tumors. In a series of patients who received hypofractionated radiotherapy (50 Gy in 3.3 Gy over 3 weeks), 15 of 28 patients experienced sensorineural hearing loss on the irradiated side.38 In contrast, irradiation up to 60 Gy at 2 Gy per fraction was not associated with significant hearing loss.39 Amendment of the irradiation schedule at the Christie Hospital, Manchester, United Kingdom from 50 Gy in 3 weeks to 45–47.5 Gy in 20 fractions over 4 weeks has eliminated audiovestibular problems, although otitis externa may be seen in patients with wax plugs in the external auditory meatus. Fourteen percent of 51 patients who received adjuvant radiotherapy following resection of recurrent PSA developed otitis externa.21
 Xerostomia
Xerostomia
The threshold for radiation-induced xerostomia is 26 Gy and will be exceeded by the radiotherapy dose required to treat a PSA. The consequences of reduced saliva flow include the unpleasant sensation of a dry mouth, which is often associated with a reduction in patients’ perception of their quality of life and an increased risk of dental caries, in particular.40 Intensity-modulated radiotherapy (IMRT) is widely believed to hold advantages in terms of sparing normal tissue in cancers of the parotid.41 Despite this, the current uncertainties pertaining to the risk of second malignancy associated with the irradiation of increased volumes of normal tissues to a low dose mean that IMRT is unlikely to be adopted for benign salivary gland tumors that arise in young patients in the near future.
 Vascular Problems
Vascular Problems
There is increasing awareness of the risk of vascular damage following radiotherapy. A case of carotid artery stenosis, potentially leading to a thrombotic cerebrovascular accident, has been reported following irradiation for a parotid PSA.42 There have also been several case reports of temporal lobe necrosis following irradiation for PSA of the parotid gland.43 In one center, three patients were noted to develop serious radiation-related side effects. One patient received 20 Gy in 10 fractions with a 300-kV lateral field. A year later, he was treated again with a wedged pair of photon beams to 40 Gy in 20 fractions. Ten years after this, he developed temporal bone necrosis. A second patient developed temporal bone necrosis 6 years after receiving 60 Gy. The third patient received 55 Gy in 25 fractions and developed temporal bone necrosis after 3 years. Sixteen years later, the same patient developed cerebellar and temporal lobe necrosis. There have been no such reports in the last decade, however, and the adoption of CT planning, with better immobilization and accuracy of delivery and a reduction in the radiation dose and hypofractionation, may have rendered these complications largely theoretical. Radionecrosis of the skin was reported in one patient among 52 who received postoperative radiotherapy following resection for a recurrent PSA,21 but again this is now very rare.
 Radiation-Induced Malignancy
Radiation-Induced Malignancy
The most serious late effect of radiotherapy that needs to be discussed with patients is a radiation-induced malignancy. These concerns involve both a de novo radiation-induced second malignancy and radiation-induced malignant transformation of the PSA. Fortunately, these are rare and may be amenable to curative surgery. An incidence of 0.7% of second malignancies (sarcomas, squamous cell carcinomas, and salivary gland tumors) arising in a previously irradiated area at a median of 36.5 years after radiotherapy and with a median survival of 3.5 years has been reported in patients who received radiotherapy for a primary cancer in the head and neck region.44 In the series reported by Dawson and Orr, radium needle implants were used in the majority of cases.45 Four patients (1%) developed a second malignancy. The malignancies included a sarcoma after 14 years, two adenocarcinomas (after 18 and 30 years of follow-up), and one expleomorphic adenoma after 14 years. In the series reported by Barton et al., one patient developed a malignant transformation of a PSA.33 Chen describes one patient who developed a second malignancy, a low-grade mucoepidermoid cancer of the parotid gland, 14 years after treatment.36 This may be an unusual histology for a radiation-induced cancer, and the original histology should be reviewed in case the original tumor was misclassified. There are other case reports—for example, of a carcinosarcoma arising in a patient treated with adjuvant irradiation for PSA.46 Two series have described in-field malignancy rates of 1%–2%.21,47 These data suggest that patients should be counseled for an overall incidence of radiation-induced malignancy of about 1% over 10–20 years following radiotherapy.
 Other Toxicities
Other Toxicities
Other significant radiation-induced toxicities reported in the series of 115 patients treated with radiotherapy for incomplete surgery or tumor spillage included two cases of radionecrosis, one complete seventh cranial nerve injury, one salivary fistula, and one case of Frey syndrome.33 In modern radiotherapy practice, such complications are very rarely seen. Liu et al. reported one case of osteoradionecrosis that healed spontaneously, and no malignancies.10 With modern CT–based planning of photon beams, osteoradionecrosis of the mandible is now very rare.26
With regard to cosmesis, long-term skin changes are unusual, given the lower radiotherapy doses. Telangiectasia is a possible late effect, which may be a concern for the predominantly female patients, especially if a bolus is required when the skin is thought to be at risk. In male patients, there may be loss of facial hair in the beard area.
 Toxicities using Alternative Radiation Techniques
Toxicities using Alternative Radiation Techniques
Particle therapy, utilizing neutrons, protons, or carbon ions, is a theoretical option where available. The lack of exit dose from proton radiotherapy is appealing as a way of reducing normal tissue toxicity. However, neutrons have generally been abandoned due to the high incidence of grade III/IV complications. Douglas et al. reported on a series of 16 patients treated between 1986 and 1993 (University of Washington, Seattle).48 The median dose was 18.9 neutron Gy (range 17–22 neutron Gy). Two patients were lost to follow-up, but the remaining 14 patients had local tumor control at a median follow-up of 83 months. At 15 years, the local control rate was 85%, one patient having died from a skull base recurrence. One other patient developed osteoradionecrosis, and the 15-year rate of Radiation Therapy Oncology Group/European Society for Therapeutic Radiology and Oncology (RTOG/ESTRO) nonaudiological grade III/IV complications was 21%.48
 All the evidence points to complete surgical excision being the optimal treatment for benign salivary gland tumors.
All the evidence points to complete surgical excision being the optimal treatment for benign salivary gland tumors.
 In difficult surgical cases, such as multinodular recurrent disease, in which all the tumor cells cannot be removed and further recurrence is likely, adjuvant radiotherapy should be considered.
In difficult surgical cases, such as multinodular recurrent disease, in which all the tumor cells cannot be removed and further recurrence is likely, adjuvant radiotherapy should be considered.
 Patient management is now generally discussed in a multidisciplinary team setting in which the particular features of the resection can be discussed between the head and neck surgeon and the radiation oncologist.
Patient management is now generally discussed in a multidisciplinary team setting in which the particular features of the resection can be discussed between the head and neck surgeon and the radiation oncologist.
 As the mechanisms of PSA recurrence are not yet fully elucidated, there is a view that the benefit of adjuvant radiotherapy for a benign condition does not outweigh the potential risk to the patient.28
As the mechanisms of PSA recurrence are not yet fully elucidated, there is a view that the benefit of adjuvant radiotherapy for a benign condition does not outweigh the potential risk to the patient.28
 Immediate postoperative irradiation appears to be more successful and is associated with less toxicity than irradiation at recurrence.33
Immediate postoperative irradiation appears to be more successful and is associated with less toxicity than irradiation at recurrence.33
 Modern radiotherapy planning and delivery, along with refinements in the surgical approach, should significantly enhance local control rates while simultaneously reducing the late effects of therapy.
Modern radiotherapy planning and delivery, along with refinements in the surgical approach, should significantly enhance local control rates while simultaneously reducing the late effects of therapy.
< div class='tao-gold-member'>
Stay updated, free dental videos. Join our Telegram channel

VIDEdental - Online dental courses


 The indications for radiotherapy may include unclear surgical margins, tumor spillage, and recurrent pleomorphic adenoma of the salivary glands.
The indications for radiotherapy may include unclear surgical margins, tumor spillage, and recurrent pleomorphic adenoma of the salivary glands.