Fig. 3.1
Retrograde cavity filled with Mineral Trioxide Aggregate (MTA) showing the formation of mineralized tissue close to the filling material and periapical repair. Original magnification × 100. H&E (Reprinted from Tanomaru-Filho et al. [173] copyright 2006, with permission from Elsevier)
The periapical tissue response of other retrofilling materials such as intermediate restorative material (IRM), thermoplasticized gutta-percha, reinforced zinc oxide cement (super EBA), and mineral trioxide aggregate (MTA) was studied in a dog model. Better reestablishment of the periapical tissues was observed with IRM and MTA. New hard tissue formation was observed directly over the material surface only in the MTA sections. MTA exhibited the most favorable periapical tissue response (Fig. 3.2) when comparing the biocompatibility with other tested materials [192].

Fig. 3.2
Histologic section (magnification × 4) of a retrograde cavity filled with MTA in dog’s teeth shows newly formed cement (NRC) growing from the periphery over the mineral trioxide aggregate surface and islands of newly formed bone (NFB) in close vicinity to the mineral trioxide aggregate surface (Reprinted from Wälivaara et al. [192] copyright 2012, with permission from Elsevier)
MTA has also been designed to seal communication between pulp cavity and periodontium, and the histologic response to intentional perforation in the teeth’s furcations of dogs repaired immediately with amalgam or MTA showed that MTA is more suitable than amalgam for perforation repair, particularly when it is used immediately after perforation [73]. The periradicular tissue response to MTA exhibited less periradicular inflammation and more fibrous capsules adjacent to MTA in comparison to amalgam. In addition, the presence of cementum over the MTA surface was observed [176]. The healing process of intentional lateral root perforation repaired with mineral trioxide aggregate (MTA) was observed in dogs’ teeth (Fig. 3.3) showing no inflammation and deposition of cementum over MTA [88]. The biological response of the periodontium adjacent to furcation perforations in rat molars filled with Endo CPM Sealer, MTA Angelus, or zinc oxide-eugenol cement has also been evaluated. The greatest number of inflammatory cells was observed at 7 days, especially in the zinc oxide-eugenol (ZOE) group where the inflammatory process was related to a significant increase in the number of osteoclasts and, thereby, in an increase in the width of the periodontal space. At 60 days, no significant differences in osteoclast numbers among Endo CPM Sealer, MTA, and control groups were evident. The periodontal space was also significantly reduced in the experimental groups in comparison with the initial periods. The periodontium adjacent to perforations filled with MTA and Endo CPM Sealer exhibited clear evidence of reestablishment and thus demonstrated better biocompatibility than zinc oxide-eugenol (Fig. 3.4) [51]. Contaminations of lateral perforations can impair the healing process. Regardless the use of MTA, contaminated lateral root perforations immediately sealed with MTA presented worse repair than the non-contaminated ones [87].
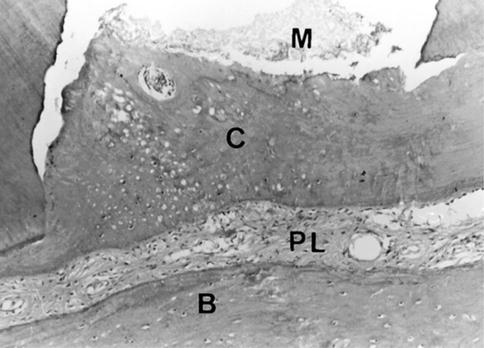
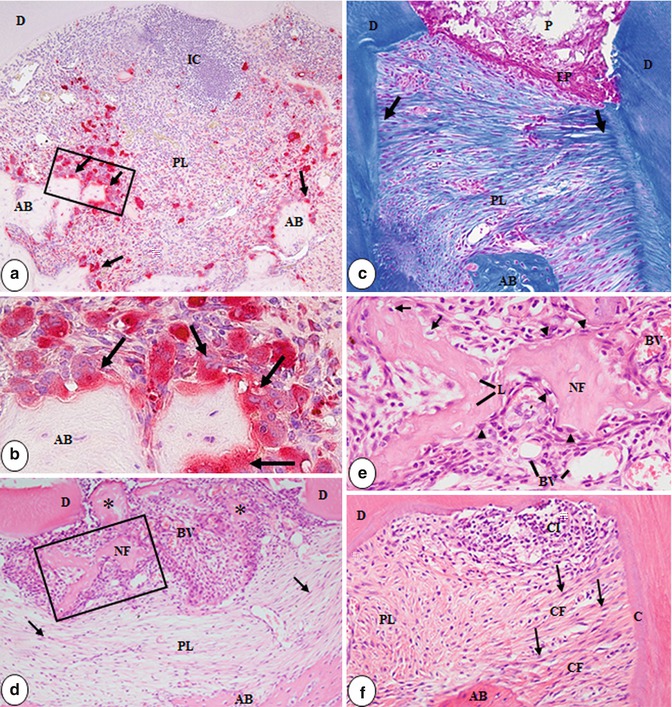

Fig. 3.3
Lateral root perforation repaired with mineral trioxide aggregate (MTA) in dogs’ teeth. Cementum (C) over the filling material (M), periodontal ligament (PL), and bone (B). Hematoxylin and eosin; original magnification 3100 (Reprinted from Holland et al. [88] copyright 2001, with permission from Elsevier)

Fig. 3.4
Light micrographs of furcation region of molar teeth perforated and filled with MTA and examined after 7 (a, b), 15 (c), 30 (d, e), and 60 days (f). In (a), numerous inflammatory cells (IC) are present in the wide periodontal space. Tartrate-resistant acid phosphatase (TRAP)-positive osteoclasts (arrows) are next to the alveolar bone (AB) surface. TRAP reaction counterstained with hematoxylin. D dentin, PL periodontal ligament (bar, 100 lm). In (b), outlined area of (a), TRAP-positive osteoclasts (arrows) are adjacent to the bone surface. AB, alveolar process (bar, 50 lm). (c) Shows a layer of two to three elongated cells (EP) between the perforation in the floor of the pulp chamber (P) and the periodontal ligament (PL). In the PL, some bundles of collagen fibers (CF) penetrate into the root surfaces (arrows). D, dentin; AB, alveolar process. Masson’s trichrome (bar, 100 lm). In (d), acidophilic and irregular material (NF) fills partially the perforation of the floor of the pulp chamber and is surrounded by numerous mononuclear cells and blood vessels (BV). Collagen fibers and fibroblasts (arrows) seem to be surrounding the material “NF.” PL, peridontal ligament; D, dentin; AB, alveolar process. H&E (bar, 100 lm). In (e), outlined area of (d), homogeneous material “NF” contains lacunae (L) and, sometimes, cells inside them (arrows). Elongated and rounded cells (arrowheads) are in close juxtaposition to the irregular surface of “NF.” BV, blood vessels. H&E (bar, 50 lm). In (f), some inflammatory cells (IC) are adjacent to the root surface. In the furcation region, fibroblasts (arrows) and bundles of collagen fibers (CF); some of them penetrate into the alveolar bone (AB) and cementum (C). D, dentin. H&E (bar, 100 lm) (Reprinted with permission from da Silva et al. [51] © 2010 International Endodontic Journal)
The systemic toxic effect of gray ProRoot Mineral trioxide aggregate (MTA) on the liver and kidney of rats was evaluated after 7 and 30 days and compared to DiaRoot BioAggregate. The materials were inserted into Teflon tubes and implanted subcutaneously. Blood samples were taken to investigate changes of kidney and liver functions. MTA induced side effects on the liver and kidney that were significantly more severe than BioAggregate (Verio Dental Co. Ltd., Vancouver, Canada) but with no permanent damage [97].
Clinical research on MTA is presented as specific case reports or randomized clinical trials. The case reports present a very low level of evidence due to lack of standardization as opposed to randomized clinical trials. From 1995 to date there have been 50 clinical trials on MTA. Most of the clinical trials were performed on treatment of primary teeth with a total of 27 reports (Table 3.1), 26 out of which MTA was used for pulpotomies and one report for MTA used as a direct pulp capping agent [186]. The clinical trials and outcome of clinical reports assessing the success rate of MTA used for the treatment of primary teeth are shown in Table 3.1. The assessment period ranged from 3 months to 84 months. Dentin bridge formation and absence of clinical and radiographic symptoms were used to assess the success rates. MTA was shown to exhibit same outcome as of other materials tested in 17 from the 26 cases reviewed. Two studies did not use a control material but assessed MTA only; thus, no comparisons could be made [110, 118]. Calcium hydroxide and MTA may be suitable materials for the treatment of the immature teeth. However, more studies evaluating the factors influencing success and failure in teeth should be conducted [46].
Table 3.1
Clinical trials evaluating the success rates of MTA used in treatment of primary teeth
|
Year
|
Author
|
Assessment period (months)
|
Control material
|
Outcome
|
|---|---|---|---|---|
|
2013
|
Mehrdad et al. [114]
|
24
|
CEM
|
Same; calcific bridge formation
|
|
2012
|
Sushynski et al. [171]
|
6–24
|
FC
|
Gray MTA better than formocresol
|
|
Fernández et al. [72]
|
24
|
FC, FS, NaOCl
|
Same
|
|
|
Odabaş et al. [127]
|
12
|
FS
|
Same
|
|
|
Airen et al. [4]
|
6, 12, 24
|
FC
|
MTA better
|
|
|
2011
|
Liu et al. [105]
|
/
|
Ca(OH)2
|
MTA better; crown discoloration noted
|
|
Malekafzali et al. [108]
|
12, 24
|
CEM
|
Same
|
|
|
Srinivasan and Jayanthi [167]
|
3, 6, 9, 12
|
FC
|
MTA better
|
|
|
Erdem et al. [66]
|
6, 12, 24
|
FC, FS, ZnO
|
Same. MTA better than ZnO
|
|
|
Cardoso Silva et al. [39]
|
84
|
GMTA, WMTA
|
Same, gray better
|
|
|
2010
|
Zealand et al. [196]
|
6
|
FC
|
Same; MTA 22 % dentin bridge formation
|
|
Ansari and Ranjpour [11]
|
6, 12, 24
|
FC
|
Same
|
|
|
Doyle et al. [60]
|
24
|
FS (±eugenol)
|
MTA better; outcome worse with eugenol free FS
|
|
|
2009
|
Subramaniam et al. [170]
|
24
|
FC
|
MTA better
|
|
Sakai et al. [154]
|
6, 12, 18, 24
|
Portland cement
|
Same; Portland cement caused more pulp obliteration
|
|
|
2008
|
Sonmez et al. [166]
|
24
|
FC, FS, Ca(OH)2
|
Same; Ca(OH)2 was the least appropriate
|
|
Moretti et al. [117]
|
3, 6, 12, 18, 24
|
FC, Ca(OH)2
|
MTA same as FC; better than Ca(OH)2
|
|
|
Noorollahian [124]
|
6, 12, 24
|
FC
|
Same
|
|
|
Tuna and Olmez [186]
|
1–24
|
Ca(OH)2
|
Same
|
|
|
2007
|
Aeinehchi et al. [2]
|
3, 6
|
FC
|
Same
|
|
2006
|
Percinoto et al. [135]
|
3, 6, 12
|
Ca(OH)2
|
Same
|
|
2005
|
Farsi et al. [70]
|
24
|
FC
|
Clinical outcome same; MTA better radiographically
|
|
Maroto et al. [110]
|
6
|
/
|
Dentin bridge formation; crown discoloration
|
|
|
Holan et al. [86]
|
38
|
FC
|
Same
|
|
|
Naik and Hegde [118]
|
6
|
/
|
Successful
|
|
|
2004
|
Agami et al. [3]
|
12
|
FC
|
Gray better than white; MTA better than FC
|
|
2001
|
Eidelman et al. [62]
|
6–30
|
FC
|
Same
|
There have been five reports on the use of MTA for direct pulp capping [1, 67, 69, 84, 93] and two for indirect pulp capping [103, 136]; four for apexification [52, 64, 158, 163] and six for apexogenesis [14, 16, 64, 78, 125, 138]; and five studies for use as a root-end filler [47, 48, 104, 165, 191] and one for perforation repair [77]. Taking into consideration the randomized clinical trials with specific MTA use, it can be concluded that MTA was shown to be a successful root-end filling material [156] and was comparable to IRM and super EBA cement [47, 104, 165]. Furthermore, the type of tooth treated did not affect the outcome of treatment. MTA was shown to be better than Retroplast as a root-end filling material [191]. High success rates were reported, and the need to use a root-end filling material as opposed to resection and burnishing gutta-percha was highlighted [48].
Pulp capping with MTA is recommended for teeth with carious pulp exposures especially immature teeth with high potential for healing [70]. When used as a pulp capping agent, MTA exhibited comparable clinical and radiographic success rates to calcium hydroxide [103, 136], while in other clinical studies, MTA was shown to have better clinical success rates than calcium hydroxide [67, 84]. Both ProRoot MTA and MTA Angelus exhibited similar success rates [1]. No statistically significant differences were found for superficial and deep inflammatory cell response, presence of a dentin bridge, and pulp vitality between MTA and calcium hydroxide. A statistically significant difference was found for the diameter of exposure. Only a minimal association between clinical and histological findings could be established for either material [93].
For treatment of immature permanent teeth, cases treated with MTA exhibited complete root formation earlier. A good success and an effective option for apexification with the advantage of reduced treatment time, good sealing ability, and biocompatibility providing a barrier for immediate obturation were demonstrated from MTA [52]. One-visit apexification procedures are possible with MTA [163]. Although MTA was more successful, it is more expensive, and dentin bridges developed over canal orifices, complicating future root canal therapy [78]. MTA was comparable to calcium hydroxide [16, 65, 138] and calcium-enriched mixture in treatment of immature permanent molars by pulpotomy [14, 125].
3.15 Conclusions
Although there are no standards for root-end filling materials or materials based on Portland cement to which MTA can conform, MTA was shown to exhibit adequate physical, chemical, antimicrobial, and biological properties which are comparable to other dental materials used for the same purpose. The clinical studies on MTA demonstrate similar data to the in vitro studies.
References
1.
Accorinte ML, Loguercio AD, Reis A, Bauer JR, Grande RH, Murata SS, Souza V, Holland R. Evaluation of two mineral trioxide aggregate compounds as pulp-capping agents in human teeth. Int Endod J. 2009;42(2):122–8.PubMed
2.
Aeinehchi M, Dadvand S, Fayazi S, Bayat-Movahed S. Randomized controlled trial of mineral trioxide aggregate and formocresol for pulpotomy in primary molar teeth. Int Endod J. 2007;40(4):261–7.PubMed
3.
Agamy HA, Bakry NS, Mounir MM, Avery DR. Comparison of mineral trioxide aggregate and formocresol as pulp-capping agents in pulpotomized primary teeth. Pediatr Dent. 2004;26(4):302–9.PubMed
4.
Airen P, Shigli A, Airen B. Comparative evaluation of formocresol and mineral trioxide aggregate in pulpotomized primary molars – 2 year follow up. J Clin Pediatr Dent. 2012;37(2):143–7.PubMed
5.
Akbari M, Rouhani A, Samiee S, Jafarzadeh H. Effect of dentin bonding agent on the prevention of tooth discoloration produced by mineral trioxide aggregate. Int J Dent. 2012;2012:563203.PubMedCentralPubMed
6.
Alanezi AZ, Zhu Q, Wang YH, Safavi KE, Jiang J. Effect of selected accelerants on setting time and biocompatibility of mineral trioxide aggregate (MTA). Oral Surg Oral Med Oral Pathol Oral Radiol Endod. 2011;111(1):122–7.PubMed
7.
Al-Hezaimi K, Al-Shalan TA, Naghshbandi J, Oglesby S, Simon JH, Rotstein I. Antibacterial effect of two mineral trioxide aggregate (MTA) preparations against Enterococcus faecalis and Streptococcus sanguis in vitro. J Endod. 2006;32(11):1053–6.PubMed
8.
Al-Hezaimi K, Al-Shalan TA, Naghshbandi J, Simon JH, Rotstein I. MTA preparations from different origins may vary in their antimicrobial activity. Oral Surg Oral Med Oral Pathol Oral Radiol Endod. 2009;107(5):e85–8.PubMed
9.
Al-Nazhan S, Al-Judai A. Evaluation of antifungal activity of mineral trioxide aggregate. J Endod. 2003;29(12):826–7.PubMed
10.
Andreasen JO, Munksgaard EC, Bakland LK. Comparison of fracture resistance in root canals of immature sheep teeth after filling with calcium hydroxide or MTA. Dent Traumatol. 2006;22:154–6.PubMed
11.
Ansari G, Ranjpour M. Mineral trioxide aggregate and formocresol pulpotomy of primary teeth: a 2-year follow-up. Int Endod J. 2010;43(5):413–8.PubMed
12.
Antunes Bortoluzzi E, Jua´rez Broon N, Antonio Hungaro Duarte M, de Oliveira Demarchi AC, Monteiro Bramante C. The use of a setting accelerator and its effect on pH and calcium ion release of mineral trioxide aggregate and white Portland cement. J Endod. 2006;32:1194–7.PubMed
13.
Arruda RA, Cunha RS, Miguita KB, Silveira CF, De Martin AS, Pinheiro SL, Rocha DG, Bueno CE. Sealing ability of mineral trioxide aggregate (MTA) combined with distilled water, chlorhexidine, and doxycycline. J Oral Sci. 2012;54(3):233–9.PubMed
14.
Asgary S, Eghbal MJ. Treatment outcomes of pulpotomy in permanent molars with irreversible pulpitis using biomaterials: a multi-center randomized controlled trial. Acta Odontol Scand. 2013;71(1):130–6.PubMed
15.
Balto HA. Attachment and morphological behavior of human periodontal ligament fibroblasts to mineral trioxide aggregate: a scanning electron microscope study. J Endod. 2004;30(1):25–9.PubMed
16.
Barrieshi-Nusair KM, Qudeimat MA. A prospective clinical study of mineral trioxide aggregate for partial pulpotomy in cariously exposed permanent teeth. J Endod. 2006;32(8):731–5.PubMed
17.
Bates CF, Carnes DL, del Rio CE. Longitudinal sealing ability of mineral trioxide aggregate as a root-end filling material. J Endod. 1996;22(11):575–8.PubMed
18.
Belı´o-Reyes IA, Bucio L, Cruz-Chavez E. Phase composition of ProRoot mineral trioxide aggregate by X-ray powder diffraction. J Endod. 2009;35:875–8.
19.
Belobrov I, Parashos P. Treatment of tooth discoloration after the use of white mineral trioxide aggregate. J Endod. 2011;37(7):1017–20.PubMed
20.
Bernabé PF, Gomes-Filho JE, Bernabé DG, Nery MJ, Otoboni-Filho JA, Dezan-Jr E, Cintra LT. Sealing ability of MTA used as a root end filling material: effect of the sonic and ultrasonic condensation. Braz Dent J. 2013;24(2):107–10.PubMed
21.
Bidar M, Naderinasab M, Talati A, Ghazvini K, Asgari S, Hadizadeh B, Gharechahi M, Mashadi NA. The effects of different concentrations of chlorhexidine gluconate on the antimicrobial properties of mineral trioxide aggregate and calcium enrich mixture. Dent Res J (Isfahan). 2012;9(4):466–71.
22.
Bortoluzzi EA, Araújo GS, Guerreiro Tanomaru JM, Tanomaru-Filho M. Marginal gingiva discoloration by gray MTA: a case report. J Endod. 2007;33(3):325–7.PubMed
23.
Bortoluzzi EA, Souza EM, Reis JM, Esberard RM, Tanomaru-Filho M. Fracture strength of bovine incisors after intra-radicular treatment with MTA in an experimental immature tooth model. Int Endod J. 2007;40(9):684–91.PubMed
24.
Bramante CM, Kato MM, Assis GF, Duarte MA, Bernardineli N, Moraes IG, Garcia RB, Ordinola-Zapata R, Bramante AS. Biocompatibility and setting time of CPM-MTA and white Portland cement clinker with or without calcium sulfate. J Appl Oral Sci. 2013;21(1):32–6.PubMedCentralPubMed
25.
Bueno CE, Zeferino EG, Manhães Jr LR, Rocha DG, Cunha RS, De Martin AS. Study of the bismuth oxide concentration required to provide Portland cement with adequate radiopacity for endodontic use. Oral Surg Oral Med Oral Pathol Oral Radiol Endod. 2009;107(1):e65–9.PubMed
26.
Camargo CH, Fonseca MB, Carvalho AS, Camargo SE, Cardoso FG, Valera MC. Microhardness and sealing ability of materials used for root canal perforations. Gen Dent. 2012;60(6):e393–7.PubMed
27.
Camilleri J. Hydration mechanisms of mineral trioxide aggregate. Int Endod J. 2007;40:462–70.PubMed
28.
Camilleri J. Characterization of hydration products of mineral trioxide aggregate. Int Endod J. 2008;41:408–17.PubMed
29.
Camilleri J. Evaluation of the physical properties of an endodontic Portland cement incorporating alternative radiopacifiers used as root-end filling material. Int Endod J. 2010;43:231–40.PubMed
30.
Camilleri J. Evaluation of the effect of intrinsic material properties and ambient conditions on the dimensional stability of white mineral trioxide aggregate and Portland cement. J Endod. 2011;37(2):239–45.PubMed
31.
Camilleri J. The color stability of white mineral trioxide aggregate in contact with hypochlorite solution. J Endod. 2014;40(3):436–40.PubMed
32.
Camilleri J, Gandolfi MG. Evaluation of the radiopacity of calcium silicate cements containing different radiopacifiers. Int Endod J. 2010;43(1):21–30.PubMed
33.
Camilleri J, Kralj P, Veber M, Sinagra E. Characterization and analyses of acid-extractable and leached trace elements in dental cements. Int Endod J. 2012;45(8):737–43.PubMed
Stay updated, free dental videos. Join our Telegram channel

VIDEdental - Online dental courses


