(1)
Department of Orthodontics, Universidad Intercontinental, Mexico, D.F., Mexico
Abstract
The most frequent iatrogenic problem in orthodontics is white spot lesions (WSLs). Maintenance of an adequate and constant presence of fluoride ions in the vicinity of the enamel on the periphery of bracket bases helps protect against the development of WSLs. Resin-modified glass ionomer cements (RMGICs) minimize the development of WSLs due to their continuous fluoride uptake from the environment and re-release, but the perception that orthodontic brackets bonded with RMGICs frequently fail due to low initial bracket shear bond strength persists. This perception is correct if the clinician were to bond these materials as recommended by the manufacturers, which includes conditioning the enamel with an extremely weak 10 % polyacrylic acid instead of the traditional 37 % phosphoric acid etch. In order to successfully use resin-modified glass ionomer cements, it is recommended to first deproteinize the enamel surface by applying 5.25 % sodium hypochlorite for 1 min to remove the acquired dental pellicle (which impedes proper etching of the enamel surface), followed by a 15–30-s etching with 37 % phosphoric acid (so the resulting etch patterns on the enamel surface are types 1 and 2, not type 3, thereby increasing bracket bond strength), and followed by moistening the enamel surface to further increase this strength. In following these recommendations, the clinician will minimize the risk to patients of developing lifelong WSLs and their consequences. In the final analysis the degree of damage provoked by WSLs is vastly more significant on the health of the enamel than the bonding and debonding process. It is the author’s hope that this review of the scientific literature will help clinicians achieve the best results for their patients.
1.1 Introduction
Orthodontic treatment success can be jeopardized by the development of enamel white spot lesions (WSLs) on the periphery of, or beneath, orthodontic bracket bases. WSLs are clinically defined as opaque, white areas caused by the loss of minerals below the outermost enamel layer [1]. WSLs are the earliest sign of the caries process, which starts with enamel demineralization. The severity of WSLs can be classified numerically using a 4-point scale (Gorelick’s scale) in which # 1 shows no enamel demineralization, # 2 slight, # 3 severe, and # 4 cavitation [2].
Brackets and bands create areas on the tooth surfaces which favor the increase of plaque and food accumulation due to the restrictive access for self-cleaning [3]. Carbohydrate fermentation by the bacteria in the dental plaque causes a decrease in the pH. This in turn results in the loss of mineral ions from the enamel to the oral environment, a process known as demineralization. Through the buffering action of saliva, the pH can increase again allowing the teeth to incorporate free ions. This process is called remineralization. There is a constant ionic exchange between the dental tissues and the environment, which will ultimately reach an ionic equilibrium. Progressive demineralization without adequate remineralization results in the development of WSLs [4, 5].
The formation of WSLs remains an unfortunately common complication during orthodontic treatment and is particularly prevalent in patients with poor oral hygiene. The initial lesions can be visible as soon as the 4th week after the placement of fixed orthodontic appliances [6]. Richter et al. [7] reported that nearly 75 % of patients who underwent comprehensive orthodontic treatment developed new WSLs because of prolonged plaque retention on the periphery of bracket bases. Therefore, the first step to prevent WSLs is to educate patients on the importance of maintaining proper oral hygiene throughout their orthodontic treatment. It is the orthodontist’s responsibility to minimize the risk of patients developing enamel demineralizations as a consequence of orthodontic treatment. This can be achieved by brushing with fluoride-containing toothpaste after every meal and snack, rinsing daily with fluoride-containing mouthwash and by dietary modification, and limiting contact with sugar-containing products. Unfortunately, patient compliance is generally quite poor, particularly among the adolescent population.
The inhibitory effect of fluoride on bacterial activity and on demineralization of enamel has been well established [8]. The main mechanism by which fluoride works is by maintaining the plaque supersaturated with respect to fluorapatite, hence tipping the balance of the caries process against demineralization and in favor of remineralization [9]. Thus, continuous contact with fluorides is critical to protect the enamel against the development of WSLs during treatment with fixed orthodontic appliances. The presence of fluoride will minimize the ionic loss from the tooth structure until the pH of the plaque becomes as low as 4.5. At that level, even the presence of adequate fluoride concentration in the oral environment will have a minimal beneficial effect on the process of remineralization [10]. But before reaching such a critically low pH level, the availability of fluoride ions in the oral environment will enhance remineralization [11]. Resin-modified glass ionomer cements (RMGICs) can be used to bond orthodontic brackets. They have an anticariogenic effect because they release fluoride into the immediate environment of the bracket base, and they do so in a sustained fashion [12]. Therefore, maintaining an adequate and constant presence of fluoride ions in the vicinity of the enamel is critical to the ability of enamel to remineralize. It has been suggested that fluorides will have a substantial inhibitory effect on the rate of demineralization of enamel even if it is in the sub-ppm level, i.e., as low as 0.02–0.06 ppm [13].
The frequent application of fluorides is the most efficient method for preventing demineralization as well as enhancing remineralization of carious lesions. A potential method of providing a sustained concentration of fluoride ions over a prolonged period is to have a slow fluoride-releasing system incorporated in the bracket bonding material. Furthermore, the ability of some bonding materials for absorbing fluoride and then releasing it, acting as a fluoride pump, will help interrupt the development of WSLs, thus decreasing the risk of caries [14]. As a result, a few fluoride-releasing bonding systems have been developed. The fluoride ions released from these materials penetrate and diffuse into the tooth structure and prevent WSLs by reinforcing the mineral content of the tooth structure. The most effective fluoride-releasing materials in a descending order are glass ionomer cements (GICs), resin-modified glass ionomer cements (RMGICs), and compomers [14].
Resin composites to which fluoride has been added, called compomers, were not found to be effective in providing a sustained rate of fluoride ions [15, 16]. Thus, GICs were originally recommended as bracket adhesives to minimize, or even prevent, WSLs from developing due to their continuous fluoride release, but frequent bracket failure occurred due to their low initial shear bond strength (SBS). The reason for this low initial SBS is that glass ionomers harden through a slow acid-base setting reaction which requires 24 h to complete [17]. To increase the initial SBS of the GICs, 4–6 % photosensitive composite resin was added to be able to obtain a faster initial hardening of the adhesive’s resinous portion through photo-curing [18]. The addition of this resinous component converted GICs into RMGICs. These hybrid adhesives have allowed orthodontists to take advantage of the positive features of conventional GICs combining them with the mechanical and physical properties of resin composites.
Thus, the use of RMGICs to bond brackets is highly recommended because of their continuous fluoride uptake from the environment and re-release. However, these cements, in spite of the added resinous component, still have a relatively low initial bracket SBS [17].
To increase the initial bracket SBS to clinically reliable levels when using RMGICs, removal of organic material (deproteinization) from the enamel surface with 5.25 % sodium hypochlorite is highly recommended [19]. By applying 5.25 % sodium hypochlorite for 1 min, prior to phosphoric acid etching, temporary elimination of the acquired pellicle from the enamel surface occurs. This in turn allows the phosphoric acid to etch the enamel surface more effectively creating better etching patterns which increase bracket SBS [20–22].
The important topic of the acquired pellicle is presented later in this chapter. For the moment suffice to state that professional tooth cleaning by the use of a rubber cup or rotary brush with pumice does not completely remove the pellicle from the enamel surface; that the pellicle layer on the enamel surface confers resistance against chemical dissolution and attack by acidic agents, so 37 % phosphoric acid is not able to etch the enamel surface in areas covered with the organic material of the pellicle; and that temporary elimination (deproteinization) of the acquired pellicle from the enamel surface where the bracket base will be bonded is a must to be able to obtain etching patterns that allow effective bracket bonding with RMGICs.
To further increase bracket SBS, GC Corp., Tokyo, Japan, the manufacturer of Fuji Ortho LC, an RMGIC, recommends moistening the enamel surface with a water-moistened cotton roll before bonding, as verified by Rodríguez [23].
Clinicians typically require that a bonding material have sufficient initial bracket SBS to be able to tie arch wires into the brackets immediately after having bonded them, but since the glass ionomer fraction of RMGICs takes 24 h to set, clinicians prefer to continue using resin composites for bonding brackets to lower the risk of bracket bond failures, even if they do not provide a sustained fluoride release to protect the enamel from developing WSLs.
In the following subchapter, the reader will become aware of how severe and widespread the incidence and prevalence of WSLs are in individuals who had orthodontic treatment and will hopefully decide to incorporate the recommended modified method of RMGIC use to protect orthodontic patients against WSL development.
1.2 Detection
Among the most common methods of detection are clinical inspection and photographs. Quantitative light-induced fluorescence (QLF) has been recently suggested as a more accurate method of detecting WSLs [24]. The QLF method consists of illuminating the teeth with a blue laser light. Tooth dentin contains atoms called fluorophores which fluoresce green when illuminated with a blue laser light. This green light is blocked from exiting the enamel where a WSL is present. Thus, the WSL appears as a black area surrounded by green color. QLF technology is expensive so it is not widely used.
WSL detection can be a challenge when enamel decalcifications are in their initial stages. Before orthodontic treatment begins, the clinician should document the extent and severity of any WSL present through clinical inspection and with the aid of intraoral photographs. These photographs can be used for comparative purposes both during and at the end of treatment for patient education as well as for the documentation of their presence.
1.3 Incidence
A review of the literature indicates that there is a high incidence of WSLs that develop during comprehensive orthodontic treatment. Richter et al. [7], using the photographic method to detect WSLs, found that 72.9 % of 350 orthodontic patients treated with comprehensive orthodontics between 1997 and 2004 in the Department of Orthodontics at the University of Michigan had developed new WSLs. These 350 patients were selected at random from the photographic records of 2,300 patients treated at that institution. Boersma et al. [25], using the quantitative light-induced fluorescence method to detect WSLs, found that 97 % of patients who were evaluated immediately following comprehensive orthodontic treatment were affected with WSLs.
The next subchapter will address the question of WSL prevalence. It is an important subject because some reports indicate that a natural remineralization process occurs after orthodontic treatment which diminishes the prevalence of WSLs.
1.4 Prevalence
A review of the literature reveals that in spite of some WSL natural remineralization occurring post-orthodontic bracket removal, these lesions generally do not disappear. Van der Veen et al. [26] used the quantitative light-induced fluorescence method to determine whether WSLs diminish after orthodontic treatment (through the natural remineralization process). They found that 6 months after bracket debonding, while 33 % of WSLs did remineralize somewhat (lesion regression), the majority of WSLs remained unchanged, and 10 % worsened (lesion progression). Ogaard [27], in a study of 51 patients treated with comprehensive orthodontics, used clinical inspection to detect WSLs and found that the prevalence of WSLs on vestibular surfaces 5 years posttreatment was significantly higher than in a matched control sample of untreated individuals.
The results from the abovementioned studies indicate why methods of prevention or treatment for WSLs must be strongly considered.
1.5 Treatment
Methods to treat WSLs post-orthodontic treatment can be divided, according to Guzman et al. [28], into conservative and aggressive. The conservative treatment methods include oral hygiene instruction, dietary modification, chewing gum to increase salivary output (preferably gum containing xylitol, not sugar), remineralization with fluorides (in dentifrices, in varnishes, in sealants, and in mouth rinses with low-concentration solutions of less than 50 ppm), the use of antimicrobials (chlorhexidine), and casein derivatives (although recent research by Huang et al. [29] casts doubt regarding casein derivatives’ effectiveness in remineralizing WSLs).
More aggressive treatment methods suggested by Guzman et al. [28] include external bleaching, micro-abrasion, composite restorations, and porcelain veneers.
Clinicians should recognize that the best policy is to prevent WSLs from occurring since the methods mentioned in the last paragraph are quite aggressive. It is therefore best to allow the natural remineralization process to take place as described in the next subchapter.
1.6 Natural Remineralization
Before attempting to use any of the WSL aggressive treatment methods, Guzman et al. [28] and Bishara and Ostby [30] recommended allowing the natural remineralization process to occur. Saliva contains minerals including calcium, phosphates, and fluoride ions, all of which help to remineralize WSLs [4, 5, 8–14]. During the potential natural remineralization period, good oral hygiene is essential. A reduction of exposure to sugared beverages should be implemented. Chewing sugarless gum is also recommended because it stimulates saliva production. In addition, daily oral rinses with low fluoride concentration and consumption of fluoridated water can be helpful. This should be combined with brushing teeth with low-concentration fluoride dentifrices to promote the remineralization process. Although it remains controversial whether high or low fluoride concentrations should be used, low fluoride concentration is generally recommended so that the external enamel surface remains permeable for the minerals in saliva to penetrate the damaged enamel surface [29].
It has been reported that 33 % of the WSLs improve with time as long as there are no enamel cavitations [26]. Unfortunately, many WSLs persist years after orthodontic treatment, in spite of the natural remineralization process [27]. Based on these facts, prevention or minimization of WSL development during the course of orthodontic treatment should be regarded as a factor of critical importance.
1.7 Prevention
Many methods have been proposed to prevent or minimize WSL development during orthodontic treatment. These can be divided into compliant and noncompliant methods [28].
1.7.1 Compliant Methods
Compliant methods include maintenance of good oral hygiene using fluoride-containing dentifrices, brushing well immediately after every meal and snack, diet modification to limit contact with sugar-containing products, and daily oral rinses containing fluorides. Continuous contact with fluoride is important because it protects the enamel by converting hydroxyapatite into fluorapatite crystals, which have a lower solubility in the oral environment than hydroxyapatite crystals [30].
In a systematic review published by Benson et al. [31], the researchers concluded that there is some evidence that the daily use of .05 % NaF mouth rinse, or bonding brackets with a glass ionomer cement, might reduce the occurrence and severity of WSLs during orthodontic treatment.
Topical application of stannous fluoride in particular may have a plaque-inhibiting effect by interfering with the adherence of plaque bacteria to the enamel surface. Tin atoms in stannous compounds also block the passage of sucrose into bacterial cells, thereby inhibiting acid production and diminishing the acidogenicity of plaque [32].
More recently, it has been suggested that the compound casein phosphopeptide-amorphous calcium phosphate (CPP-ACP) may also reduce the incidence of demineralization. The theoretical basis of this arose from the observation that dairy products are anticariogenic [33]. They work in a way similar to fluoride by maintaining the saturation of calcium and phosphate in plaque fluid, thereby discouraging the dissolution of these elements and also promoting remineralization if they are lost [34].
Unfortunately, compliant methods require patient cooperation, which is frequently lacking. Geiger et al. [35, 36] evaluated patient compliance in a preventive fluoride-rinse program. The degree of compliance with the home-care preventive protocol was poor in half of the patients, and less than 15 % of orthodontic patients used daily fluoride rinses as instructed.
1.7.2 Noncompliant Methods
1.7.2.1 RMGICs
In view of well-documented poor patient compliance, RMGICs have been proposed as bracket bonding materials due to their continuous fluoride-releasing properties throughout the orthodontic treatment.
RMGICs act as fluoride pumps due to the fact that they continuously absorb fluoride from the environment (e.g., fluoride in dentifrice, in oral rinse, and in potable fluoridated water) and subsequently re-release it precisely in the areas most susceptible to WSLs. These are the bracket perimeter and voids beneath the bracket base. In vivo [37, 38], ex vivo [39, 40], and in vitro [41] studies plus systematic reviews [31, 42] have documented that RMGICs do protect the enamel from the development of WSLs. These studies confirm that less demineralization occurs during fixed orthodontic appliance treatment with RMGICs than with traditional resin-based adhesives.
It is the author’s opinion that orthodontic care should include protection of enamel from developing WSLs. This implies using fluoride-releasing RMGICs as bracket bonding agents. However, the current recommended method by the manufacturer needs to be modified to increase bracket SBS to clinically reliable levels. This can be achieved by deproteinizing the enamel surface with 5.25 % sodium hypochlorite (NaOCl) for 1 min, etching the enamel surface with a 15–30-s application of 37 % phosphoric acid, and moistening the enamel surface with a water-moistened cotton roll (the rationale for these three steps is explained in the following subchapters). Figures 1.1, 1.2, 1.3, 1.4, 1.5, and 1.6 show posttreatment intraoral photographs of patients who were treated by the author. These patients had bicuspid extractions followed by comprehensive orthodontic treatment using brackets cemented with Fuji Ortho LC, an RMGIC. It can be observed that no WSLs developed.
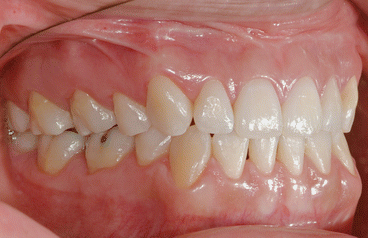
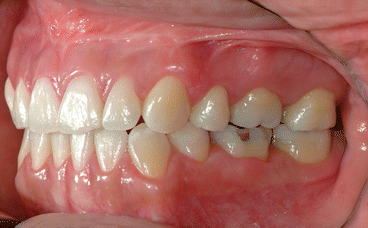
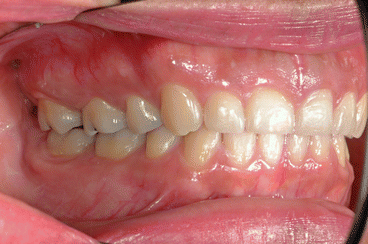
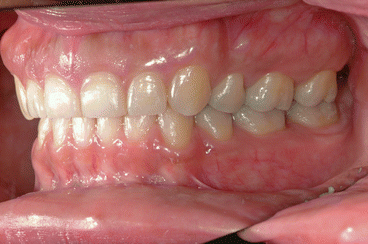
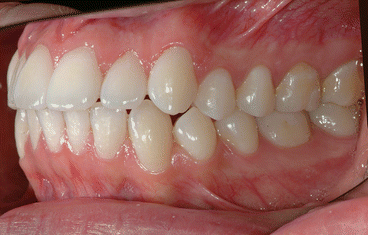
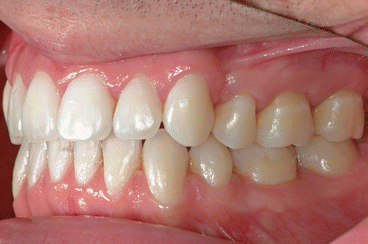

Fig. 1.1
Posttreatment intraoral right side view of a patient treated with extraction of four first bicuspids and full fixed orthodontic appliances for 24 months using Fuji Ortho LC as a bracket bonding agent. No WSLs can be observed

Fig. 1.2
Posttreatment intraoral left side view of the same patient

Fig. 1.3
Posttreatment intraoral right side view of a patient treated with extraction of four first bicuspids and full fixed orthodontic appliances for 28 months using Fuji Ortho LC as a bracket bonding agent. No WSLs can be observed

Fig. 1.4
Posttreatment intraoral left side view of the same patient

Fig. 1.5
Pre-treatment intraoral photograph of a patient who had previously been treated orthodontically using non-extraction therapy. The patient complained about having developed a double protrusion. In addition, the patient experienced three WSLs during this first treatment. These can be observed in the cervical third of the crowns of the second maxillary bicuspid and on the first and second maxillary molars

Fig. 1.6
Posttreatment intraoral photograph of the same patient after orthodontic re-treatment with extraction of four first bicuspids and full fixed orthodontic appliances for 30 months using Fuji Ortho LC as a bracket bonding agent. The same WSLs can be observed with no progression whatsoever and no new WSLs
In addition to the use of fluoride-releasing RMGICs as bracket bonding materials, three other materials that also release fluorides include the application of fluoride-releasing varnish on the periphery of the bracket bases, the use of fluoride-releasing composite resin as a bracket bonding agent, and the use of fluoride-releasing sealants. These products can also be employed to help reduce the development of WSLs.
1.7.2.2 Fluoride-Releasing Varnishes
Schmit et al. [43] carried out an ex vivo study to evaluate the effect of a fluoride-releasing cavity varnish on inhibition of enamel demineralization adjacent to orthodontic brackets bonded with a composite resin (Transbond XT) and with an RMGIC (Fuji Ortho LC). Brackets were bonded to 48 extracted human third molars. Half were bonded with the composite resin and half with the RMGIC. Each group was further divided into 2, with half receiving an application of Duraflor (Pharmascience, Montreal, Quebec, Canada), a fluoride-releasing varnish. The samples were cycled in an artificial caries solution for an hour twice daily for 31 days. After each caries challenge, the teeth were brushed with a soft toothbrush to simulate normal mechanical wear of the varnish. The loss of fluoride varnish was timed. Teeth were sectioned longitudinally and photographed under polarized light microscopy. Mean lesion depth was measured. The authors reported that teeth bonded with composite resin showed a 35 % reduction in mean lesion depth when fluoride-releasing varnish was applied. Teeth bonded with Fuji Ortho LC showed no significant differences in lesion depth between varnish and non-varnish groups. Both RMGIC groups had 50 % smaller mean lesion depth when compared with the composite resin group without fluoride varnish. Samples bonded with RMGIC had lesion depths shallower near the bracket; depths increased as lesions extended farther from the bracket, illustrating a wedge effect—the protective effect to the enamel of the fluoride released from the RMGIC decreased with the increasing distance from the bracket. The protective effect of the fluoride released from the RMGIC decreased from the edge of the bracket out to 1 mm away from the bracket. The hypothesis that RMGIC, when used to cement orthodontic brackets to human enamel, can significantly reduce the depth of an enamel demineralization lesion adjacent to the bracket when compared with a composite resin-cemented bracket was supported in this study. The results suggest that RMGIC is more effective at inhibiting enamel demineralization around the bracket base than is the fluoride varnish. The varnish was removed from the teeth by brushing in just a few days; so it has to be frequently reapplied and it has a brown color that patients might find objectionable.
Thus, monthly application of fluoride-releasing varnishes is not necessary when RMGICs are used, but essential when composite resin adhesives are used, particularly in patients with inadequate oral hygiene. Guzman et al. [28] also suggested monthly application of fluoride-releasing varnish around brackets bonded with composite resins as a method to prevent WSLs in case of poor patient compliance with using preventive protocols at home.
Stecksén-Blicks et al. [44] carried out a clinical study whose aim was to evaluate the efficacy of topical fluoride varnish applications on WSL formation in adolescents during treatment with fixed orthodontic appliances, using brackets bonded with composite resin adhesive (Victory Twin APC II 3M Unitek, Monrovia, USA). The study design was a double-blinded randomized placebo-controlled trial with two parallel arms. The subjects were 273 consecutive orthodontic patients aged 12–15 years, who were randomly allocated to either the experimental or control groups with topical applications of either a fluoride varnish (Fluor Protector, Ivoclar Vivadent, Schaan, Liechtenstein) or a placebo varnish every 6th week during the treatment period. The outcome measures at debonding were incidence and progression of WSL on the upper incisors, cuspids, and premolars as scored from before and after digital photographs by two experienced and calibrated independent clinician judges. The sample attrition rate was 5 %. The mean number of varnish applications was 10 (range 4–20) in both groups. The incidence of WSLs during the treatment with fixed appliances was 7.4 % in the fluoride varnish compared to 25.3 % placebo group (p < 0.001). The mean progression score was significantly lower in the fluoride varnish group than in the placebo group, 0.8 ± 2.0 vs. 2.6 ± 2.8 (p < 0.001). The authors concluded that there were significantly fewer new WSLs in the patients that had the application of the fluoride varnish at each visit compared with the placebo varnish. The results strongly suggest that regular topical fluoride varnish applications during treatment with fixed appliances may reduce the development of WSLs adjacent to the bracket base and that application of fluoride varnish should be advocated as a routine measure in orthodontic practice.
Unfortunately, these frequent varnish applications demand extra time and work for both the orthodontist and the office staff and an increased cost to the patient, so the conclusion can be drawn that fluoride-releasing varnishes are not likely to be as efficiently used or as effective in protecting the enamel against WSLs as RMGICs.
1.7.2.3 Fluoride-Releasing Composite Resins
Composite resins with fluoride-releasing potential have been designed for bonding orthodontic attachments. They are called compomers or polyacid-modified composite resins [32]. Unfortunately, most of the fluoride from compomers is released and depleted during the first few days or weeks after bonding brackets. In addition, compomers do not have the critical ability to absorb fluoride ions from the environment [15, 16, 45–47]. Thus, fluoride-releasing composite resins do not provide the long-term cariostatic effect needed to prevent the development of WSLs.
In vitro data provided by Chin et al. [41] determined that RMGICs (Fuji Ortho LC and Ketac Cem μ) are more effective at inhibiting enamel demineralization adjacent to orthodontic brackets, because they maximized the release of fluoride and resulted in the least peri-bracket enamel demineralization in an artificial caries solution, when compared to a fluoride-releasing resin composite (Light Bond, Reliance Orthodontic Products, Itasca, IL).
Wilson and Donly [39] demonstrated that Fuji Ortho LC exhibits significant inhibition of demineralization adjacent to the bracket base, compared to Light Bond, a fluoride-releasing composite resin, and to Concise, a non-fluoride-releasing composite resin.
The conclusion can be therefore drawn that fluoride-releasing composite resins are not as effective in protecting the enamel against WSLs as RMGICs.
1.7.2.4 Fluoride-Releasing Sealants
Soliman et al. [48] measured the in vitro rate and amount of fluoride ions released from Pro Seal (Reliance Orthodontic Products Inc, Itasca, IL), a fluoride-releasing orthodontic sealant, over a period of 17 weeks. These researchers also measured the recharging ability of this sealant when fluoride ions were reintroduced into the environment. They found that this sealant did release fluoride ions. However, the release decreased significantly from a high of 0.07 ppm/week/mm2 to a low of 0.01 by the end of the 17th week. They also found that this sealant had the ability to be recharged with fluoride ions from a foaming solution of acidulated phosphate fluoride, but not from brushing with fluoridated toothpaste. Unfortunately, the sealant had to be frequently recharged with the acidulated phosphate fluoride because even though the mean fluoride ion release was 0.354 ppm/week/mm during the 1st week after the fluoride application, it decreased to 0.014 by the 8th week.
1.7.2.5 Non-fluoride-Releasing Sealants
Non-fluoride-releasing sealants have also been developed. The application of these sealants around and/or beneath orthodontic brackets has been proposed as a method to prevent caries in orthodontic patients. Filled resin sealants have shown better retention and increased resistance to mechanical abrasion than unfilled resin sealants. Gizani [49] conducted a systematic review to evaluate the effectiveness of orthodontic sealants in preventing the occurrence of enamel demineralization in patients with fixed orthodontic appliances during treatment. The results were contradictory, with half of the studies showing significant reductions in the incidence of enamel demineralization for the sealed teeth compared with the control group, while the remaining half did not show any important differences. Gizani stated that it was impossible to make any reliable recommendations on the usage of these sealants during orthodontic treatment for the prevention of WSL development.
A recent prospective trial of 62 patients was carried out by O’Reilly et al. [50] to determine the effectiveness of a non-fluoride-releasing orthodontic sealant (BisCover LV, Bisco, Schaumberg, Ill) to prevent WSLs for the full duration of orthodontic treatment with fixed appliances. The researchers found a slightly lower incidence of WSLs on treated teeth (13.5 %) compared with the control teeth (17.7 %). WSL severity was nearly the same for treated and control teeth. They concluded that the sealant did not prevent all WSLs for the full duration of treatment but did demonstrate a clinically small but statistically significant ability to prevent WSLs.
The conclusion can be drawn that orthodontic sealants, whether fluoride-releasing or not, have limited and contradictory evidence to suggest protection of the enamel against WSLs and are not as well supported by the literature as are RMGICs.
1.8 Composition of Resin-Modified Glass Ionomer Cements
The main component of Fuji Ortho LC powder (LC stands for “Light Cured”) is a finely ground fluoroaluminosilicate glass. The liquid contains polyacrylic acid, water, monomer, and an activator. The resin component of Fuji Ortho LC is a mixture of three monomers, with 2-hydroxyethyl-methacrylate (HEMA) being the major constituent. The HEMA provides for a sharp setting reaction of the material when exposed to visible light irradiation. In addition, a very small quantity of camphorquinone is contained in the liquid as a photoinitiator [51].
Among the advantages of RMGICs when used as bracket adhesives are the protection against the development of WSLs, the ability to bond in a humid environment (particularly useful when bonding brackets in the lower molar area), the ease of debonding brackets, the easier enamel cleanup since no primer is used, and the reduction of damage to the enamel surface during adhesive removal [51].
1.9 Setting Reaction of Fuji Ortho LC
The setting chemistry of Fuji Ortho LC, an RMGIC, differs from that of GICs, in that it involves both an acid-base reaction and a polymerization reaction. There are three stages involved in the setting reaction. The first stage is photopolymerization resulting from exposure to the light-curing unit (wavelength: 470 nm) which initiates free radical polymerization of HEMA, and two other monomers, to form a poly-HEMA matrix that hardens the material. This first stage, the formation of the poly-HEMA resin matrix, gives dimensional stability and an early set strength. The second stage reaction is a self-cure of the resin monomers. The third stage is the final setting of the acid-base reaction of the GIC fraction in the polymer matrix. The poly-HEMA and the polyacrylic metal salt ultimately form a homogeneous matrix that surrounds the glass particles. As a result the light-activated polymerization reaction is well harmonized with the acid-base reaction. Moisture contamination of the adhesive is encouraged in order to displace water-soluble monomers that may inhibit the glass ionomer set [52]. The light-initiated reaction also allows for early placement of arch wires, while the acid-base reaction occurs simultaneously, but far more slowly, and continues for a period of time after the mass has been cured by light irradiation.
1.10 Preparation of the Enamel Surface
Unfortunately, in spite of the addition of the photosensitive composite resin to the GICs, the initial SBS of RMGICs was still not high enough for clinical use. So it was suggested by Toledano et al. [53] that in order to increase the SBS, 37 % phosphoric acid be used for 15 s to etch the enamel surface instead of conditioning the enamel surface with the manufacturer-recommended 10 % polyacrylic acid for 20 s. These researchers found that brackets bonded with Fuji Ortho LC obtained similar SBS as composite resins 24 h after bonding, when the enamel surface was etched with 37 % phosphoric acid.
Ogaard et al. state that phosphoric acid applied to the enamel surface cleans the surface and dissolves the minerals to create multiple microporosities which result in micromechanical retentions; that it is important that the bonding material reach deeply into the etched areas and polymerize to give retention; that the bonding material must therefore be able to wet the surface, either by the surface having a higher tension than the bonding material or by using material that is sufficiently soluble in the components of the surface; and that phosphoric acid etching of the enamel creates this high surface tension that “sucks” the material into the roughened surfaces [32].
Thus, bonding brackets with RMGICs by etching the enamel surface with 37 % phosphoric acid, instead of conditioning it with polyacrylic acid, increases the bracket SBS during the critical 24-h setting time of the glass ionomer fraction in RMGICs so that the initial SBS is sufficient to withstand forces that otherwise would cause bracket failure.
Kakaboura and Vougiouklakis [54] have suggested that GICs do not adhere to the enamel through micromechanical retentions when conditioning the enamel with a 20-s, 10 % polyacrylic acid solution. They state that GICs bond chemically to the enamel through ionic bonding such as calcium bridges, hydrogen bonds, or van der Waals forces. These chemical bonds have a low initial SBS, however, which results in high initial bracket bond failure rates.
The recommendation by various researchers [19, 53] to etch the enamel surface, instead of conditioning it with 10 % polyacrylic acid, to create micromechanical retentions for adhesives, composite resins, and RMGICs, seems logical. However, despite phosphoric acid etching of the enamel surface, RMGICs still have a lower initial SBS than composite resins. Thus, clinicians have been reluctant to switch from using composite resin, an adhesive they have relied on since the beginning of their orthodontic education, to RMGICs. Historically, clinicians have tended to lay the blame for WSL development on poor patient compliance with oral hygiene instructions during orthodontic treatment and cited the risk of bracket failure rates as reason to defer the use of RMGICs, even though it is now known that RMGICs help minimize the development of WSLs. After review of the literature, the clinician should be more amenable to discontinue the use of composite resin in favor of RMGICs, as appropriate employment of RMGICs minimizes the risk of bracket bond failure while greatly reducing the risk of WSLs. Specifically, deproteinizing the enamel surface, etching of the enamel surface with phosphoric acid, and moistening of the enamel surface prior to bonding allow adequate SBS to minimize the risk of bracket bond failure to a clinically acceptable rate.
1.11 Increase in Bracket Shear Bond Strength with Deproteinization
Justus et al. [19] carried out an ex vivo study to determine whether deproteinization of the enamel surface, with 5.25 % sodium hypochlorite (NaOCl) before etching, increases bracket SBS of composite resin (Transbond XT) and RMGIC (Fuji Ortho LC). Brackets were bonded to 76 extracted premolars. Half were bonded with the composite resin and half with the RMGIC. Each group was randomized, with half receiving a 1-min application of NaOCl on the enamel surface. The findings indicated that by applying 5.25 % NaOCl for 1 min to the enamel surface with a microbrush (Figs. 1.7 and 1.8), followed by a 30-s acid etching with 37 % phosphoric acid and then moistening the enamel surface, the mean SBS of brackets bonded using Fuji Ortho LC was 9.64 ± 5.01 MPa, which exceeds the minimum mean tensile bond strength of 5.9 MPa, recommended by Reynolds [55]. In contrast, the Fuji Ortho LC group, in which NaOCl was not applied, had a much lower mean SBS (5.71 ± 3.87 MPa). Thus, if the clinician wishes to use RMGICs to prevent WSLs, it is recommended, based on the findings of this research, to deproteinize the enamel surface with 5.25 % NaOCl for 1 min before acid etching as there was a statistically significant difference between the 2 Fuji Ortho LC groups (control and experimental). The mean SBS increased 3.9 MPa (from 5.7 to 9.6 MPa) in the Fuji Ortho LC experimental group (with NaOCl) while increasing only 1.3 MPa (from 8.1 to 9.4 MPa) in the Transbond XT experimental group (with NaOCl).
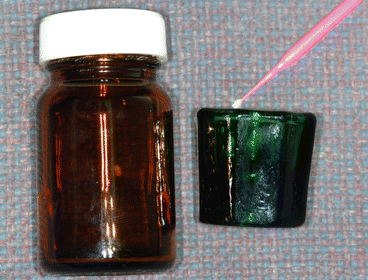
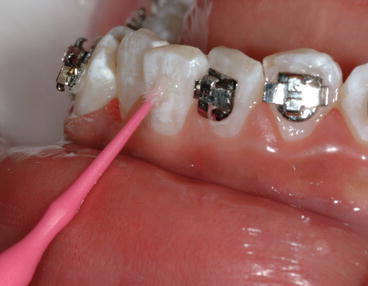
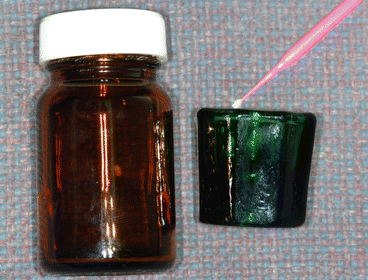
Fig. 1.7
Photograph on the left shows a glass container with a 5.25 % sodium hypochlorite (NaOCl) solution. This dark container helps prevent the deactivation of this solution by light. Photograph on the right shows a Dappen Dish containing the NaOCl solution and a microbrush used to transport it to the labial/buccal surfaces of the teeth

Fig. 1.8
Clinical example of enamel deproteinization by applying 5.25 % NaOCl solution to the enamel surface for 1 min with a microbrush. The objective is to eliminate the acquired pellicle so the 37 % acid etch can create improved etching patterns on the enamel surface to increase bracket SBS (Reprinted with permission from Justus et al. [19])
The authors believe that the small increase in the experimental Transbond XT group is because a primer is used in this system. The primer is an unfilled resin with a low viscosity. Even though the etching pattern is probably type 3 when no NaOCl is applied, the primer is able to penetrate deeply into the microporosities created during the acid-etching process on the enamel surface. This allows for the incorporation of resin “tags” into the enamel, thereby creating microscopic mechanical interlocks between the enamel and resin providing as a result adequate SBS, in spite of not deproteinizing the enamel surface. In contrast Fuji Ortho LC does not use a fluid primer.
Thus, if RMGICs are to be used, it is critical that the enamel etching pattern be of type 1 or 2 (to increase SBS), and not of type 3. The authors consider that the application of NaOCl to achieve a better etching pattern is important for RMGICs to be clinically useful. As the test specimens in the present study were stored in distilled water, the organic elements on the enamel surfaces might have been partially lost. Thus, the authors believe that the in vivo application of NaOCl might result in even greater SBS than demonstrated in this ex vivo study.
A modified adhesive remnant index (ARI) was used to quantify the amount of remaining adhesive. The scores for the Fuji Ortho LC group, in which the enamel surface was deproteinized, and the Transbond groups (with and without deproteinization) were similar. Bracket failure was seen more often at the bracket-adhesive interface, indicating a good bond between the adhesives and the enamel.
Stay updated, free dental videos. Join our Telegram channel

VIDEdental - Online dental courses


