Introduction
Class III malocclusion is characterized by a composite of dentoskeletal patterns that lead to the forward positioning of the mandibular teeth in relation to the maxillary teeth and a concave profile. Environmental and genetic factors are associated with this condition, which affects 1% of the population in the United States and imposes significant esthetic and functional burdens on affected persons. The purpose of this study was to capture the phenotypic variation in a large sample of white adults with Class III malocclusion using multivariate reduction methods.
Methods
Sixty-three lateral cephalometric variables were measured from the pretreatment records of 292 white subjects with Class II malocclusion (126 male, 166 female; ages, 16-57 years). Principal component analysis and cluster analysis were used to capture the phenotypic variation and identify the most homogeneous groups of subjects to reduce genetic heterogeneity.
Results
Principal component analysis resulted in 6 principal components that accounted for 81.2% of the variation. The first 3 components represented variation in mandibular horizontal and vertical positions, maxillary horizontal position, and mandibular incisor angulation. The cluster model identified 5 distinct subphenotypes of Class III malocclusion.
Conclusions
A spectrum of phenotypic definitions was obtained replicating results of previous studies and supporting the validity of these phenotypic measures in future research of the genetic and environmental etiologies of Class III malocclusion.
A disproportionate facial appearance often accompanies a severe Class III malocclusion and can result in a significant burden on the quality of life for those affected. Current therapies for this condition are aimed at treatment rather than prevention; thus, patients undergo years of orthodontic or orthopedic treatment, with many requiring surgical correction in adulthood. Studies since the 1970s have provided evidence that Class III skeletal characteristics have a strong genetic component. To elucidate preventive strategies and improve treatment modalities for these patients, studies identifying the genetic etiology of Class III malocclusion are warranted. However, detection of human susceptibility genes for Class III malocclusion is in its initial stages, since no etiologic mutations have yet been identified.
The few genetic mapping studies of Class III malocclusion thus far have found genetic linkages of mandibular prognathism to chromosome loci 1p22.1, 1p36, 3q26.2, 4p16, 6q25, 11q22, 12q13.13, 12q23, 14q 24.3, and 19p13.2 and have found positive association signals of mandibular height and prognathism to genes GHR , Matrilin-1 , EPB41 , TGFB3, LTBP , and MYO1H , indicating that molecular pathways implicated in bone ( TGFB3 , LTBP ) and cartilage ( GHR , Matrilin-1 ) development are plausible candidates for mandibular size discrepancies and should be considered in future research.
Although informative, the few genetic studies to date have limitations including modest sample sizes, exclusion of environmental effects, unknown generalization of results to other ancestries, and, finally and perhaps more importantly, limited phenotypes that cannot capture the complexities of Class III malocclusions. The success of genetic studies aimed at identifying causative genes for complex traits such as malocclusion depends greatly on a well-characterized phenotype to reduce heterogeneity. Studies of cross-sectional and retrospective longitudinal samples with conventional cephalometry or shape-analysis methods have attempted to characterize the dentoskeletal morphology in Class III children and adults of different ethnicities. In general, most of these studies have shown great variation in dentoskeletal morphology, yet the most common features in Class III subjects include a short anterior cranial base with an acute saddle angle, maxillary retrusion with a normal or protruded mandible, mandibular protrusion with a normal maxilla, and combinations of these anteroposterior discrepancies with a normal, excessive, or deficient vertical facial dimension along with protrusive maxillary incisors and retrusive mandibular incisors. Most of these components are present in the majority of Class III patients regardless of ethnic background, appear early in development, and tend to worsen with age.
Recently, studies using multivariable methods such as principal component analysis and cluster analysis applied to data from cephalometric radiographs have provided further insight into the characterization of Class III malocclusion phenotypes beyond traditional cephalometric methods. Principal component analysis essentially decomposes the correlations of a set of variables into orthogonal linear combinations of these variables (called components ). The information captured by the components decreases with the component order. Each component has scoring coefficients, or weights, for the included variables that allow for constructing a linear index that reflects a phenotypic axis of variation in the variables. In other words, principal component analysis accounts for the overall morphologic variation in the craniofacial complex. On the other hand, cluster analysis complements principal component analysis by identifying groups of subjects of similar phenotypes and allowing for traditional case-control comparisons.
Mackay et al studied morphologic variation in craniofacial forms using cluster analysis in 50 severe, nongrowing Class III patients requiring surgical correction and identified 5 subgroups. These findings provided good evidence that different forms of Class III malocclusion exist and can successfully be divided into groups based on similar phenotypes. Hong and Yi used cluster analysis to illustrate that different patterns of skeletal architecture—beyond the current simple classification based on the positions of the maxilla and the mandible, dentoalveolar units, and vertical relationships—contribute to the development of the Class III deformity. They identified 7 clusters in their Asian sample of 106 untreated Class III subjects with a mean age of 21 years (range, 16-32 years). Their clusters showed that in addition to the facial bones and dentition, the cranial base, cranial vault, and cervical spine were also involved in different but specific architectural patterns.
Abu Alhaija and Richardson studied 115 Class III children (aged 11.6-12.7 years) with cluster analysis and discriminant function analyses to differentiate favorable and unfavorable growers. They found 3 main clinical clusters according to long, short, and intermediate facial heights and determined that the power of discriminant function analysis to discriminate between favorable and unfavorable growers increases from 80% to 100% in some cases when cluster analysis is applied before discriminant function analysis.
The most recent article and the one most directly relevant to our study was by Bui et al in 2006; they characterized Class III malocclusion phenotypes using cluster analysis and principal component analysis of 67 cephalometric variables derived from 309 Class III subjects. Their sample included a wide age range (average, 19.1 years; range, 5.9-56.3 years) and was racially and ethnically diverse, consisting of 73% white subjects, 17% African Americans, 5% Asians, 3% Hispanics, and 2% another race or ethnicity. Subjects with previous orthodontic treatment, congenital abnormalities, trauma, or incomplete or low-quality cephalograms were excluded. Five clusters were identified, representing distinct subgroups of Class III malocclusion. In addition to the spectrum of phenotypic variation evidenced by the clusters, the investigators found that the first 5 principal components derived from the data explained 67% of the variation in the sample. Based on these combined findings, the authors suggested that different genes might be involved in controlling dimensions vs structures, and they questioned current treatment modalities that target the growth of the maxillary or mandibular skeletal structures. Although these data are informative, the sample included subjects who were still growing and did not have fully expressed phenotypes. In addition, the few ethnicities represented might not be large enough to be statistically meaningful, thus increasing phenotypic heterogeneity and limiting generalizability. Still, that study clearly demonstrated that Class III malocclusion exists in morphologically diverse patterns that can be classified into phenotypes using multivariable methods such as cluster analysis and principal component analysis.
Although previous studies have contributed to our understanding of the craniofacial components of the Class III malocclusion, there are limitations in sample sizes, sample selection criteria such as including growing subjects and not excluding other genetic or environmental traits such as missing or impacted teeth, heterogeneity due to race or ethnicity, and lack of or limited standardization of data with respect to key variables such as age and sex before applying the data reduction methods. Therefore, there is uncertainty regarding the extent to which the results from previous work, particularly from the most recent and methodologically advanced study of Bui et al, are generalizable to other samples and populations, and whether one can identify additional phenotypic variation in other samples.
In this study, we aimed at extracting phenotypes that could best capture the phenotypic variation in a large sample of white adults with Class III malocclusion using multivariate reduction methods. Using similar methods to those of Bui et al, one goal was to evaluate whether their phenotypes replicate in an ethnically homogeneous sample limited to postpubertal subjects. In light of the uncertainty about the generalizability of previous findings, replication studies are essential to evaluating the validity of this approach for phenotypic characterization. Another goal was to see whether we could explain meaningful additional variation in this sample. Such improvement in phenotypic variation can be important both clinically and for increasing the power of genetic studies. We applied rigorous sample inclusion criteria and carefully accounted for age and sex effects to increase the precision of the estimation. Our work builds on previous studies and provides a comprehensive set of Class III phenotypes that can be readily applied for phenotypic characterization of Class III subjects in other samples; this would facilitate large future collaborations of genetic studies.
Material and methods
The study protocol was reviewed and approved by the institutional review board at the University of Iowa. Our sample included Class III adults who were seeking treatment at the University of Iowa’s Orthodontic Graduate Clinic or Hospital Dentistry Clinic, or private practice clinics in the surrounding area. The sample consisted of 292 white postpubertal subjects (126 male >18, 166 female >16; age range, 16-57 years) who would have completed 98% of their growth at the time of initial records and met our eligibility criteria ( Table I ), which selected for moderate (ANB angle or Wits appraisal of 0-3 mm or degrees) to severe (ANB angle or Wits appraisal, <3 mm or degrees) Class III malocclusion with a skeletal component. The pool of available subjects included 311 patients; 18 who were not white were excluded because of lack of power, and 1 additional subject was found to be ineligible based on the inclusion criteria.
| Inclusion criteria | Exclusion criteria |
|---|---|
| Adult (female, ≥16 y; male, ≥18 y) | History of severe facial trauma |
| At least 2 of the following clinical criteria were required | Previous orthodontic treatment |
| ANB ≤0° | Facial syndrome |
| Overjet ≤0 mm | Missing or poor-quality records |
| At least edge-to-edge or anterior crossbite | Missing or impacted teeth other than third molars |
| Wits appraisal (female, ≤0 mm; male, ≤−1 mm) | Retained deciduous teeth |
| Angle Class III molar or canine relationship on at least 1 side | |
| Concave profile |
Two-dimensional pretreatment lateral cephalometric films of 292 Class III adults were digitized using Dolphin Imaging (version 11.0; Dolphin Imaging & Management Solutions, Chatsworth, Calif). Sixty-three cephalometric measurements were made representing distance, degree, percentage, and difference measures between the cephalometric landmarks, which were derived from commonly used lateral cephalometric analyses ( Table II ). The data were obtained from 2 sources (film and digital radiographs). All films taken on conventional or analog cephalometric units from either the College of Dentistry Graduate Orthodontic Clinic or the Hospital Dentistry Clinic were scanned into the Dolphin system with a 100-mm ruler and corrected for magnification by 12% and 13%, respectively. Distance measures for film radiographs were scaled (multiplied by 0.8929 for 12% magnified cephalometric radiographs from the College of Dentistry Graduate Clinic and 0.8850 for 13% magnified cephalometric radiographs from the Hospital Dentistry Clinic) to match the digital radiographs that were not corrected for magnification. To reduce landmark identification errors, all scanned analog films were traced twice (by K.C.V.), and the average value for each variable was used in the data analysis.
| Cranial base | Intermaxillary | Dental |
|---|---|---|
| Saddle/sella angle (SN-Ar) (°) | ANB (°) | U1-SN (°) |
| Anterior cranial base (SN) (mm) | Facial plane to AB (AB-NPg) (°) | U1-NA (°) |
| Posterior cranial base (S-Ar) (mm) | Facial plane to SN (SN-NPg) (°) | U1-NA (mm) |
| Midface length (Co-A) (mm) | U1-FH (°) | |
| Maxilla | Posteroanterior face height (S-Go/N-Me) (%) | IMPA (L1-MP) (°) |
| SNA (°) | Y-axis (N-S-Gn) (°) | L1-NB (°) |
| Convexity (NA-APg) (°) | Maxillomandibular difference (Co-Gn–Co-ANS) (mm) | L1-NB (mm) |
| N-A || HP (mm) | Wits appraisal (AO-BO) (mm) | L1 protrusion (L1-APg) (°) |
| A to N perp (FH) (mm) | Anterior face height (N-Me) (mm) | L1 protrusion (L1-APg) (mm) |
| Maxillary unit length (Co-ANS) (mm) | Upper face height (N-ANS) (mm) | FMIA (L1-FH) (°) |
| Lower face height (ANS-Me) (mm) | Interincisal angle (U1-L1) (°) | |
| Mandible | Nasal height (N-ANS/N-Me) (%) | UADH (U1-PP) (mm) |
| SNB (°) | PFH:AFH (Co-Go/N-Me) (%) | LADH (L1-MP) (mm) |
| Facial angle (FH-NPg) (°) | FMA (FH-MP) (°) | UPDH (U6-PP) (mm) |
| Gonial/jaw angle (Ar-Go-Me) (°) | SN-GoGn (°) | LPDH (L6-MP) (mm) |
| Chin angle (Id-Pg-MP) (°) | Occlusal plane to SN (°) | Overjet (mm) |
| Ramus height (Ar-Go) (mm) | Occlusal plane to FH (°) | Overbite (mm) |
| Length of mandibular base (Go-Pg) (mm) | FH-SN (°) | |
| Facial taper (N-Gn-Go) (°) | Soft tissue | |
| Articular angle (S-Ar-Go) (°) | Upper lip to E-plane (mm) | |
| N-B || HP (mm) | Lower lip to E-plane (mm) | |
| N-Pg || HP (mm) | Upper lip to ST N perp (FH) (mm) | |
| B to N perp (FH) (mm) | Lower lip to ST N perp (FH) (mm) | |
| Pg to N perp (FH) (mm) | ST Pg to ST N perp (FH) (mm) | |
| Mandibular unit length (Co-Gn) (mm) | ||
| Pg-NB (mm) | ||
| Posterior facial height (mm) (Co-Go) |
Reliability in landmark location and resulting calculation of craniofacial measurements was determined by interrater and intrarater methods with the intraclass correlation (ICC) and difference testing. A sample of 15 random cephalometric radiographs was traced by 2 raters (K.C.V. and L.M.M.) to assess interrater reliability and traced twice at least 3 weeks apart by the same rater (K.C.V.) to assess intrarater reliability. In addition, systematic differences between raters and between the first and second ratings were assessed with the Wilcoxon rank sum procedure. All analyses were performed using SAS software for Windows (version 9.3; SAS Institute, Cary, NC), and a type I error of 0.05 was assumed.
Statistical analysis
Principal component analysis and cluster analysis were used to capture the most significant components of variation and identify the most homogeneous groups of patients representing distinct Class III phenotypes to reduce the genetic heterogeneity. The data were standardized using a linear model to assess the possible effects of age and sex and to consider the possibility of age-by-sex interactions. A separate model was fitted for each of the 63 cephalometric measures using standard multiple regression methods. In all, 4 configurations of covariate adjustment were used among the 63 models: all included an adjustment for sex, some also required an age adjustment, and others needed an additional consideration of sex-by-age interaction: ie, a different age adjustment for each sex. Model diagnostic procedures were performed on all standardization models, and the assumptions were validated. The studentized (normalized) residuals were extracted from these models and used as the standardized data for the principal component analysis. Standardized principal component analysis scores were the basis for the formation of clusters defining different phenotypes of Class III malocclusion. Criterion-based model selection methods were used to determine the cluster configuration that illustrated the most distinct clusters graphically. Cluster analysis was performed via a partitional cluster analysis of extracted principal components using the SAS version 9.3 software with methods based on the leader and the k-means algorithms according to the method of Anderberg, called nearest centroid sorting .
To visualize the cluster analysis results, a canonical discriminant analysis was performed, and scored canonical variables were computed. The scored canonical variables were used to plot pairs or triads of canonical variables to aid in the visual interpretation of cluster differences. R statistical program along with the rgl package were used to produce 3-dimensional graphs of the data.
The k-means clustering algorithm is sensitive to extreme values as a consequence of the least squares condition; however, no subjects in this data set appeared to represent extreme observations. The clustering algorithm was performed separately for a range of numbers of clusters, from 3 to 7 clusters. The criterion-based methods of pseudo F statistic, approximate expected overall R 2 , and cubic clustering criterion (valid because of the uncorrelated nature of the principal components) as well as data visualization techniques of scored canonical variables were used to determine the appropriate number of clusters. Of the range of clusters considered, the 5-cluster model best optimized the criterion and presented the most distinct clusters graphically. Cluster validation was performed by locating subjects closest to the final cluster means and examining their cephalometric data and profile to ensure that the clusters represented distinct clinical phenotypes. All analyses were done with SAS version 9.3 software with a 0.05 level of significance.
Results
Reliability testing of landmark location and derived craniofacial measurements showed interrater reliability ICC values from 0.8594 to 0.9987, with only 4 variables for which the ICC was less than 90%. Intrarater reliability values ranged from 0.9021 to 0.9999, with only 2 variables less than 94%. In general, interrater and intrarater reliability values are deemed acceptable when they are above 85%. Thus, excellent agreement between the 2 measures was achieved for all 63 variables. Results from difference testing showed 16 significant differences between the 2 sets of measures for interrater reliability using the Wilcoxon signed rank test. The median difference was greater than 0.5 mm for only 7 of the 16 variables. When intrarater reliability was assessed, 5 significant differences were found between the 2 sets of measures. The median difference for midface length was 0.5 mm; the median difference for SNA angle was 0.1°. After examining variables with significant differences, outliers were identified, and techniques were used to improve the reliability to acceptable values. In general, discrepancies in cephalometric measurements of 0.5 to 1 mm are acceptable because of the inherent difficulties in landmark location.
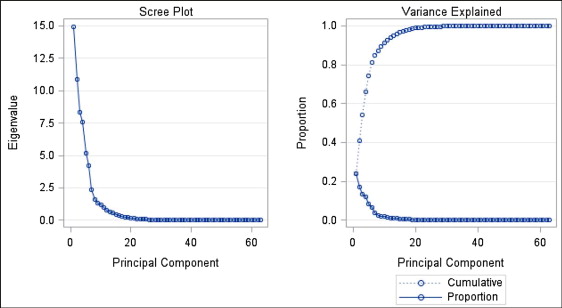
The results of the principal component analysis showed that 6 principal components accounted for 81.2% of the total variation in the data ( Fig 1 ). The first 6 principal components were selected because they explained the most variation in the data set and were specific in their anatomic explanations. As shown in Figure 1 , principal components beyond the sixth component were deemed not informative because the additional variation explained decreased significantly. About half of the variation in this sample was explained by the anteroposterior position of the mandible in relation to the cranial base, the size of the maxillomandibular horizontal discrepancy, and the mandibular incisor position and its effect on lower lip protrusion. Table III gives the variation explained by each of the 6 components and the set of cephalometric variables that contributed the most to each principal component. Figure 2 displays the cephalometric profiles of subjects with extreme principal components score values (ie, most negative and most positive scores) for each of the 6 principal components and the highest loading cephalometric variables in each principal component.
| Principal components | ||||||
|---|---|---|---|---|---|---|
| 1 | 2 | 3 | 4 | 5 | 6 | |
| Variance explained | 0.2374 | 0.1729 | 0.1325 | 0.1199 | 0.0825 | 0.0665 |
| Cumulative variance | 0.4103 | 0.5428 | 0.6627 | 0.7452 | 0.8117 | |
| Variables ∗ | Facial plane to SN (SN-NPg) (°) | Mandibular unit length (Co-Gn) (mm) | L1 protrusion (L1-APg) (mm) | IMPA (L1-MP) (°) | U1-NA (°) | FH-SN (°) |
| N-Pg || HP (mm) | Posterior facial height (mm) (Co-Go) | L1-NB (mm) | Maxillomandibular difference (Co-Gn-Co-ANS) (mm) | U1-NA (mm) | Saddle/sella angle (SN-Ar) (°) | |
| Y-axis (N-S-Gn) (°) | Midface length (Co-A) (mm) | Lower lip to ST N perp (FH) (mm) | Chin angle (Id-Pg-MP) (°) | A to N perp (FH) (mm) | Occlusal plane to FH (°) | |
| N-B || HP (mm) | Maxillary unit length (Co-ANS) (mm) | L1-NB (°) | Wits appraisal (AO-BO) (mm) | SNA (°) | Upper lip to ST N perp (FH) (mm) | |
| SNB (°) | Ramus height (Ar-Go) (mm) | Pg-NB (mm) | Facial taper (N-Gn-Go) (°) | N-A || HP (mm) | Lower lip to ST N perp (FH) (mm) | |
Stay updated, free dental videos. Join our Telegram channel

VIDEdental - Online dental courses


