Chapter 8 Obesity management
1 INTRODUCTION
Evaluation and management of obesity is an essential step in the treatment of patients with obstructive sleep apnea (OSA) and snoring based on three important facts. First, 66% of US adults are currently categorized as overweight or obese,1 placing obesity as one of the most common conditions seen among primary care providers and medical and surgical specialists. Second, obesity is a contributor to poor quality of life and increased morbidity and mortality that involves nine organ systems. Obesity, particularly upper body obesity, is a well-documented risk factor for OSA and is reported to be present in 60% to 90% of OSA patients evaluated in sleep clinics.2 Additionally, the severity of OSA is directly related to increasing body weight. Population-based studies have consistently shown an increased incidence of sleep disordered breathing with weight gain and obesity.3–5 Third, weight reduction by dietary and surgical treatment has been shown to improve co-morbid conditions including sleep disordered breathing and sleep quality.6 For these reasons, a thorough evaluation and treatment plan for OSA should specifically address weight loss for patients who are overweight or obese. This chapter will review current evaluation and treatment guidelines for overweight and obesity with a special focus for the otolaryngologist. For a more comprehensive review, readers are referred to the American Medical Association’s Assessment and Management of Adult Obesity: A Primer for Physicians.7
2 EVALUATION OF THE OVERWEIGHT AND OBESE PATIENT
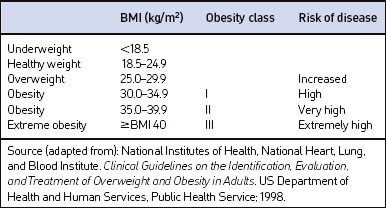
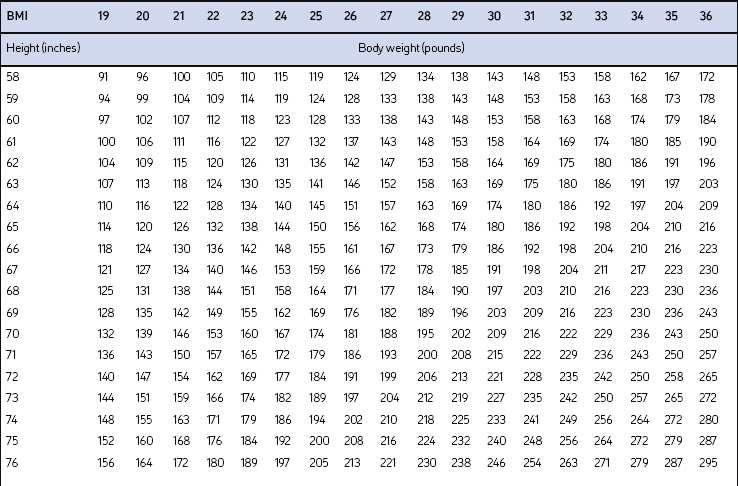
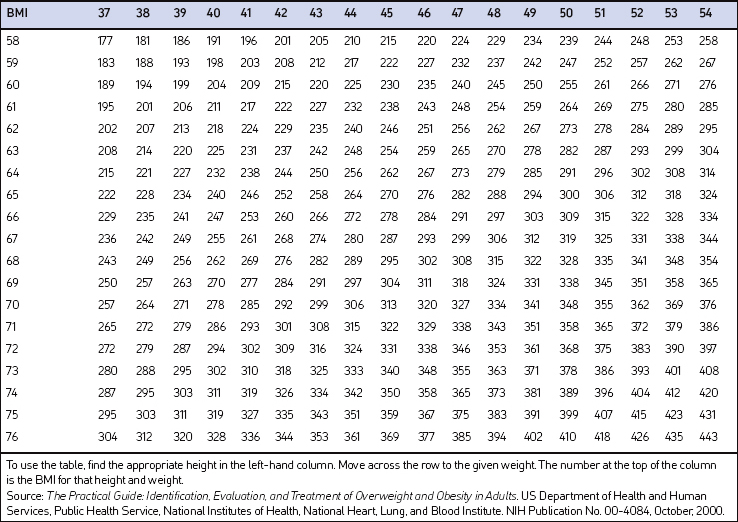
Screening for overweight and obesity should not rely on subjective visual inspection alone. Rather, screening is carried out by performing three simple anthropometric measurements: weight, height and waist circumference. These measurements can be easily obtained by ancillary office personnel. The Body Mass Index (BMI), calculated as weight (kg)/height (m)2, or as weight (pounds)/height (inches)2×703, is used to define classification of weight status and risk of disease (Table 8.1). A BMI table is more conveniently employed for simple reference (Table 8.2). BMI is used since it provides an estimate of body fat and is related to risk of disease.
Excess abdominal fat, assessed by measurement of waist circumference or waist-to-hip ratio, is independently associated with higher risk for metabolic abnormalities, diabetes mellitus, cardiovascular disease and OSA.8 Although protocols and guidelines differ among investigators, measurement of waist is most commonly performed in the horizontal plane above the iliac crest or at the narrowest point between the costal margin and iliac crest. Cut points that define higher risk are a waist circumference >102 cm (>40 inches) in men and >88 cm (>35 inches) in women when measured superior to the iliac crest.9 Overweight persons with waist circumferences exceeding these limits should be urged more strongly to pursue weight reduction since it categorically increases disease risk for each BMI class. Measurement of waist circumference should be obtained in those individuals with a BMI ≤35 kg/m2.
Although not specified in the clinical guidelines, it may be useful to measure neck circumference in obese patients presenting with signs and symptoms of OSA. Large neck girth in both men and women who snore is highly predictive of sleep apnea.10 Some studies suggest that neck circumference is a more useful predictor of sleep apnea than BMI11 or waist circumference.12 In general, a neck circumference of 17 inches in men or 16 inches in women indicates a higher risk for sleep apnea.
3 TAKING AN OBESITY-FOCUSED HISTORY
3.1 ASSESSING RISK
The medical history, physical examination and laboratory evaluation should be focused on assessing obesity co-morbid diseases. There is no single laboratory test or diagnostic evaluation that is indicated for all patients with obesity. The specific evaluation performed should be based on presentation of symptoms, risk factors and index of suspicion. However, based on several other screening guideline recommendations, all patients should have a fasting lipid panel (total, LDL and HDL cholesterol and triglyceride levels) and blood glucose measured at presentation along with blood pressure determination. Symptoms and diseases that are directly or indirectly related to obesity are listed in Table 8.3. Although individuals will vary, the number and severity of organ specific co-morbid conditions usually rise with increasing levels of obesity.
| Cardiovascular | Respiratory |
| Hypertension Congestive heart failure Cor pulmonale Varicose veins Pulmonary embolism Coronary artery disease |
Dyspnea Obstructive sleep apnea Hypoventilation syndrome Pickwickian syndrome Asthma |
| Endocrine | Gastrointestinal |
| Metabolic syndrome Type 2 diabetes Dyslipidemia Polycystic ovarian syndrome (PCOS)/angrogenicity Amenorrhea/infertility/menstrual disorders |
Gastroesophageal reflux disease (GERD) Non-alcoholic fatty liver disease (NAFLD) Cholelithiasis Hernias Colon cancer |
| Musculoskeletal | Genitourinary |
| Hyperuricemia and gout Immobility Osteoarthritis (knees and hips) Low back pain Carpal tunnel syndrome |
Urinary stress incontinence Obesity-related glomerulopathy End-stage renal disease Hypogonadism (male) Breast and uterine cancer Pregnancy complications |
| Psychological | Neurologic |
| Depression/low self-esteem Body image disturbance Social stigmatization |
Stroke Idiopathic intracranial hypertension Meralgia paresthetica Dementia |
| Integument | |
| Striae distensae (stretch marks) Stasis pigmentation of legs Lymphedema Cellulitis Intertrigo, carbuncles Acanthosis nigricans Acrochordon (skin tags) Hidradenitis suppurativa |
An increased waist circumference has been found to be predictive of a constellation of metabolic risk factors termed the metabolic syndrome that includes elevated blood pressure, impaired fasting glucose or glucose intolerance, hypertriglyceridemia, and low HDL cholesterol.6 Although the components and cutoff values selected to define the metabolic syndrome are useful for clinical practice, the constellation of abnormalities associated with insulin resistance is much broader. These ‘non-traditional risk factors’ include increased biomarkers of chronic inflammation (C-reactive protein, tumor necrosis factor-a, interleukin-6), a prothrombotic state (increased plasma plasminogen activator inhibitor (PAI)-1 and fibrinogen), endothelial dysfunction (decreased endothelium-dependent vasodilatation), hemodynamic changes (increased sympathetic nervous activity and renal sodium retention), hyper-uricemia, and non-alcoholic fatty liver disease (NAFLD). OSA often co-exists with these traditional and non-traditional cardiovascular risk factors, leading one author to coin the term ‘Syndrome Z’.13
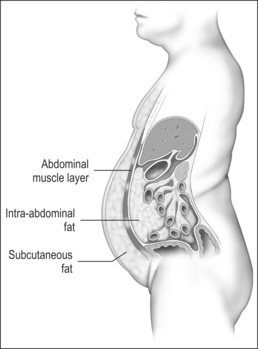
Measurement of waist circumference is a surrogate marker for visceral adipose tissue (VAT) which refers to adipose tissue located within the abdominal cavity, below abdominal muscles, and comprising omental and mesenteric adipose tissue, as well as adipose tissue of the retroperitoneal and perinephric regions (Fig. 8.1). Visceral fat accumulation has also been shown to have a significant negative impact on glycemic control in patients with type 2 diabetes. Recent studies have linked the metabolic and inflammatory abnormalities seen in abdominal obesity to the secretion of adipocyte and adipose connective tissue products called adipokines. Secreted factors include leptin, IL-6, TNF-α, angiotensinogen, PAI-1, transforming growth factor (TGF)-β, and adiponectin among many others.14 Secretion of these products results in altered endocrine, paracrine and autocrine functions. The metabolic dysregulation and systemic inflammation seen in OSA is likely related, in part, to VAT- derived secretory products.15–17
4 TREATMENT OF THE OVERWEIGHT AND OBESE PATIENT
Guidance for weight loss should be part of the non-surgical treatment for all obese patients with OSA.18 The evidence that treatment of obesity improves OSA is reasonably well established. Both medical and surgical approaches to weight loss have been associated with a consistent but variable reduction in number of respiratory events, as well as improvement in oxygenation.10 A critical review of the literature by Strobel and Rosen6 concluded that obesity treatments have shown varying degrees of improvement in sleep disordered breathing, oxygen hemogloblin saturation, sleep fragmentation, and daytime performance. Dietary weight losses of 9–18% body weight have been associated with reductions in AHI ranging from 30% to 75%. The authors note, however, that it is presently unclear how much weight loss is necessary to achieve significant improvements in sleep disordered breathing and which patients are most likely to benefit from weight loss. In a population-based study, a 10% weight loss was associated with a 26% decrease in the AHI.5 In general, surgical weight loss interventions have shown greater improvements in the AHI compared to dietary treatments. A recent systematic literature review of bariatric gastric banding surgery noted improvement in OSA in a number of studies.19 This was consistent with another bariatric surgical meta-analysis that showed striking results with over 85% of patients having complete resolution of the disorder.20 The authors also report data from a sub-analysis of four studies (92 subjects) that showed a mean decrease of 33.85 apneas or hypopneas per hour after bariatric surgery.
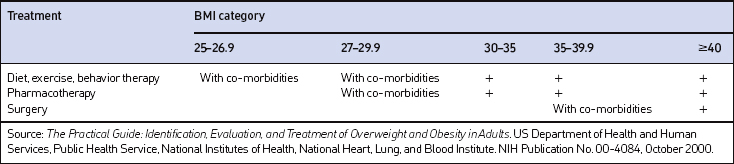
The decision of how aggressively to treat patients and which modalities to use is determined by the patient’s risk status, their abilities and desires, and by what resources are available. Table 8.4 provides a guide to selecting adjunctive treatments based on the BMI category. Equally important in choosing a treatment approach is the patient’s interest and ability to comply with the regimen and their perceptions of its effectiveness and safety. Therapy for obesity always includes lifestyle management and may include pharmacotherapy or surgery (Fig. 8.2). Since the otolaryngologist is unlikely to manage obesity by him or herself, it is essential to identify resources, e.g. registered dietitian, physician specialist, commercial or internet program, that can provide the needed assistance, monitoring and accountability for successful obesity care. The primary role of the otolaryngologist is to recognize the importance of weight loss, to incorporate weight loss into the treatment plan, and be supportive of the patient’s initiatives.
< div class='tao-gold-member'>
Stay updated, free dental videos. Join our Telegram channel

VIDEdental - Online dental courses