1. Microbial factors
(a) Antibiotic resistant mutant strains
MRSA
Vancomycin resistant enterococci
(b) Antibiotic resistant mechanisms
Exchange of genetic materials
Deficiency of specific porin channels
Promotion of active drug efflux
Thickening of the peptidoglycan layer of the outer wall
(c) Structure and organization of microbes
Variation in the outer wall in different classes of bacteria and fungi
Formation of biofilms
Protozoal existence as tropic feeding stage of resting cystic stage
2. Antibiotic misuse
Excessive or inappropriate prescription
Failure to complete treatment regimen
Widespread use of antibiotics in livestock feedstuff
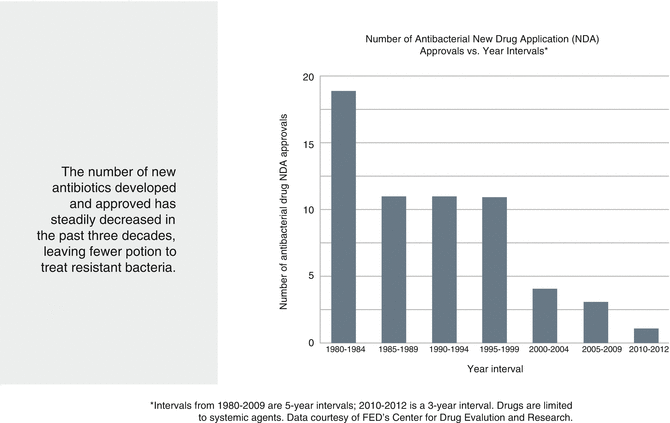
Fig. 6.1
Tomorrow’s antibiotics: the drug pipeline (Adapted from Prevention CfDCa [7]. With permission from CDC.org)
6.2 Antibacterial Nanoparticles
Nanoparticles (NPs) are microscopic particles with one or more dimensions in the range of 1–100 nm [10]. Nanotechnology, literally, is derived from the words “nano” and “technology” [11]. Nano is defined as one billionth of a quantity, represented mathematically as 10−9. NPs possess properties that are very unique from their bulk counterparts. A wide range of applications has been explored with NPs. Some of their applications are drug/gene delivery, fluorescent labeling for imaging, detection of pathogens, probing DNA structure, tissue engineering, tumor destruction, etc. [12]. NPs that display antibacterial properties may be termed antibacterial NPs. These NPs have been found to have a broad spectrum of antimicrobial activity and far lesser propensity to induce microbial resistance when compared to antibiotics.
Metallic nanoparticles of copper, gold, titanium, and zinc have attracted particular attention, with each of them having different physical properties and spectra of antimicrobial activity [13, 14]. It is known that magnesium oxide (MgO) and calcium oxide (CaO) slurries acted upon both gram-positive and gram-negative bacteria in a bactericidal manner [15], while zinc oxide (ZnO) slurry acted in a bacteriostatic manner and exhibited stronger antibacterial activity against gram-positive than gram-negative bacteria [15]. The antibacterial powders of magnesium oxide (MgO), calcium oxide (CaO), and ZnO generated active oxygen species, such as hydrogen peroxide and superoxide radical, which is responsible for their antibacterial effect. These NPs of metallic oxides with their high surface area and charge density exhibited greater interaction with bacteria and subsequently produced markedly high antibacterial efficacy [16]. Furthermore, it is apparent that bacteria are far less likely to acquire resistance against metallic nanoparticles than other conventional antibiotics.
Heavy metal ions are known to have different cytotoxic effects on bacterial cell functions [13, 17]. Copper ions may induce oxidative stresses [13] and affect the redox cycling, resulting in cell membrane and DNA damages. Zinc ions applied above the essential threshold level inhibit bacterial enzymes including dehydrogenase [18], which in turn impede the metabolic activity [13]. Silver ions inactivate proteins and inhibit the ability of DNA to replicate [14]. NPs synthesized from the powders of Ag, CuO, and ZnO are currently used for their antimicrobial activities. The electrostatic interaction between positively charged NPs and negatively charged bacterial cells and the accumulation of large numbers of NPs on the bacterial cell membrane have been associated with the loss of membrane permeability and cell death [17].
6.3 Bacterial Biofilms as the Therapeutic Target for Antimicrobials
Bacteria have been associated with almost 80 % of human infectious diseases [2, 19, 20]. Biofilm is the preferred mode for bacterial growth in oral disease conditions such as dental caries, periodontal disease, and apical periodontitis (endodontic disease) [21]. Bacteria persisting as biofilms have received major attention due to the difficulty in the clinical management of biofilm-mediated infections. Costerton defined biofilms as “a structured community of bacterial cells enclosed in a self-produced polymeric matrix and adherent to an inert or living surface” [22]. Typically, bacteria occupy 10–20 % of the total volume of the biofilm, and the remainder is the extracellular matrix [23, 24]. The extracellular matrix of a biofilm is polyanionic exopolysaccharides secreted by the bacteriumitself. They consist of polysaccharides, proteins, nucleic acids (extracellular DNA), and salts, totally making up to 95 % of the biofilm volume [24]. This hydrated envelope not only protects the bacteria from noxious threats but also acts as a scavenger to trap and concentrate nutrients for favorable growth [8]. These hydrated structures are known to possess water channels that facilitate efficient interchange of nutrients and waste between bacterial cells and bulk fluid. These channels also act as primitive circulatory system and transfer information between bacterial microcolonies in a biofilm.
The bacterial cells in a biofilm are heterogeneously arranged as microcolonies that are adherent to a solid surface (Fig. 6.2) [21]. These microcolonies are dense aggregates of bacteria assembled based on the physiological and metabolic states within the biofilms. Depending upon the environmental conditions and fluid shear forces, mature biofilms can detach microcolonies. This detachment of cells from matured biofilms termed seeding dispersal, could be either a continuous process such as erosion or massive loss of biofilm (sloughing). The microcolonies that are shed from the biofilm possess traits of the parent biofilm bacteria; consequently, they may lead to a distant infection [21, 25]. The antimicrobial resistance in a biofilm is a complex phenomenon, wherein one or more mechanisms go hand in hand. The mechanisms that contribute toward antimicrobial resistance in biofilm bacteria are categorized as
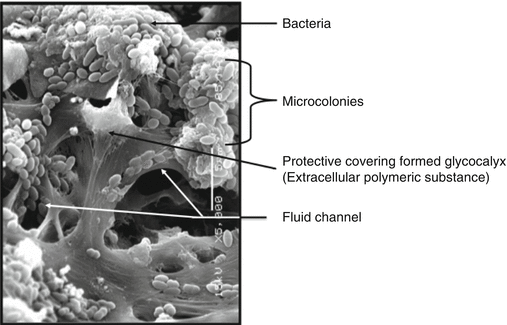

Fig. 6.2
SEM image showing ultrastructure of bacterial biofilm formed on dentin. The major components of biofilm structures are indicated (Courtesy of Anil Kishen)
1.
Protection by Exopolysaccharide (EPS) matrix: An EPS matrix is known to impart resistance to antimicrobials by acting as a physical barrier or neutralizing the chemicals applied. The diffusion of antimicrobials is limited to the surface of a biofilm structure enabling bacteria to survive deeper in the biofilms. The anionic EPS also acts as a chemical barrier. The charge and interwoven dense structure reduces the penetration of antimicrobials’ by ionic or electrostatic interactions [26, 27]. Mostly the antimicrobials used are positively charged hydrophilic compounds that can bind with EPS. Antimicrobial agents such as iodine, chlorine, and peroxygens have been shown to be neutralized by constituents of a biofilm matrix [28].
2.
Physiological state of bacteria: Persister cells are a small population of nongrowing cells within a biofilm that contribute to antibiotic tolerance [29]. These phenotypically different bacteria have very low metabolic activity, which allows them to enter a dormant state and survive with minimal nutrient requirement. As most antibiotics target actively growing cells, these persister cells are spared. However, once the antibiotic is stopped and a favorable condition is provided, these persisters are known to repopulate the biofilm, leading to treatment relapse [29].
3.
Altered microenvironment and phenotype: The mature biofilm consists of multilayered microcolonies of bacteria embedded in an EPS matrix. This creates a gradient of nutrients and redox potential that offers surface bacteria advantage of higher growth rates [30]. The deep-lying bacteria have limited access to nutrients and oxygen and the metabolic rates are reduced, thus allowing bacteria to resist antimicrobials. The enhanced tolerance to antibiotics has been suggested as these chemicals target bacterial cellular processes such as DNA replication or translation as in actively growing cells. The thicker the biofilms with an abundant EPS matrix, the more tenacious they become to be removed by antimicrobials. In addition, exposure to stress or low levels of antimicrobials is known to result in the expression of certain stress genes, shock proteins, and multidrug efflux pumps in biofilm bacteria [31]. The multidrug efflux pumps are membrane-bound active pumps found in both gram-positive and gram-negative bacteria and have been considered as one of the key factors in case of biofilm resistance [32, 33].
6.3.1 Endodontic Biofilms
Apical periodontitis (endodontic disease) is a bacterial biofilm–mediated infection [34]. In this disease process, the causative bacteria exist within the apical region of the root canals and release toxins/irritants, which induce periapical inflammation and host immune response [35, 36]. Till date, no specific correlation has been established between bacterial species in the root canal and clinical presentation of apical periodontitis [37]. In 1987, Nair and colleagues showed that bacteria in infected root canals exist as clusters of bacterial aggregates, a morphology analogous to biofilm bacteria [38]. Today, based on different histopathological studies, it is stressed that apical periodontitis is a biofilm-mediated disease [39–41].
A root canal system exhibits a tough environmental condition with regard to the availability of nutrients and oxygen [42]. Thus, an infected root canal contains a restricted assortment of bacterial species (approximately 5–10) [43, 44], which is markedly less when compared to more than 500 diverse species of bacteria in the oral cavity. No single microorganism is associated with the occurrence of disease rendering them with pathogenicity. The selection of particular combinations of bacteria could be attributed to the endodontic milieu that provides selective habitat, which supports the development of specific proportions of strict and facultative anaerobic flora [43, 45] (Fig. 6.3). Based on the “community-as-pathogen concept,” it is possible at this point to infer that some communities are more related to disease than others [46]. The main treatment goals of endodontic disease is thus to eliminate microbial biofilms from the infected root canals. Unfortunately, complete elimination of bacterial biofilms from the complex anatomy of root canals has been a daunting task for clinicians.
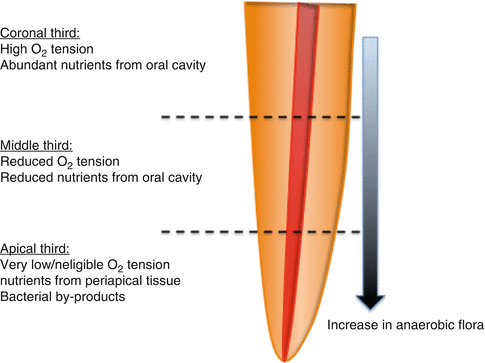

Fig. 6.3
The root canal space presents with selective pressures allowing specific bacteria to survive depending on their nutritional and environmental requirements (Adapted from Chavez de Paz [42]. With permission from Elsevier)
Previous studies showed that the presence of viable microbes at the time of filling was one of the crucial factors for the successful outcome of endodontic treatment [47, 48]. About 79 % of endodontically treated teeth with the presence of bacteria showed periapical lesions as compared to 28 % in those treated without bacteria [47]. Ricucci and Siqueira studied the prevalence of biofilms in primary and persistent infections and reported intraradicular biofilms in almost 74 % of the treated root canals with apical periodontitis at the apical segment [39]. Though extraradicular biofilms have been reported as a causative factor for persistent infections [49, 50], their prevalence has been reported to be on the lower side (6 %). As bacteria persisting within the treated root canals have been considered as the major cause for treatment failure [42, 51, 52], research has focused toward (a) understanding the limitations of the current root canal irrigation methods and (b) development of newer antibiofilm strategies for endodontic application.
6.4 Antimicrobials in Root Canal Therapy
Topical antimicrobials used as irrigants and medications are suggested for effective elimination of bacteria within the root canals [53, 54]. The success rate of root canal treatment is suggested to be 74–95 % [55–58]. The current level of evidence showed that despite the advancements in treatment strategies, the success rates have not increased for the past four to five decades [59]. This could be mainly attributed to the limitations of current technologies to deal with the challenges of the disease process [60]. Clinical outcome studies confirmed that the prognosis of endodontic treatment depend on the stage of disease process and whether the treatment is primary or retreatment [55–58, 61–66]. Follow-up study after 4–6 years of initial endodontic treatment revealed that the presence of periapical infection at the time of treatment reduced the healing rate from 92 to 74 % [61].
Endodontic therapy treats a complex tissue system consisting of the tooth–pulp–periradicular complex. Conventionally, chemical antimicrobials (topical) are used within the root canals in combination with mechanical instrumentation to achieve “microbe-free” root canals prior to filling the root canal with an inert filling material [2, 8, 67]. Ethylenediaminetetraacetic acid (EDTA) and sodium hypochlorite (NaOCl) are commonly used for root canal debridement. NaOCl is a deproteinating agent that results in the heterogeneous removal of organic substrate from dentin, whereas EDTA is a chelating agent that demineralizes the dentin and exposes the surface collagen fibrils [68]. These irrigants alone or in combinations are inadequate in achieving the above treatment goals. Therefore, newer antimicrobials are warranted for root canal disinfection. An effective treatment strategy that possesses marked antibiofilm capability (to eliminate residual endodontic bacteria/biofilm) without producing undue damaging effects on the dentin hard tissue and cytotoxic effect on periapical tissue will overcome the shortcomings associated with the current antimicrobial strategy in endodontic treatment.
Generally, therapeutic strategies for biofilms focus on (1) destroying or inactivating the resident bacteria within the biofilm structure or (2) disrupting the biofilm structure and simultaneously killing the resident microbes. Figure 6.4 shows the schematic representation of different antibiofilm approaches [67]. It includes the application of topical antimicrobials that produce slow destruction of the biofilm structure, antimicrobials that destroy persister cells or quorum-sensing signals in a biofilm, antimicrobial application in combination with strategies that enhance its diffusion into the biofilm structure, antimicrobials that diffuse into the biofilm structure producing biofilm bacterial killing and antimicrobial that destroys both the biofilm matrix and resident bacteria from a biofilm structure [69].
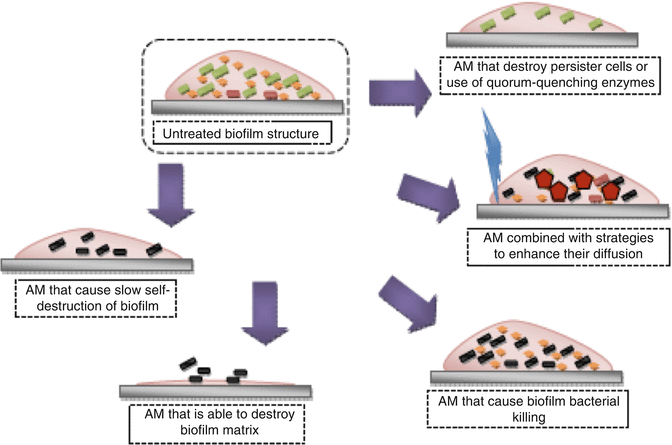

Fig. 6.4
Schematic diagram showing different antibiofilm strategies. AM antimicrobial (Adapted from Kishen [67]. With permission from John Wiley & Sons)
The requirements of an ideal root canal irrigant have been suggested to be (1) broad antimicrobial activity to eliminate biofilms of anaerobic and facultative anaerobic bacteria and yeasts; (2) dissolved necrotic pulp tissue; (3) ability to inactivate the endotoxins; (4) ability to remove smear layer following instrumentation and provide a clean root canal surface; and (5) biocompatibility to vital periapical tissues if extruded beyond the root canal space [70]. Till date, there is no antimicrobial irrigant that could perform all these functions single-handedly. Therefore, combinations of two or more irrigants are used to achieve a bacteria-free root canal space prior to filling [70]. Studies have shown that despite thorough instrumentation and irrigation procedures, bacteria still exist in significantly high numbers within the uninstrumented areas of root canals such as isthmuses and lateral canals [52, 54, 71, 72].
The current limitations in the endodontic disinfection strategies are not only due to the biofilm mode of growth within the root canals but also due to the anatomical complexities of root canal systems, dentin structure/composition, and factors associated with the chemical disinfectants collectively [67]. Microcomputed tomography–based studies revealed that 35–42 % of the root canal surfaces remain untouched by the instruments invariable to the instrumentation methods used [73]. Histological sections of the apical portions of the prepared root canals with instrumentation and irrigation procedures revealed the presence of bacteria in 86 % of cases mainly as biofilms in the complex anatomical sites [52]. In terms of bacterial reentry/recolonization, studies comparing the efficacy of different obturation techniques and materials have shown that microbial leakage occurs through the filled root canals [74]. Therefore, it may be concluded that even after intracanal medications, complete elimination of bacteria from the root canal system could not be achieved [48, 75]. Due to the shortcomings of the current antibiofilm strategies during root canal treatment, advanced disinfection strategies are being developed and tested. The newer disinfection strategies should aim to circumvent these challenges by eliminating biofilm bacteria not only from the main canals but also from the uninstrumented portions and anatomical complexities of the root canal system without inducing untoward effects on dentin substrate and periradicular tissue. In the following paragraphs, newer nanoparticles that hold significant potential for eliminating endodontic biofilms will be reviewed.
6.5 Classification and Types of Antibacterial Nanoparticles
Nanomaterials could be classified according to composition and various morphologies such as spheres, rods, tubes, and prisms. The nanoparticles based on different composition that are being developed for antibacterial purposes are shown in Table 6.2. Most of these newly developed nanoparticles are in the initial phases of laboratory testing. Table 6.3 shows the list of literature pertinent to the use of various nanoparticles used for improving the root canal disinfection.
Table 6.2
Nanoparticles available based on the composition
|
Nanoparticles based on composition
|
||||
|---|---|---|---|---|
|
Inorganic
|
Metallic
|
Polymeric
|
Quantum dots
|
Functionalized
|
|
Zinc oxide
|
Gold
|
Alginate
|
Cadmium sulfide
|
With:
|
|
Iron oxide
|
Silver
|
Chitosan
|
Cadmium selenide
|
Drugs
|
|
Titanium dioxide
|
Iron
|
Photosensitizers
|
||
|
Cerium oxide
|
Copper
|
Antibodies
|
||
|
Aluminum oxide
|
Magnesium
|
Proteins
|
||
Table 6.3
List of literature available on the use of nanoparticles for antibacterial purpose to achieve root canal disinfection
|
Author
|
Nanoparticles used
|
Bacteria tested
|
Observations
|
|---|---|---|---|
|
Waltimo et al. (2007)
|
Bioactive glass
45S5
|
E. faecalis in planktonic
|
Micron to nano size increased the killing
|
|
Kishen et al. (2008)
|
ZnO
Chitosan
|
E. faecalis in planktonic
|
CS -highest antibacterial
Combination-highest leaching property
|
|
Waltimo et al. (2009)
|
Bioactive glass- Nano/Micron combination
|
3 weeks old E. faecalis biofilm in root canals
|
Nano BAG did not show any antibacterial efficacy
|
|
Mortazavi et al. (2010)
|
BAG of different sizes
|
Planktonic E. coli, P. aeruginosa, S. typhi, and S. aureus
|
Antibacterial activity decreased with decrease in size
|
|
Pagonis et al. (2010)
|
MB loaded PLGA
|
3 days old E. faecalis biofilm in root canals
|
Significant reduction of biofilm
|
|
Chogle et al. (2011)
|
C18 organoclay
|
E. faecalis in planktonic
|
Reduced apical microleakage when mixed with polymer
|
|
Shrestha et al. (2010)
|
ZnO
Chitosan
|
E. faecalis in planktonic and biofilms
|
Total elimination of planktonic
Significant reduction of biofilms
Retained antibacterial property after aging
|
|
Shrestha and Kishen (2012)
|
Chitosan in the presence of tissue inhibitors
|
E. faecalis in planktonic
|
Pulp and BSA significantly inhibited the antibacterial effect
|
|
DaSilva et al. (2013)
|
Sealer incorporated with chitosan nanoparticles with/without canal surface treatment with different formulations of CS
|
E. faecalis 7 days in a chemostat-based biofilm fermentor
|
Inhibition of biofilm formation within the sealer-dentin interface
Surface treatment with chitosan showed higher inhibition of biofilm formation
|
|
Abramovitz et al. (2013)
|
Quaternary ammonium polyethyleneimine nanoparticles (QPEI NP) incorporated into standard TRMs
|
S. mutans and E. faecalis in planktonic form
|
Incorporation of 2 % wt/wt QPEI NP significantly increased the sealing ability of TRMs
|
|
Shrestha et al. (2013)
|
Rose bengal functionalized chitosan nanoparticles (CSRBnp)
|
E. faecalis in planktonic and biofilms
|
Photoactivation of CSRBnp resulted in reduced viability of biofilms and disruption of biofilm structure.
|
|
Shrestha (Kishen*) et al. (2013)
|
Rose bengal functionalized chitosan nanoparticles (CSRBnp) in the presence of tissue inhibitors
|
E. faecalis in planktonic form
|
Pulp and BSA significantly inhibited the antibacterial effect
|
|
Shrestha (Kishen*) et al. (2014)
|
Photoactivated rose bengal functionalized chitosan nanoparticles (CSRBnp)
|
E. faecalis in planktonic and 21 days biofilms
|
CSRBnp displayed properties of both CS and RB in a photoactivable nano-structure that performed the dual function of targeted elimination of bacterial-biofilms
|
|
Shrestha and Kishen (2014)
|
Rose bengal functionalized chitosan nanoparticles (CSRBnp)
|
Multispecies biofilms (Streptococcus oralis, Prevotella intermedia, and Actinomyces naeslundii) grown on dentin sections for 21 days
|
CSRBnp showed complete disruption of multispecies biofilm with a reduction in viable bacteria and biofilm thickness
|
6.5.1 Chitosan Nanoparticles
Chitosan (poly (1, 4), β-d glucopyranosamine), a derivative of chitin, the second most abundant natural biopolymer, has received significant interest in biomedicine [76–78]. The industrial extraction of chitin is generally obtained from crustaceans such as crabs, lobsters, and shrimps, making up to 1013 kg in the biosphere [79]. The structure of chitin closely resembles that of cellulose, and both act as a structural support and defense material in living organisms. Chitin has two reactive groups: primary (C-6) and secondary (C-3) hydroxyl groups allowing for various chemical modifications. Chitosan has an additional amino (C-2) group on each deacetylated unit.
Chitosan is known as a versatile biopolymer that could be synthesized in various forms such as powder (micro- and nanoparticles), capsules, films, scaffolds, hydrogels, beads, and bandages [77]. Chitosan has a structure similar to extracellular matrix components and hence is used to reinforce the collagen constructs [80]. This hydrophilic polymer with large numbers of hydroxyl and free amino groups can be subjected to numerous chemical modifications and grafting [81–83]. Nanoparticles of chitosan have been developed mainly for drug/gene delivery applications.
Nanoparticles of chitosan could be synthesized or assembled using different methods depending on the end application or the physical characteristics required in the nanoparticles [77]. Chitosan nanoparticles (CS NPs), by virtue of their charge and size, are expected to possess enhanced antibacterial activity. In addition, chitosan possesses several characteristics such as being nontoxic toward mammalian cells, color compatibility to tooth structure, cost effectiveness, availability, and ease of chemical modification. CS NPs can be delivered within the anatomical complexities and dentinal tubules of an infected root canal to enhance root canal disinfection [84].
Antibacterial Properties
Chitosan and its derivatives such as carboxymethylated chitosan showed a broad range of antimicrobial activity, biocompatibility, and biodegradability [85–88]. The exact mechanisms of antibacterial action of chitosan and its derivatives are still not vivid. However, a more commonly proposed mechanism is contact-mediated killing that involves the electrostatic attraction of positively charged chitosan with the negatively charged bacterial cell membranes (Fig. 6.5). This might lead to the altered cell wall permeability, eventually resulting in rupture of cells and leakage of the proteinaceous and other intracellular components [89, 90]. Rabea et al. proposed that chitosan, due to its chelating property, sequesters trace metals/essential nutrients and inhibits enzyme activities essential for bacterial cell survival [89]. Under transmission electron microscopy, the bacterial cells were seen to be completely enveloped in the chitosan forming an impermeable layer [86]. This could have resulted in the prevention of transport of essential solutes leading to cell death. In case of fungi, chitosan was hypothesized to enter cells and reach nuclei, bind with DNA, and inhibit RNA and protein synthesis.
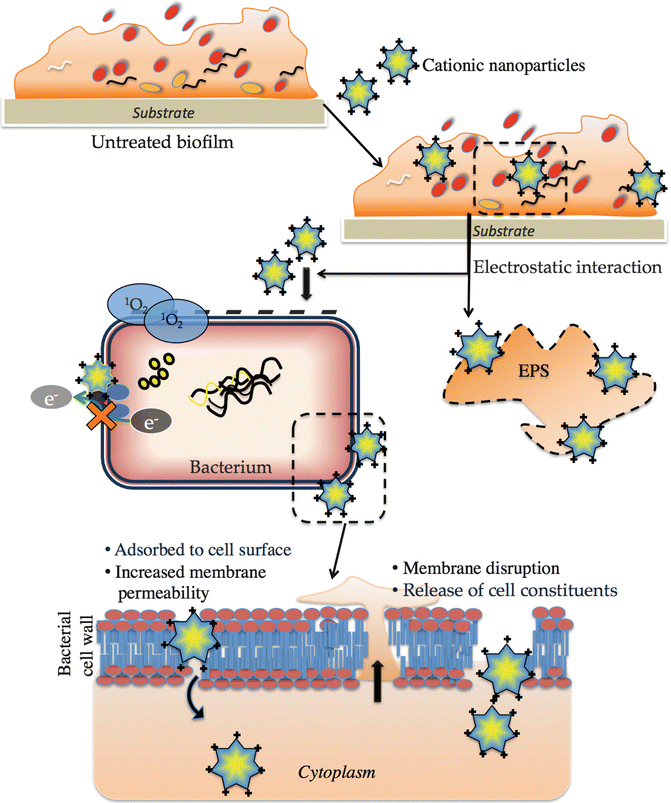

Fig. 6.5
A schematic diagram illustrating the antibacterial mechanism of nanoparticles with positive charge (e.g. Chitosan). Mature bacterial biofilm consisting of abundant EPS and bacteria enclosed. When cationic nanoparticles are introduced for treatment of biofilms, it can interact with both EPS and bacterial cells. The initial electrostatic interaction between positively charged nanoparticles and negatively charged bacterial surface. Bacterial killing occurs upon contact mediated lipid peroxidation via production of reactive oxygen species (ROS). The membrane damage and increased permeability of unstable membrane eventually leads to ingress of nanoparticles into the cytoplasm and release of cytoplasmic constituents. EPS secreted by bacteria in biofilm may interact with the nanoparticles and prevent from interacting with bacteria and thus reducing the antibacterial efficacy
Chitosan has excellent antibacterial, antiviral, and antifungal properties [89]. In case of bacteria, gram-positive bacteria were more susceptible than gram-negative ones. The minimal inhibitory concentrations ranged from 18 to 5,000 ppm depending upon the organism, pH, degree of deacetylation (DD), molecular weight, chemical modifications, and presence of lipids and proteins [89, 91]. The DD is known to influence the antibacterial activity. With higher DD, the number of amine groups increases per glucosamine unit, and thus, chitosan showed higher antibacterial efficacy [92]. Another study tested the efficacy of CS NPs and ZnO NPs in disinfecting and disrupting biofilm bacteria and the long-term efficacy of these nanoparticulates following aging. Enterococcus faecalis (ATCC & OG1RF) in planktonic and biofilm forms were tested in this study. These studies demonstrated that the rate of bacterial killing by NPs depended on the concentration and duration of interaction. Total elimination of planktonic bacteria was observed in contrast to the biofilm bacteria, which survived even after 72 h (Fig. 6.6). Both CS NPs and ZnO NPs were found to retain their antibacterial properties after aging for 90 days [85].
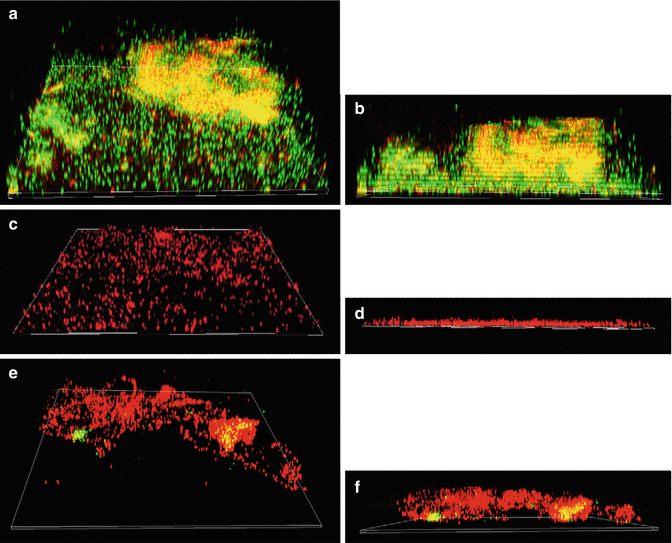

Fig. 6.6
The 3-dimensional CLSM reconstruction of E. faecalis (ATCC 29212) biofilm (a, b) and after treatment with antibacterial ZnO-np (c, d) and CS-np (e, f). The number of live bacterial cells was reduced significantly, and the 3-dimensional structure was also disrupted. (b, d, and f show the sagittal sections of the biofilm structure) (original magnification) (Adapted from Shrestha et al. [85]. With permission from John Wiley & Sons)
6.5.2 Bioactive Glass Nanoparticles
Bioactive glass (BAG) received considerable interest in root canal disinfection due to its antibacterial properties. BAG consists of SiO2, Na2O, CaO2, and P2O5 at different concentrations. Nanometric bioactive glass (BAG) used by Zehnder et al. [93] was amorphous in nature, ranging from 20 to 60 nm in size. They highlighted that the increase in pH is mainly responsible for the antimicrobial activity. Furthermore, the release of Ca2+, Na+, PO43−, and Si4+ could lead to formation of bonds with the mineralized hard tissues.
Antibacterial Properties
The antibacterial mechanism of BAG has been attributed to several factors acting together [94]:
1.
High pH: increase in pH due to release of ions in an aqueous environment.
2.
Osmotic effects: increase in osmotic pressure above 1 % is inhibitory for many bacteria.
3.
Ca/P precipitation: induced mineralization on the bacterial surface.
BAG was used for in vitro root canal disinfection studies [93, 95, 96]. When compared with calcium hydroxide, the latter showed significantly more antibacterial effect than BAG in preventing residual bacterial growth [93]. Waltimo et al. [97] suggested that an ideal preparation of 45S5 bioactive glass suspensions/slurries for root canal disinfection should combine high pH induction with capacity for continuing release of alkaline species. They demonstrated that BAG nanometric slurry had a 12-fold higher specific surface area than the micrometric counterpart. However, the latter had a considerably higher alkaline capacity and disinfected significantly better. The nano-BAG was found to be less effective in eliminating biofilms [97] as compared to the planktonic counterparts [98, 99]. The contradictory result in the antibacterial efficacy of BAG with transition from micron- to nano-sized has been contributed to the tenfold increase in silica release and solution pH elevation by more than three units [98].
6.5.3 Silver Nanoparticles
Use of silver compounds and nanoparticles is widespread mainly owing to its antibacterial property. As early as the 1800s, silver has been used for the prevention of infection in burn cases [100]. Prior to the introduction of antibiotics in the early twentieth century, oral intake of aqueous colloidal dispersions of silver for the prevention of infection has been reported [101]. In dentistry, silver and its nanoparticles have been tested for application as dental restorative material, endodontic retrofill material, dental implants, and caries inhibitory solution [102].
Antibacterial Properties
The antibacterial property of silver is mainly due to its interaction with the sulfhydryl groups of proteins and DNA, altering the hydrogen bonding/respiratory chain, unwinding of DNA, and interference with cell wall synthesis/cell division [103, 104]. At the macroscopic level, these silver nanoparticles are known to destabilize the bacterial membrane and increase permeability leading to leakage of cell constituents [105]. However, the toxicity of silver ions has been a major limitation for its widespread use. Research has been directed toward developing silver nanoparticles with specific antibacterial activity and lower cytotoxicity to host cells [106].
Silver in its metallic state is inert but in the presence of moisture is ionized and forms silver ions. These silver ions are highly reactive, bind to tissue proteins, and induce tissue changes. These silver ions bring about structural changes in the bacterial cell wall and nuclear membrane, disrupt the membrane barrier, and result in cell death [107
Stay updated, free dental videos. Join our Telegram channel

VIDEdental - Online dental courses


