Fig. 2.1
Number of (a) publications and (b) citations related to “nanomaterials” or “nanotechnology” or “nanoparticles” and tissue engineering/dental according to ISI Web of Science (Data obtained March 2014). A steady increase in the number of publication in the area of nanomaterials for dental research indicates growing interest in the field
This chapter reviews recent developments in the area of nanomaterials and nanotechnologies for dental restoration. We will focus on two major types of nanomaterials, namely, bioinert and bioactive nanomaterials. Bioactive nanomaterials include hydroxyapatite, tricalcium phosphate, and bioglass nanomaterials, whereas bioinert nanomaterials include alumina, zirconia, titanium, and vitreous carbon. Emerging trends in the area of dental nanomaterials and future prospects will also be discussed.
2.2 Anatomy and Development of the Tooth
To design advanced biomaterials for dental repair, it is important to understand the chemical and physical properties of native tissues. Teeth are three-dimensional complex structures consisting of the crown, neck, and root. Oral ectodermal cells and neural crest–derived mesenchyme are the primary precursors for mammalian teeth. Tooth development can broadly be divided into five major stages: dental lamina, initiation, bud stage, cap stage, bell stage and eruption (Fig. 2.2). Initially, ameloblasts and odontoblasts differentiate at the junction between epithelium and mesenchyme to form enamel and dentin, which are the tooth-specific hard tissues. Following this, root formation is initiated by differentiation of cementoblasts from dental follicle mesenchyme to form cementum, which is the third hard tissue of the tooth. As teeth erupt into the oral cavity and the roots reach their final length, substantial amounts of epithelial cells are lost [21].
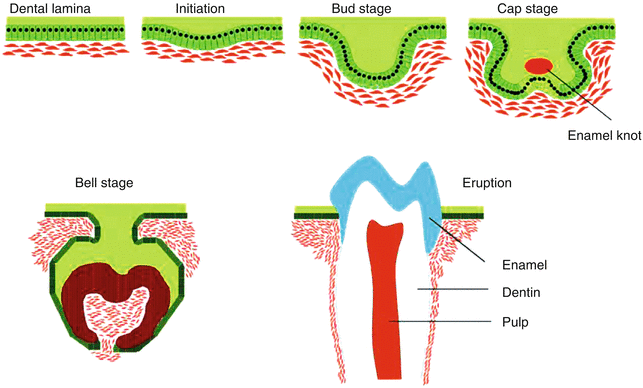

Fig. 2.2
Stages of tooth morphogenesis (Adapted from Nakashima and Reddi [97]. With permission from Nature Publishing Group)
A range of soft and hard dental tissues is observed, depending on the anatomical location. For example, the tooth crown with a mechanical stiffness of 100 GPa is composed of the mineralized outer enamel layer with 0.6 % of organic matter and 0.36 % of proteins. Underlying the enamel layer, the mineralized dentin layer has moderate mechanical stiffness (80 GPa), and the inner pulp dental tissue is very soft (~65 GPa) [22]. The enamel layer is mechanically stiff as it is composed of mineralized tissue that is produced by specific epithelial cells called ameloblasts. This layer consists of specialized enamel proteins such as enamelin, amelogenin, and ameloblastin. These proteins participate in helping structural organization and biomineralization of the enamel surface [16]. The underlying dentin layer is 75 % mineralized tissue containing dental-derived mesenchymal cells called odontoblasts on the dental papilla [23, 24], while the inner dental pulp tissue present in the root canal consists of dentin, cementum, and periodontal ligament layers, which secures the tooth to the alveolar bone [4].
Most of the mineralized structure observed in dental tissue is composed of nonstoichiometric carbonated apatite known as nanocrystalline hydroxyapatite (HA) in a highly organized fashion from micrometer to nanometer-length scale. The nanocrystalline HA particles are rod-shaped particles 10–60 nm in length and 2–6 nm in diameter [25]. The second most important component of dental tissue is cells. Cells present in dental tissue include osteoclasts and osteoblasts present on the face of the alveolar bone, cementoblasts on the surface of the root canal, and mesenchymal tissue in the ligament tissue that is essential for the long-term survival of these dental tissues. Molecular signals initiated by these cells trigger a set of events that regulate tissue morphogenesis, regeneration, and differentiation.
Human teeth do not have the capacity to regenerate after eruption. Therefore, biomedical engineering of human teeth using nondental cells might be a potential alternative for functional dental restorations. Here, we will discuss various types of nanomaterials that are proposed to facilitate regeneration of dental tissues.
2.3 Nanomaterials for Dental Repair
2.3.1 Hydroxyapatite as a Biomaterial for Dental Restoration
Hydroxyapatite particle (HAp) is a naturally occurring mineral form of calcium apatite, which is predominately obtained in mineralized tissue [26–28]. Hydroxyapatite is also one of the major components of dentin. Due to its bone-bonding ability, hydroxyapatite has been widely used as a coating material for various dental implants and grafts. Additionally, HAp is highly biocompatible and can rapidly osteointegrate with bone tissue. Due to these advantages, HAp is used in various forms, such as powders [29], coatings [30], and composites [26, 31] for dental restoration. Despite various advantages, hydroxyapatite has poor mechanical properties (highly brittle) and hence cannot be used for load-bearing applications [28].
A range of techniques have been developed to improve the mechanical toughness of this HAp [32]. Such hybrid nanocomposites are used to design bioactive coatings on dental implants. Brostow et al. designed a porous hydroxyapatite (150 μm)–based material by selecting a polyurethane to fabricate porous material and improve the mechanical properties of the implants [32]. Polyurethane was used because of its tunable mechanical stiffness. These hybrid nanocomposites are shown to have enhanced mechanical strength along with an interconnected porous network. It was shown that a rigid polymer with 40 % alumina contained fewer smaller closed pores, which causes an increase in Young’s modulus. Earlier studies have highlighted the use of porous materials for dental fillings [33–35]. The increase in mechanical properties is mainly used to enhance surface interactions between nanoparticles and polymers. In a similar study, Uezono et al. showed that nanocomposites have significantly higher bioactivity when compared to microcomposites, as determined by an enhanced bone-bonding ability [36]. They fabricated titanium (Ti) rod specimens with a machined surface with nHAp coating and a nanohydroxyapatite/collagen (nHAp/Col) coating and placed it under the periosteum of a rat calvariu. After 4 weeks of implantation, they observed that nHAp/Col-coated titanium rods were completely surrounded by new bone tissue.
As compared to conventional microsized hydroxyapatite, nanophase hydroxyapatite displays unique properties such as an increased surface area, a lower contact angle, an altered electronic structure, and an increased number of atoms on the surface. As a consequence, the addition of hydroxyapatite nanoparticles to a polymer matrix result in enhanced mechanical strength. For example, Liu et al. showed that the addition of nHAp to chitosan scaffolds enhances the proliferation of bone marrow stem cells and an upregulation of mRNA for Smad1, BMP-2/4, Runx2, ALP, collagen I, integrin subunits, together with myosins compared to the addition of micron HAp [37]. The addition of nHAp significantly enhances pSmad1/5/8 in BMP pathways and showed nuclear localization along with enhanced osteocalcin production. In a similar study, nHAps are used to reinforce polymeric networks and enhance bioactive characteristics [38]. The addition of nHAps to a PEG matrix results in a significant increase in mechanical strength due to physical interactions between polymers and nanoparticles (Fig. 2.3). Additionally, this cell adhesion and spreading was also enhanced due to the nHAp addition. These bioactive nanomaterials can be used as an injectable matrix for periodontal regeneration and bone regrowth. Overall, nHAp-reinforced nanocomposites or surface coating improves mechanical stiffness and bioactivity of implants and can be used for dental restoration.
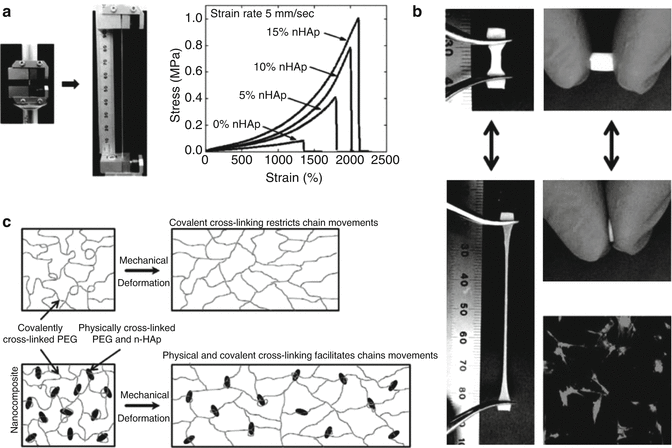

Fig. 2.3
Nanocomposite reinforced with nHAP. (a) Addition of nHAP to polymer matrix have shown to increase the mechanical strength by 5 folds. (b) The nHAP reinforced nanocomposite showed elastomeric properties indicating strong nHAP-polymer interactions at nano-length scale (c). The addition of nHAP also shown to promote cell adhesion properties of nanocomposite network (Reprinted from Gaharwar et al. [38]. With permission from American Chemical Society)
2.3.2 Dental Regeneration Using Bioactive Glass
Bioactive glass was developed by Hench et al. in 1960 with a primary composition of silicon dioxide (SiO2), sodium oxide (Na2O), calcium oxide (CaO), and phosphorous pentoxide (P2O5) in specific proportions. Bioglass is extensively used in the field of dental repair due to its bone-bonding ability [39–42]. When the bioglass is subjected to an aqueous environment, it results in the formation of hydroxycarbonate apatite/hydroxyapatite layers on the surface [43, 44]. Despite these advantages, bioglass is brittle and has a low wear resistance and thus cannot be used for load-bearing applications.
A range of techniques was developed to improve the mechanical properties of bioglasses. For example, Ananth et al. reinforced bioglass with yttria-stabilized zirconia. This yttria-stabilized zirconia bioglass is deposited on the titanium implant (Ti6Al4V) using electrophoretic deposition [45]. The yttria-stabilized zirconia bioglass (1YSZ-2BG) coating showed significantly higher bonding strength (72 ± 2 MPa) compared to yttrium-stabilized zirconia alone (35 ± 2 MPa). A biocompatibility test was performed to check for the ability to form an apatite layer on 1YSZ-2BG. A thin calcium phosphate film was observed on part of the 1 YSZ-2BG-coated surface after 7 days of immersion in simulated body fluid (SBF) without any precipitation. The apatite layer formation and size of calcium phosphate globules increased with an increase in the immersion time. The favored apatite formation on 1YSZ-2BG could be due to the Si-OH group arrangement on the bioglass (Fig. 2.4). Moreover, osteoblasts seeded on yttria-stabilized zirconia bioglass surfaces exhibited flattened morphology with numerous filopodial extensions. After 21 days of culture, the cells spread readily the on yttria-stabilized zirconia bioglass surface and resulted in the production of mineralized nodules. The enhanced mineralization of these bioactive surfaces is mainly attributed to the release of ions from the bioglass that facilitated the mineralization process.
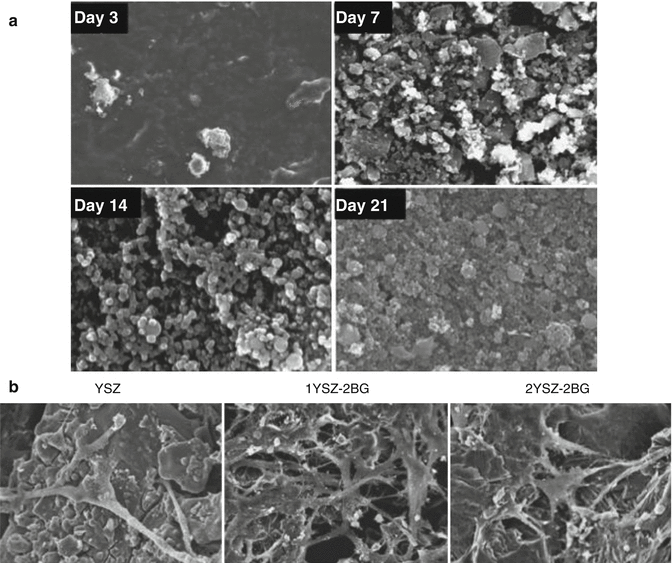

Fig. 2.4
Bioglass reinforced yttria-stabilized composite layer deposited on Ti6Al4V substrates. (a) SEM surface images of Ti6Al4V coated with 1YSZ–2BG immersed in simulated body fluid (SFB) for day 3, 7, 14, and 21. The formation CaP onto the surface of the 1YSZ–2BG indicates the catalytic effect of Si–OH, Zr–OH and Ti–OH groups on the apatite nucleation. (b) Morphological of osteoblast cells cultured on the Ti6Al4V surface coated with YSZ, YSZ–BG and 2YSZ-2BG (Adapted from Ananth et al. [45]. With permission from Elsevier)
Bioactive glass releases several ions that include sodium, phosphate, calcium, and silicon. Silicon in particular plays a central role as a bioactive agent. When released, it forms silanols in the near-liquid region above the glass surface. These silanols spontaneously polymerize to form a silica gel layer for the eventual nucleation and growth of a “bone-like” apatite. These same silanols also appear to influence collagen matrix synthesis by various cells types. In previous work, it was found that the ionic products from bioactive glass dissolution enhances the formation of a dense, elongated collagen fiber matrix, which was attributed to the presence of ionic Si in vitro [46, 47]. Moreover, it was found that these ions combinatorially effected the expression of genes associated with osteogenesis. The combinatorial concept of gene expression involves the use of gene regulatory proteins, which can individually control the expression of several genes, while combining these gene regulatory proteins can control the expression of single genes. Analogously, it was shown that Si and Ca ions can combinatorially regulate the expression of osteocalcin and this combinatorial effect can also enhance the mineralized tissue formation [48, 49]. Therefore, these ions can be used as an inductive agent to enhance the formation of mineralized tissue through the combinatorial of osteogenic gene expression.
2.3.3 Nanotopography Improves Biointegration of Titanium Implants
Titanium is one of the inert metals that is extensively used in the field of dental and musculoskeletal tissue engineering for load-bearing applications due to its excellent biocompatibility, high toughness, and excellent corrosion resistance and osteointegration [50–53]. When exposed to an in vivo microenvironment, a titanium surface spontaneously forms an evasive TiO2 layer that is highly bioinert and provides excellent corrosion resistance [54]. Despite these favorable characteristics, the chemically inert surface of titanium implants is not able to form a firm and permanent fixation with the biological tissue to last the lifetime of the patient. Additionally, fibrous tissue encapsulation around the titanium implants leads to implant failure [51].
One of the possible techniques to reduce fibrous capsule formation and enhance tissue integration is to modulate surface structure and topography that directly aid in cell adhesion and mineralization [20]. Buser et al. observed that modifying the titanium implant surface with microtopography resulted in enhanced implant integration of underlying bone tissue in vivo [55, 56]. This is a widely accepted technique to enhance tissue integration of dental implants; however, these surface modification techniques at a micro length scale are far from satisfactory in preventing bone resorption and result in limited interaction with the natural tissues [57, 58].
Recent studies have shown that nanotopography of dental implants is far more effective in overcoming these problems. It is observed that nanoengineered surfaces are able to induce osteoinductive signals to cells and facilitate their adhesion to the implant surface [20]. Nanotopography significantly modifies the biochemical and physiochemical characteristics of the implant surface and directly interacts with cellular components, favoring extracellular matrix deposition or formation of mineralized tissue at the dental implant surface [20]. Moreover, nanostructured titanium implants promote cell adhesion, spreading and proliferating as these nano surfaces directly interact with membrane receptors and proteins [20].
Apart from physical modification of titanium implant surfaces, chemical techniques to modify the surface characteristics have also been shown to influence dental tissue integration. For example, Scotchford et al. modified surface characteristics by functional groups (RGD) using molecular self-assembly to improve the osteointegration of a titanium implant surface [59, 60]. The RGD domains are immobilized on the titanium surface via a silanization technique using 3- aminopropyltriethoxysilane. Another method to impart nanotopography on the titanium implants includes chemical etching [61]. In this technique, the implant surface is treated with NaOH, which results in the formation of nanoetched structures and the formation of a sodium titanate layer. When this treated surface is subjected to simulated body fluid (SBF), nHAp crystals are deposited that aid in osteointegration of dental implants. In another study, Wang et al. showed that surface etching techniques to obtain nanostructure on the surface of implants can also improve mineralization on the implant surface [62]. They observed that the amorphous TiO2 resulting due to the etching of the implant surface by H2O2 results in formation of mineralized tissue [63, 64].
Deposition of bioactive ceramic nanoparticles on the implant surface can also enhance the bone-bonding ability [65, 66]. Nanosized calcium phosphate is deposited on the implant surface using the sol–gel transformation method to promote the formation of mineralized tissue on the implant surface [65, 66]. The implant surface modified with calcium phosphate promotes osteoblast attachment and spreading as shown by elongated filopodia. This resulted in interlocking of the implant with the adjoining bone in a rat model.
In a similar approach, Jiang et al. showed that combining nano- and microtopography, significantly enhanced osteointegration can be obtained (Fig. 2.5) [50]. They subjected the titanium implant surface to a dual chemical treatment consisting of acid etching followed by an NaOH treatment. The dual treatment resulted in nanostructured pores 15–100 nm in size and a microporous surface with a 2- to 7-μm size. The treated implant surface showed improved hydrophilicity, enhanced bioactivity, and increased corrosion resistance. Due to an increase in surface area, a significant increase in protein adsorption was observed on the nano/micro titanium implant. Thus, with the surface-functionalized and surface topography–modified titanium substrate, this ceramic-–based material is clinically successful as a dental implant in regeneration of endosseous tissues.
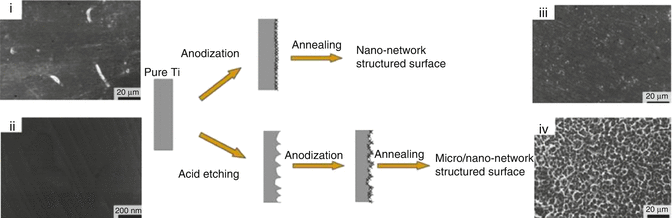

Fig. 2.5
Schematics showing surface modification of titanium implant using anodization and chemical surface etching. SEM images showing (i, ii) blank titanium, (iii) nano structured modified titanium surface and (iv) micro/nano modified surface (Adapted from Jiang et al. [50]. With permission from Elsevier)
2.3.4 Bioinert Zirconia Nanoparticles in Dentistry
Zirconia (or zirconium dioxide) is a polycrystalline biocompatible ceramic with low reactivity, high wear resistance, and good optical properties and thus extensively used in dental implantology and restorations [67, 68]. The mechanical properties of zirconia can be improved by phase transformation toughening using stabilizers such as yttria, magnesia, calcium, and ceria to stabilize the tetragonal phase of zirconia [69–72]. However, tetragonal zirconia is sensitive to low temperature degradation and results in a decrease in mechanical strength and surface deformation [68, 73]. By reducing the grain size of zirconia to nanoscale, phase modification can be arrested. Garmendia et al. showed that nanosized yttria-stabilized tetragonal zirconia (Y-TZP) can be obtained by spark plasma sintering [74, 75]. Nanosized Y-TZP showed significantly higher temperature stability and crack resistance compared to macrosized Y-TZP and zirconia [76].
Another approach to increase the mechanical properties of zirconia is to incorporate various nanoparticles such as carbon nanotubes and silica nanoparticles. For example, Padure et al. improved the toughness of nanosized Y-TZP by incorporating single-wall carbon nanotubes (SWCNTs) [76]. The addition of SWCNTs as a reinforcing agent to Y-TZP results in enhanced mechanical strength and making it attractive for dental restoration. In a similar approach, Guo et al. fabricated Y-TZP nanocomposite by reinforcing with silica nanofiber. The reinforced nanocomposite showed significant increase in the flexural modulus (FM), fracture toughness, flexural strength, and energy at break (EAB) compared to Y-TZP.
Nanosized zirconia can be used as a reinforcing agent in various dental fillers. Hambire et al. incorporated zirconia nanoclusters within a polymer matrix to obtain dental filler. The addition of zirconia nanoparticles resulted in significant increases in mechanical stiffness and enhanced tissue adhesion. Lohbauer et al. showed that a zirconia-based nanoparticle system can be used as dental adhesive (Fig. 2.6) [77]. Zirconia nanoparticles (20–50 nm) were prepared via a laser vaporization method. These nanoparticles are incorporated within the adhesive layer and have shown to increase tensile strength and promote mineralization after implantation.
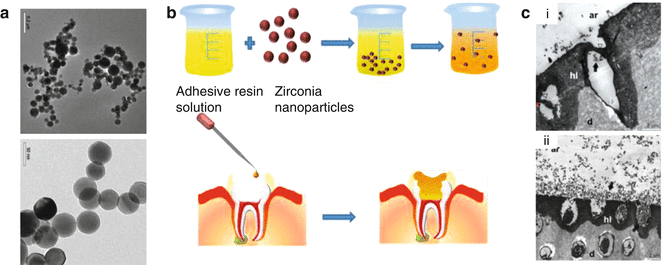

Fig. 2.6
(a) TEM images of zirconia nanoparticles prepared by laser vaporization. (b) Resin solution acting as adhesive is combined with zirconia nanoparticles. The composite resin loaded with zirconia nanoparticle showed uniform dispersion after ultrasonication. The adhesive dental resin loaded with nanoparticles are incorporated within to etched dentin with the implant. (c) TEM photographs showing dental resin composite with (i) 5 % wt and (ii) 20 % wt zirconia nanoparticles. The nanocomposite loaded with zirconia nanoparticles showed formation of submicron crystal enhancing remineralization and bioactivity at the implant-dentin interface. Adhesive resin (ar), hybrid layer (hl), demineralized dentin (d) are represented in image. Black arrow represents nanoparticles and open arrow represent formation of hybrid layer. (Adapted from Lohbauer et al. [77]. With permission from Elsevier)
Overall, nanostructured Y-TZP and nanosized TZP are extensively used in the field of dentistry due to the bioinert characteristic and aesthetic quality. The addition of zirconium nanoparticles to dental filler and incorporation into the dental tissue layers significantly enhance the mechanical stiffness and can promote bone bonding, restoring the dental defect and promoting accrual bone growth.
2.3.5 Antimicrobial Silver Nanoparticles for Dental Restoration
Over the centuries, silver has been used extensively in the field of medicine owing to its antimicrobial property, unique optical characteristic, thermal property, and anti-inflammatory nature [78]. Nano and micro particles of silver are used in conductive coatings, fillers, wound dressings, and various biomedical devices. Recently, there has been a growing interest in using silver nanoparticles in dental medicine, specifically for the treatment of oral cavities due to the antimicrobial property of silver nanoparticles [79]. The antimicrobial property of silver is defined by the release rate of silver ions. Although metallic silver is considered to be relatively inert, it gets ionized by the moisture, which results in a highly reactive state. This reactive silver interacts with the bacterial cell wall and results in structural changes by binding to the tissue protein and ultimately causing cell death [80].
Due to silver’s antimicrobial properties, it is used in dental fillers, dental cements, denture linings, and coatings [81]. For example, Magalhaeces et al. evaluated silver nanoparticles for dental restorations [82]. They incorporated silver nanoparticles in glass ionomer cement, endodontic cement, and resin cement. The antimicrobial properties of these dental cements were evaluated against the most common bacterial species of Streptococcus mutans that are responsible for lesions and tooth decay. All these cements doped with silver nanoparticles showed significantly improved antimicrobial properties when compared to cement without any nanoparticles. In a similar study, Espinosa-Cristóbal et al. investigated the inhibition ability of silver nanoparticles toward Streptococcus mutans [83]. They evaluated the antimicrobial properties of silver on the enamel surface, which is commonly affected by primary and secondary dental caries. They evaluated three different sizes of silver nanoparticles (9.3, 21.3, and 98 nm) to determine minimum inhibitory concentrations (MICs) in Streptococcus mutans (Fig. 2.7). Due to the increase in surface area–to-volume ratio, silver nanoparticles with smaller size showed significantly enhanced antimicrobial properties.


Fig. 2.7
(a) Different size of silver nanoparticles characterized using TEM and DLS. (b) SEM images showing the inhibition efficiency of silver nanoparticles towards Streptococcus mutans species (i) 9.3 nm (ii) 21.3 nm (iii) 98 nm (iv) negative control (Adapted from Espinosa-Cristóbal et al. [86]. With permission from Elsevier)
Another application of these silver nanoparticles is evaluated in hard and soft tissue lining. Dentures (false teeth) are the prosthetic devices created to replace damaged teeth and are supported by hard and soft tissue linings present in the oral cavity [80]. These soft and hard tissue linings are subjected to higher mechanical stress during chewing and colonization of species, and this soft lining is one of the major issues due to invasion of fungi or plaque leading to mucosa infection. Candida albicans is the commonly found fungal colony near the soft linings. Chladek at al. modified these soft silicone linings of dentures with silver nanoparticles [84]. They showed that the addition of silver nanoparticles significantly enhances the antifungal properties and can be used for dental restorations. In a similar study, Torres et al. also evaluated antifungal efficiency of silver nanoparticles by incorporating it within denture resins [85]. They prepared denture resins by reinforcing silver nanoparticles within polymethylmethacrylate (PMMA). The addition of silver nanoparticles facilitates the release of silver ions and enhances antimicrobial activity. The denture resin loaded with silver nanoparticles showed improved inhibition of C. albicans on the surface along with an increase in flexural strength.
Apart from incorporating antimicrobial properties, silver nanoparticles also improve the mechanical strength of dental materials. Mitsunori et al. showed that the addition of silver nanoparticles to porcelain significantly enhances mechanical toughness [86
Stay updated, free dental videos. Join our Telegram channel

VIDEdental - Online dental courses


