Chapter 58 Multi-modality management of nasopharyngeal stenosis following uvulopalatopharyngoplasty
1 INTRODUCTION
Nasopharyngeal stenosis (NPS) after uvulopalatopharyngoplasty (UPPP) is a serious problem. More palatal procedures are being performed leading to an increased incidence of nasopharyngeal stenosis, but little has been written about this severe complication and its management.
Historically, adult NPS was a consequence of maxillofacial trauma or severe infection such as syphilis. Now NPS is seen as a complication of surgery such as tonsillectomy and adenoidectomy in the pediatric population, and UPPP in the adult population with obstructive sleep apnea. Rhinoscleroma, lupus, diphtheria, tuberculosis, acid burn, scarlet fever and aggressive velopharyngeal repair can also cause nasopharyngeal stenosis.1,2
The complication of severe nasopharyngeal stenosis following UPPP is well understood. Most cases are problems of surgical technique. The management of intraoperative or postoperative bleeding with nasopharyngeal packing and electrocautery is an important cause of NPS. Adenoidectomy in conjunction with UPPP carries an increased risk of nasopharyngeal stenosis. Other technical errors include excessive excision and cauterization of the posterior tonsillar pillars or undermining the posterior and lateral pharyngeal walls. Wound dehiscence, infection, necrosis and scarring in keloid-forming patients are some other causes of acquired NPS. Also, the use of KTP (potassium titanyl phosphate) laser to perform adenoidectomy is associated with an extremely high incidence of NPS.1
2 SURGICAL TECHNIQUE
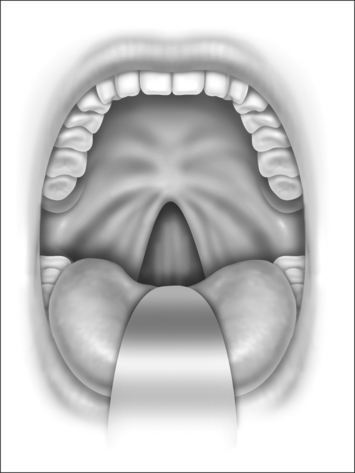
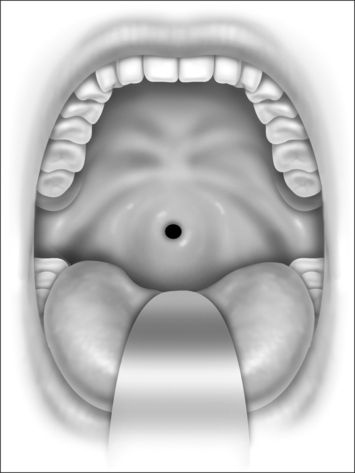
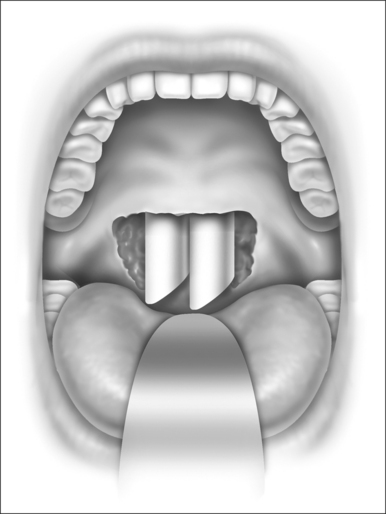
Nasopharyngeal stenosis is graded as type I (mild) when the lateral aspects of the palate adhere to the posterior pharyngeal wall. Type II (moderate) is the occurrence of circumferential scarring with small central opening of the soft palate. This circumferential opening is between 1 and 2 cm in diameter. Type III (severe) is when the entire palate fuses with the posterior and lateral palatal wall, leaving a residual opening less than 1 cm (Fig. 58.1&). Treatment for type I stenosis can be carried out as an outpatient, whereas treatment for type II and III stenosis may require general anesthesia in an inpatient setting. CO2 laser is used in conjunction with a scanning device at 30 watts, with a pharyngeal handpiece in a continuous mode in type I patients. During surgery, two nasopharyngeal tubes (size 6 or 7) are passed and left in place for 3–4 days as temporary stents (Fig. 58.2). A customized nasopharyngeal obturator is used in type II and III patients and inserted after removal of the stents on the third or fourth postoperative day (Figs 58.3 and 58.4). The nasopharyngeal obturator consists of an upper dental appliance with a customized nasopharyngeal extension as an obturator to stent open the nasopharynx. The patients use the stent during the day and remove it at night. The stent is left in place for 3–6 months (Fig. 58.5).
< div class='tao-gold-member'>
Stay updated, free dental videos. Join our Telegram channel

VIDEdental - Online dental courses