Fig. 12.1
The seven subsites of the oral cavity
Lip lesions frequently present earlier and are the most common sites for cancerous lesions, due to their relation to sun exposure. More advanced lesions are at risk of involving the mandible as well as being associated with the mental nerve. Oral tongue lesions may present early, but oftentimes encompass advanced disease with potential perineural invasion and regional metastasis. A depth of four to five millimeters is associated with significantly decreased rates of locoregional control. Early oral tongue OSCC is often associated with the previously discussed potentially malignant lesions, specifically erythroplakia, representing a potential target for mucosal therapy. Even more so than with lip lesions, tongue OSCC may extend to involve the mandible, and tumors with bone invasion beyond the periosteum may require a segmental mandibulectomy [10]. Floor of the mouth tumors frequently present in an advanced stage as lesions deeper than 1.5 mm are considered at greater risk for regional metastasis [11]. The retromolar trigone, an area extending from the alveolar ridges posteriorly over the mandibular ramus, is intimately associated with several nearby subsites that render most primary tumors of this area to be in an advanced stage by the time of presentation. Specifically, oropharyngeal structures such as the palatine tonsils and base of tongue, as well as oral cavity subsites such as the buccal space and floor of mouth, are directly near this area. The subsites in which oral cavity cancers arise also represent various targets for topical therapy. Specifically, numerous drugs outside the realm of cancer treatment have been designed for transmucosal delivery in the buccal, sublingual, gingival, and hard palatal areas.
12.4 Oral Mucosae and Dosage Form
The oral mucosae are about 50 cell layers thick and consist of stratified squamous epithelium. Mucosal tissue can be either keratinized or nonkeratinized and is without organized tight junctions, which renders them much more permeable than the skin. For a concise review, please see Harris and Robinson [12]. Oral cavity mucosae are generally divided into three regions: (1) sublingual or masticatory mucosa (ventral oral tongue and floor of mouth), (2) buccal mucosa (upper and lower lips and cheek lining), and (3) gingival mucosa (roof of the mouth) (Fig. 12.2) [13, 14]. Diseases of the oral mucosae are extremely common and can often be effectively treated with topical therapeutic approaches. Innovative new strategies that combine the use of small molecules or natural products and novel biomaterials have begun to emerge.
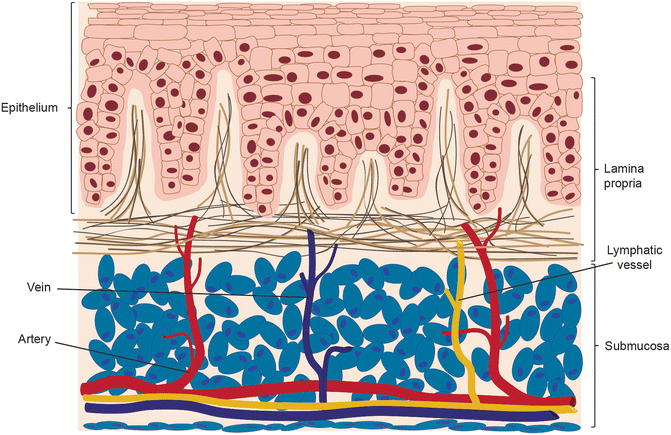

Fig. 12.2
Oral epidermis, lamina propria (dermis), and submucosa
Oral topical therapies are especially appealing in that the mouth is readily accessible for diagnosis and treatment [1, 2]. The surface area of the adult mouth is approximately 220 cm2 and is comprised of a permeable epithelial barrier that allows unidirectional small molecule (e.g., drug) transport [14]. Additionally, drugs administered to the oral mucosae, either systemically or topically, have the distinct advantage of avoiding first-pass hepatic metabolism. It has also been noted by several authors that oral/topical therapies can be quickly withdrawn at a patient’s request or in case of emergency (e.g., trauma or reaction to therapy) [15, 16]. Challenges associated with this approach are saliva (constantly wet environment), mastication, swallowing, or even talking that all act together and lead to short retention times and low therapeutic efficacy. There are a variety of mucosal therapy delivery systems described in the literature, including bioadhesive tablets that adhere to the surface until dissolution, patches and films for local and systemic release, adhesive semisolids such and ointments and gels, and liquid systems such as sprays and oral rinses or washes [15]. In our experience patients receiving mucosal therapies have reported the additional psychological benefit of feeling as though they are proactively participating in their treatment, especially during the time between diagnosis and surgical intervention. The ability to assuage patients’ fear is extremely important and may, in fact, provide a therapeutic benefit by helping them to comply with treatment regimens or to adopt healthy behavior changes. It is important that we continue to strive to identify medicinal compounds and create innovative biomaterials so that we can deliver more effective treatments.
12.5 Premalignant Lesions, the Development of OSCC, and Introduction to Chemoprevention
OSCC, through the accumulation of genetic insults, has a distinct histological and clinical transformation sequence from normal epithelial mucosa through varying degrees of dysplasia (e.g., mild, moderate, and severe) and carcinoma in situ (Fig. 12.3). What begins clinically as a leukoplakic (white) or erythroplakic (red) premalignant lesion may progress to malignancy (Fig. 12.4). Tobacco cessation plays an important role in the regression/arrest of premalignant lesions and may decrease the risk of subsequent secondary cancers and recurrences [17, 18]. Unfortunately, this is not always practical, as many patients at risk for malignant and premalignant oral lesions continue to smoke. Nonetheless, any oral cancer prevention trial would benefit from inclusion of tobacco cessation as one of the study parameters even though this could conceivably confound results [19].
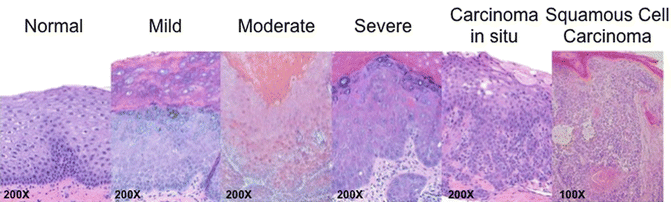
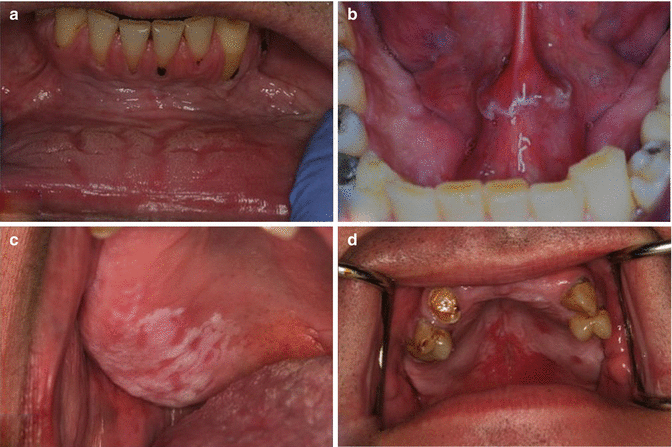

Fig. 12.3
Continuum of distinct histopathological states of oral cavity carcinogenesis. Mild dysplasia is characterized by changes in the epithelium such as basilar crowding and hyperplasia, cellular disorganization, and maturational disturbances not extending more than one-third of the epithelial thickness with little interruption of the keratin layer. Moderate epithelial dysplasia includes the above parameters more than one-third, but not extending beyond two-thirds of the entirety of the epithelium. Severe dysplasia included the above parameters extending beyond two-thirds of the epithelial thickness but not affecting the entirety of the epithelium. Additional features included frequent mitotic figures, cellular pleomorphism, nuclear atypia, and some early disturbance of the keratin layer. Carcinoma in situ appears as a full-thickness epithelial change with the above features, an expansion of multiple layers of cells into the suprabasal and intermediate layers, and with disturbance of the keratin layer but without penetration of the basement membrane

Fig. 12.4
Premalignant oral cavity lesions that are potentially amenable to mucosal therapies. (a) Leukoplakic lesion of the lower anterior vestibule. Patient is a long-term snuff user. Histopathology smokeless tobacco keratosis with mild epithelial dysplasia. (b) Leukoplakic lesion of the floor of the mouth. Patient is a former smoker with 40+ pack-year smoking history. Histopathology epithelial hyperplasia with keratosis and focal epithelial dysplasia. (c) Leukoplakic lesion associated with the edentulous ridge of the right maxilla extending from the premolar area to the soft palate. Patient is a smoker and wears a partial denture. Histopathology Moderate epithelial dysplasia with keratosis. (d) Erythroleukoplakia of the hard and soft palates. Patient is HIV positive (well controlled) and smokes cigarettes and marijuana. Histopathology Severe epithelial dysplasia (Photos courtesy of Dr. Steven Barket, DMD)
One immediate clinical advantage of treating premalignant tissues is that they are both easily visible by examination and clearly accessible for biopsy. There has not been, however, one single modality of chemoprevention that has taken over as the standard of care. Oral systemic medications and topically applied preparations have been examined in chemoprevention clinical trials, with local therapies (e.g., rinses, gels, mucoadhesive patches, and injections) having slightly better clinical results as well as more organized and tightly controlled experimental parameters [20]. There have been five major classes of local therapeutics that have been evaluated in clinical trials: (1) vitamin A derivatives, (2) adenoviruses, (3) chemotherapeutic medications, (4) cyclooxygenase (COX) inhibitors, and (5) natural products [20].
It is important to consider which type of preparation is ideal for chemoprevention and for which premalignant condition. Conceptually, the oral cavity can be considered a single continuous epithelial surface. Chronic exposure to chemical carcinogens (e.g., tobacco smoke and alcohol) in the oral cavity affects the entirety of the epithelium to varying degrees depending on the subsite (e.g., floor of the mouth). Considering that dysplastic lesions arise in a field of affected epithelial cells (i.e., field cancerization), patients may benefit from both a lesion-specific, site-directed topical therapy and a field-covering rinse. Thus, such a strategy may simultaneously treat the offending lesion and prevent the development of future lesions or recurrences. To thoroughly examine this issue, the landmark paper on field cancerization must be discussed. The topic was first introduced by Slaughter in 1953 and proposed to explain the appearance and development of multiple tumors and recurrence in oral squamous cell carcinoma [21]. There has been evidence to show that even after complete resection with negative margins, cancer can develop from genetically altered cells in the field that were left behind postoperatively [22]. In a recent review by Jaiswal et al., the presence of a field with genetically altered cells has been shown as a risk factor for cancer, evidenced by the high incidence of secondary primary tumors postoperatively [23]. Theoretically a chemopreventive rinse that acts on the entire oral cavity may prevent the outgrowth of new tumors. Combined with a lesion-specific agent impregnated in a patch or a gel that acts directly on the malignant portion, this would potentially give the patient the best chance to be cancer-free while avoiding harmful side effects from systemic chemotherapy or radiation.
Large tumors and those that involve adjacent structures, including the lymphatics and/or blood supply, are likely not amenable to adjuvant or neoadjuvant topical therapy. In contrast topical chemoprevention may be most ideal in certain disease processes, such as oral premalignant lesions including erythroplakia, and superficial malignancies (such as a carcinoma in situ). These topical gels or mucoadhesive patches could be used neoadjuvantly to simultaneously shrink the existing tumor and prevent or delay the local spread of malignant cells. Topical therapies could also be used in an adjuvant fashion by placing directly onto the healed excision site following surgery to prevent recurrence. After treatment, covert residual foci of microscopic invasion at the excision margin may be best managed by topical treatment and not additional resections or postoperative radiation. For example, the current standard of care in these types of cancers is to give postoperative radiation therapy if there are positive tumor margins on permanent section pathology. Another potential reason to give adjuvant radiation therapy (RT) is the presence of perineural invasion, which has been noted to indicate aggressive disease and a poorer prognosis [24].
There is conflicting evidence supporting a survival benefit in the use of RT for tumors with perineural invasion with negative surgical margins [25, 26]. However, RT has its own adverse effects including dysphagia, xerostomia, and resorption of the alveolar ridge associated with edentulism [26, 27]. This can also lead to anorexia and malnutrition and is a large cause of concomitant morbidity seen in head and neck cancer patients [28]. The benefit of using topical preparations in this setting can be quite large by potentially decreasing the need for radiation therapy in patients whose microscopic disease can be more simply controlled while negating the potentially harmful radiation side effects. There are evident risks, however, including the potential for mucositis that can occur with the treatment of any local preparation; established guidelines are in place for the prevention and treatment of mucositis. Further research should be undertaken to directly compare postoperative radiation with local chemoprevention for tumors with positive margins and/or perineural invasion with no other serious risk factors (i.e., lymph node extension, large tumors >4 cm, invasion of tumor into adjacent structures, extracapsular lymph node spread).
12.6 Topical Therapies to Treat Oral Premalignant Lesions and OSCC
Although theoretical considerations suggest that topical delivery systems provide superior effects on premalignant and localized and superficial malignant lesions − with potentially limited toxicities − practical considerations raise questions. An understanding of the oral mucosal drug delivery system is paramount in evaluating the available literature and identifying areas for further study [2].
The major advantage of mucosal therapy is direct treatment of superficially invasive cancers in the oral cavity using therapeutic agents with mitigated risk of systemic toxicity. Adverse effects of systemic chemotherapy include bone marrow suppression and inhibition/depletion of continuously renewing cells throughout the body. These adverse effects are less likely with locally applied therapy because the total dose is considerably lower. Alternatively, a potential adverse effect of mucosal therapy includes the localized inflammatory response (mucositis). Keefe et al. provided some clinical guidelines for the prevention and treatment of mucositis [29]. The authors published a table noting the risk of grade 3–4 oral mucositis and diarrhea with chemotherapy regimen. The data obtained from multiple studies indicated that the serious grade 3–4 oral mucositis was present in the varying chemotherapeutic regimens at a percentage ranging from approximately 1 % to as high as 13 %. The grade 3–4 oral mucositis is typically exacerbated by a falling white count associated with the chemotherapy. This pathology predisposes patients to secondary infections of the mucosal lining. However, this process is an unlikely effect of mucosal therapy given that bone marrow depletion would not occur with a mucosal therapy. In our opinion the risk of mucositis with local mucosal therapy is low (and likely manageable), but is certainly an important consideration with this treatment approach.
Mucosal absorption of applied chemotherapeutic agents will vary by compound and preparation (e.g., the vehicle) and the mode of delivery. We have observed, for example, that approximately 10 % of resveratrol administered in an oral rinse is absorbed into the mucosa (unpublished observation). The remainder is absorbed by the superficial mucosal tissues including the tumor tissue, or expectorated. Gastrointestinal absorption is not a factor. An early work discussing the permeability of oral mucosa was authored by C.A. Squier and presented in Critical Reviews in Oral Biology and Medicine [30]. He noted that keratinized areas such as gingiva and hard palate are least permeable and non-keratinized lining areas are most permeable. “Substances with different chemical properties traverse the barrier region by different routes, some crossing the cell membrane and entering the cell (a transcellular or intracellular route), others passing between by an intercellular route.” He further noted that the floor of the mouth and the lateral border of the tongue are more permeable than the buccal mucosa and stated that the examination of the kinetics of penetration of a variety of compounds across the oral mucosa under different conditions in vitro led to the conclusion that the process is most likely to be one of simple diffusion. The lining of the oral mucosa (e.g., the floor of the mouth and the lateral border of the tongue) are regions of high permeability and have been designated as high-risk areas for OSCC.
12.7 NSAID and Other Topical Chemopreventions
COX-2 expression is increased in oral cancers and correlates with poor prognosis [31, 32]. Increasing amounts of COX-2 expression (compared to normal adjacent and healthy patient controls) have been widely reported in OSCC, other head and neck SCCs, mucosal lesions, and oral intraepithelial neoplasia [31, 33, 34]. This enzyme promotes the formation of prostaglandins from arachidonic acid and can be induced by growth factors and tumor promoters. COX-2 facilitates cell division, angiogenesis, and reactive oxygen species formation and interferes with apoptosis and immune function [31, 35–38]. Hence, there is a strong theoretical basis for further investigation into interference with this system in the management and prevention of premalignant and malignant oral lesions.
Nonsteroidal anti-inflammatory drugs (NSAIDs) have been widely studied in the chemoprevention of a variety of cancers including oral cavity neoplasms. NSAIDs inhibit COX-1 and COX-2 and thereby prevent the synthesis of prostaglandins and eicosanoids such as thromboxane. Only a few studies have been dedicated to examining the impact of NSAID incorporation in an oral topical formulation for chemoprevention. In addition to any benefit that might be realized in the general “precancer” population, it is worthy to note that for patients with various risks for cardiac and GI sequelae, topical treatment with cyclooxygenase 2 (Cox-2) inhibitors presents a particularly valuable potential strategy [39].
Li and colleagues have evaluated the expression of enzymes related to arachidonic acid metabolism and the impact of chemopreventive therapies utilizing agents interfering with this COX-2 in a 7,12-dimethylbenz[a]anthracene (DMBA)-induced oral carcinogenesis model [40]. Examining oral cancer tissues, 5-lipooxygenase, and COX-2 expression was increased in OSCC specimens from hamsters [40, 41]. Topical application of zileuton, a 5-lipooxygenase inhibitor, significantly decreased the incidence of hamster OSCC, and topical application of celecoxib (Celebrex) also decreased the incidence of OSCC [42]. Combination topical therapy applied in mineral oil was further noted to have an inhibitory effect on carcinogenesis in a male Syrian golden hamster pouch model [40]. Although the preparation used did not contain NSAID, Mallery et al. reported that black raspberry extract delivered in a mucoadhesive freeze-dried black raspberry gel significantly decreased COX-2 expression in a small cohort human trial of patients with intraepithelial neoplasia (IEN) [34], further supporting the hypothesis that COX-2 is a valid target for mucosal therapy.
NSAID use has been suggested to have a chemopreventive impact in numerous cancers, most prominently in colorectal cancers. A similar trend has been suggested among patients with oral cancers. Conducting a nested case-control study of 454 patients gathered from a population-based cohort, long-term use of NSAIDs was associated with a significantly reduced risk of oral cancer, with a hazard ratio of 0.47 [43]. It should be noted that the same study demonstrated that long-term use of NSAIDs also significantly increased the risk of cardiovascular disease-related death and did not reduce mortality overall. Additional studies in both animals and humans have suggested the potential utility of NSAIDs for chemoprevention among patients with premalignant oral cavity lesions [44–46]. Although there has been study of COX inhibitors administered systemically in humans, there has been little analysis with regard to topical application. Mulshine et al. performed a prospective controlled study among 57 human patients with oropharyngeal leukoplakia that were recruited from the M.D. Anderson Cancer Center, the University of Colorado, and the NIH clinical center [47]. In this study, 38 patients were given 10 mL of a 0.1 % ketorolac rinse solution twice daily over a 3-month period, while another 19 patients were provided a placebo. The response criteria were defined as disappearance of all measurable disease for at least 30 days or decrease in the area of measurable leukoplakia by at least 50 %. No significant difference in response rate was observed upon comparison of histology results from both of these groups. In the Mulshine study, examining ketorolac rinses, the authors argued that the doses and concentration of the rinse were not sufficiently adequate to facilitate a response in leukoplakic tissue. The study led them to logically suggest pilot studies to elucidate the feasibility of utilizing a sustained release formula [46]. Furthermore, the short periods of time used for the ketorolac rinses (two rinses a day for 30 s each) might not have been sufficient to effectively deliver the drugs. Although there was not a significant difference in the response to treatment in the Mulshine study, topical application of NSAIDs is an area that requires additional study before definitive conclusions can be made. Specifically, the permeability and local absorption levels of any topically applied single agent or combination of agents need to be considered.
There is a strong theoretical and scientific foundation for the continued study and attempts to modulate COX-2 function in premalignant and malignant oral lesions. With the paucity of definitive data among human cohorts, evidence of gastrointestinal and cardiac toxicities associated with systemic therapies, and the potential advantages of topical administration, the use of topical NSAIDs represents a significant area of further ongoing inquiry.
12.8 Chemoprevention of OSCC Targeting EGFR
The epidermal growth factor receptor (EGFR) represents the only molecular target with a US Food and Drug Administration (FDA)-approved drug for use in the treatment of OSCC. EGFR expression is increased in upper aerodigestive tract malignancies and is overexpressed in leukoplakic lesions [35, 48]. Activation of EGFR can lead to COX-2 induction, further strengthening the argument for targeting arachidonic acid metabolism for the chemoprevention of oral malignancies [35]. Cetuximab was approved in 2006 for the treatment of recurrent/metastatic HNSCC alone or in combination chemoradiation; it is currently approved for intravenous administration and is the only FDA-approved EGFR inhibitor [49]. As part of a chemoradiation regimen, it significantly increased the rate of response in OSCC patients (compared to radiation alone) and increased overall survival from 29.3 to 49.0 months [50]. Despite the success of cetuximab and chemoradiation improving locoregional control and reducing mortality in patients with advanced head and neck cancers, there has been little study regarding the utility of EFGR inhibitors administered directly to affected tissues.
The toxicities associated with systemic chemotherapies, including cetuximab, make inquiry into finding acceptable topical formulations with limited adverse effects a priority. Topical EGFR inhibitors have been evaluated in several animal models. The over-the-counter laxative polyethylene glycol (PEG) has been well studied as a chemopreventive agent in colon carcinogenesis [51–53]. PEG has been reported to bind to and internalize EGFR and direct it to the 26S proteasome for degradation [54]. PEG also induces cell cycle arrest at the G1–S phase [55] and can disrupt β-catenin transcription leading to the downregulation of E-cadherin and SNAIL [51]. Using a human HNSCC line and a rat model, daily topical administration of a PEG preparation to the buccal floor and hard palate significantly reduced tumor size and multiplicity. Furthermore, PEG application reduced cell proliferation and EGFR expression in both oral cavity mucosa and tumor tissue [55]. These results suggest that PEG, possibly through its anti-EGFR activity, might elicit potent chemopreventive properties in vivo.
Sun et al. evaluated topical EGFR inhibitor application to oral premalignant lesions in an animal model. Topical application of GW2974, a dual EGFR and Neu (Erb-2) tyrosine kinase inhibitor, in hamsters three times a week decreased both the incidence and size of grossly visible tumors and suppressed the number of premalignant lesions (including hyperplasia and dysplasia) in a DMBA-induced oral carcinogenesis model. In the same study, it was observed that GW2974 reduced prostaglandin levels and cell proliferation [56]. These results provide additional evidence that underscore the role of arachidonic acid metabolism in promoting oral carcinogenesis and that agents interfering with this pathway remain a promising area of study. The success of systemic cetuximab, along with preliminary data detailed above, presents a strong rationale for the continued study of topical anti-EGFR agents for chemoprevention.
12.9 Topical Therapies and Retinoids
Retinoids (vitamin A/retinol, 13-cis retinoic acid [RA]) and other vitamin A derivatives (e.g., fenretinide) have been examined in oral cancer chemoprevention trials in recent decades. Vitamin A is an essential micronutrient in the human diet and plays an integral role in cell differentiation [57]. Exogenous treatment of normal and malignant oral epithelial cells with vitamin A treatment induced terminal differentiation [57, 58]. However, systemic and local toxicities have limited its continued use for oral cancer chemoprevention studies and the treatment of oral premalignant lesions. Recently, Mallery and colleagues have reopened the door to vitamin A analog chemoprevention with physiology-based pharmacokinetic studies of fenretinide-impregnated mucoadhesive patches [34, 59]. Nonetheless, this longstanding discussion deserves attention from practitioners and scientists involved in the study and management of OSCC, and several significant findings are highlighted below.
As previously discussed, arachidonic acid metabolism, particularly with respect to COX-2 function, has been studied in the tumorigenesis of OSCC. Long-term NSAID use has been shown to reduce the incidence of several solid malignancies, including colorectal cancer, in numerous animal studies [60], and EGF can upregulate Cox-2 expression in OSCC cell lines. In one analysis, Mestre et al. treated human OSCC cells with retinoids, demonstrating that such treatment suppressed EGF-mediated COX-2 induction [60].
Vitamin A is required and plays several important functions in the differentiation of healthy epithelial tissue. Specifically, vitamin A derivatives induce apoptosis and terminal differentiation, thereby thwarting uncontrolled proliferation, a hallmark of cancer [61, 62]. Conversely, premalignant tissues have reduced retinoic acid receptor (RAR) mRNA expression. Treatment with 13-cis retinoic acid induces the expression of RARs, thus preventing further progression of these tissues [63]. Utilizing 4-hydroxyphenyl retinamide, another retinoid derivative, Scher and colleagues noted a dose-dependent decrease in cell proliferation in OSCC cells [62]. Examining the expression of apoptotic markers in patients with oral premalignant lesions including leukoplakia, an increase in apoptotic bodies was noted with topical treatment of 13-cis-retinoic acid [64]. Another study found that two-thirds of patients administered systemic 13-cis retinoic acid had a decrease in the extent of oral leukoplakia [65]. It should be noted that these studies observed adverse effects including mucosal dryness and hypertriglyceridemia in more than half of patients.
Despite its potential for low systemic adverse effects, topical therapy still requires patient adherence to its repeated use. As noted above, mucosal dryness and other local irritation are factors that patients may need to be advised about [57]. Nonetheless, further trials with topical retinoids have shown promise in addition to those detailed above. Performed over three decades ago, Shah et al. conducted a trial where they treated 16 patients with leukoplakia using isotretinoin lozenges. Nine of these 16 patients had partial or complete responses, ranging from thinning of the leukoplakia and reduction of its surface by at least 50 % to complete remission. Similar to previously discussed studies, this cohort had a small study population and lacked a control group. It should also be noted that this study included a mix of both topical and systemic therapies.
More recent trials using topical therapies have been performed. Epstein and colleagues [58] employed a gel (0.05 % tretinoin) in 26 patients, with a mean time of participation exceeding 3 years. Complete disappearance of leukoplakic lesions was noted in 27 % of patients, while a 54 % experience a partial response. Approximately one in five patients noted local irritation, far less than the number of patients reporting side effects in other studies examining systemic administration of vitamin A derivatives. Importantly, only about 50 % of patients experienced recurrence after cessation of treatment.
Stay updated, free dental videos. Join our Telegram channel

VIDEdental - Online dental courses


