Introduction
Enamel demineralization is a problem in orthodontics. Fluoride is partially effective in addressing this problem, but additional treatment options are needed. The objective of this prospective randomized controlled trial was to determine the effectiveness of a new product, MI Paste Plus (GC America, Alsip, Ill), in the prevention or reduction of white spot lesions in orthodontic patients.
Methods
Sixty patients who were undergoing routine orthodontic treatment were recruited for this prospective randomized clinical trial. A double-blind method of randomization was used to determine whether each patient received the MI Paste Plus or a placebo paste (Tom’s of Maine, Salisbury, United Kingdom). Each patient was asked to administer the paste by using a fluoride tray for a minimum of 3 to 5 minutes each day at night after brushing. Photographic records obtained in a light-controlled environment were used to record the presence or absence of white spot lesions in both groups. The enamel decalcification index was used to determine the number of white spot lesions per surface at each time interval. Patients were followed at 4-week intervals for 3 months. A scoring system from 0 to 6 was used to determine the level of caries or cavitations. This system was also used for each tooth at each time interval.
Results
Fifty patients (26 using MI Paste Plus, 24 using the placebo paste) completed the study. The enamel decalcification index scores for all surfaces were 271 and 135 at the start of treatment and 126 and 258 at the end of treatment for the MI Paste Plus and placebo paste groups, respectively. The enamel decalcification index scores in the MI Paste Plus group reduced by 53.5%, whereas the placebo group increased by 91.1% during the study period. A 3-way analysis of variance (ANOVA) was done for the average enamel decalcification index scores. The surface type, the product/time interactions, and the product/surface interactions of the mean enamel decalcification index scores were significant ( P <0.05).
Conclusions
MI Paste Plus helped prevent the development of new white spot lesions during orthodontic treatment and decreased the number of white spot lesions already present. The placebo paste had no preventive action on white spot development during orthodontic treatment; the number of lesions actually increased. MI Paste Plus reduced white spots on the gingival surfaces; the placebo paste had the opposite effect. The incisal surface effect on the mean enamel decalcification index scores over time and between products was highly significant. The incisal enamel decalcification index scores were consistently higher than those for the other surfaces (mesial, distal, and gingival).
Enamel decalcification, or white spot lesion formation, is a phenomenon that occurs primarily on the smooth enamel surfaces of teeth, notably in the gingival third of the crown. Demineralized enamel, the precursor to caries formation, can be attributed to fixed orthodontic appliances and prolonged exposure to bacterial plaque. Bacterial plaque promotes the accumulation of acidic by-products and demineralization that leads to successive changes in the optical properties of subsurface demineralized enamel. The progression to clinically detectable white spot lesions can occur as early as 1 month after the placement of orthodontic appliances.
Over the past 30 years, several studies have reported increases in white spot lesions after orthodontic treatment. Although a large portion of the nonorthodontically treated population experiences some form of decalcification, orthodontic patients have shown increases in both new lesions and the severity of preexisting enamel opacities. Approximately 50% of orthodontic patients develop white spot lesions in at least 1 tooth, compared with only 24% in those not undergoing orthodontic treatment.
Appliance removal halts white spot formation, and further elimination of cariogenic factors through diligent oral hygiene efforts inactivates incipient lesions, which might regress over time. Complete elimination of lesions is unlikely because of the rapid remineralization of the enamel surface with a high concentration of fluorides, which restrict the passage of ions into the deeper, more affected layers. Therefore, immediate application of a high concentration of fluoride is not recommended. There might be fewer enamel discolorations with time because of further remineralization, but regression is primarily credited to gradual surface abrasion of the tooth structure.
Such problems with enamel decalcification in orthodontic patients have caused clinicians to search for a solution to orthodontic-associated demineralization. Because fluoride treatment immediately upon debonding is not advocated, clinicians have proposed fluoride treatment and fluoride-releasing materials at the commencement of therapy. Recommendations include oral hygiene instructions and reinforcement; fluoridated toothpastes, varnishes, and mouthwashes; and fluoridated water supplies. Lack of patient compliance hinders these efforts.
A new product, MI Paste Plus (GC America, Alsip, Ill), is currently available and has been shown in some initial reports to be useful in the reduction of white spot lesions. Although MI Paste Plus does not remove the need for compliance, it is hoped that it provides a new way to prevent and resolve decalcifications that develop during orthodontic treatment. MI Paste Plus reportedly restores minerals to the teeth and helps to stimulate saliva production. It contains casein phosphopeptide-amorphous calcium phosphate,a special milk-derived protein that is potentially a breakthrough in oral health care in helping to remineralize teeth. More specifically, casein phosphopeptide is produced from digestion of the milk protein casein by aggregation with calcium phosphate and purification by ultrafiltration. The casein phosphopeptide stabilizes the amorphous calcium phosphate in solution, maintaining high concentration gradients of calcium and phosphate in the white spot lesion, thus effecting high rates of enamel remineralization.
The aim of this study was to determine whether MI Paste Plus has an effect on the formation and resolution of white spot lesions in patients undergoing orthodontic treatment. It is believed to provide a new way to reduce and prevent the decalcifications that develop during orthodontic treatment.
Material and methods
The study protocol was reviewed and approved by the institutional review board of the University of Texas Health Science Center at Houston.
Sixty from a possible 65 patients who were undergoing routine orthodontic treatment were recruited for this prospective double-blind randomized clinical trial. The patients were 12 years of age and older. A caries risk assessment was used to determine their caries risks. They were carefully selected for the study and included and excluded on the following criteria. The inclusion criteria were patients (1) with permanent dentition, (2) who would be compliant with using the paste (in the investigator’s opinion), (3) who had not extensively used fluoride regimens, and (4) who were 12 years of age and older. The exclusion criteria were patients (1) with any medical or dental condition that (in the investigator’s opinion) could impact the study results during its expected length, (2) currently using any investigational drug, (3) who planned to move within 6 months of enrollment, (4) who had received or were currently receiving fluoride treatment for white spot lesions, and (5) who had an allery to IgE casein.
The patients were recruited through the Orthodontic Postgraduate Clinic at the University of Texas Health Science Center Dental Branch at Houston. Sixty-five patients who expressed an interest in the study were approached to participate. Five did not want to participate for a variety of reasons after protocol procedures were explained and did not sign the consent form. Sixty patients agreed to participate in the study. A study administrator prepared the pastes and assigned the randomizations. The process of randomization was carried out by drawing the letters X and Y from a prepared pool of letters (X was the placebo [Tom’s of Maine, Salisbury, United Kingdom], and Y was the MI Paste Plus). The assigned X and Y pastes were given to a clinician to administer to the patients.
The paste was delivered in a prefabricated fluoride varnish tray and used once daily for 3 months. Patients placed the tray with the paste into the mouth and left it for 3 to 5 minutes after they had already brushed their teeth at night. The patients were asked to expectorate and not to rinse their mouth, eat, or drink after using the paste. Patients were reviewed every 4 weeks. During the recall period, photographic records and clinical examinations were carried out.
Photographic records were used to determine the presence of the white spot lesions in the study groups from the left first premolar to the right first premolar in both the mandible and the maxilla. A standard intraoral photographic camera was used, the photographs were taken in a light-controlled environment, and the photographs were captured with a preset protocol. The enamel decalcification index score (Banks and Richmond ) was used to determine the number of white spot lesions at each time point ( Fig 1 ). The 4 time points were start of treatment (T1), 4 weeks into treatment (T2), 8 weeks into treatment (T3), and 12 weeks into treatment (T4).
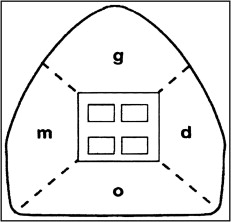
The international caries detection and assessment system was used for the clinical examinations; this is a visually based system. Only primary caries detection was used for the labial surfaces of the teeth. Essentially, a scoring system from 0 to 6 was used in determining the level of caries ( Table I ).
| ICDAS detection | ICDAS lay terms | ICDAS dental terms |
|---|---|---|
| 6 | Severe decay | Extensive cavity with visible dentin |
| 5 | Severe decay | Distinct cavity with visible dentin |
| 4 | Established decay | Underlying dentin shadow |
| 3 | Established decay | Localized enamel breakdown |
| 2 | Early stage decay | Distinct visual change in enamel |
| 1 | Early stage decay | First visual change in enamel |
| 0 | Sound | Sound |
Statistical analysis
Three-way analysis of variance (ANOVA) (StatView; SAS Institute, Cary, NC) was used to analyze the enamel decalcification index scores of the maxillary and mandibular right first premolars to the left first premolars at the 0.05 level of significance. Two-way ANOVA was used to analyze the international caries detection and assessment system scores. Fisher protected least significant difference intervals were used to compare mean enamel decalcification index scores.
The enamel decalcification index and the international caries detection and assessment system scores were randomly recorded for 5 sets of patients at the start and end of treatment. Three operators (M.A.R., J.T.N., C.H.K.) scored the photographs independently. Their scores were compared by using the t test; no statistically significant difference ( P ≥0.05) was found between the operators.
Results
This study was a prospective clinical trial with a double blind method of randomization. We found that MI Paste Plus, used as presented here, not only lessened the occurrence of white spots, but also actually reduced the number of white spots already present. The results of the statistical analysis are presented in Table II , and the reduction in scores in Tables III and IV .
| P value | Power | |
|---|---|---|
| Product | 0.61 | 0.080 |
| Time | 0.99 | 0.058 |
| Surface | <0.0001 | 1.000 |
| Product/time interaction | 0.0003 | 0.977 |
| Product/surface interaction | 0.03 | 0.696 |
| Time/surface interaction | 1.00 | 0.086 |
| Product/time/surface interaction | 0.97 | 0.166 |
| Surface | Sum of EDI at T1 | Sum of EDI at T4 | Change in EDI, T1-T4 (%) |
|---|---|---|---|
| All 4 surfaces | 271 | 126 | −53.5 |
| Mesial | 54 | 20 | −63.0 |
| Distal | 58 | 20 | −65.5 |
| Gingival | 38 | 21 | −44.7 |
| Incisal | 121 | 65 | −46.3 |
| Surface | Sum of EDI at T1 | Sum of EDI at T4 | Change in EDI, T1-T4 (%) |
|---|---|---|---|
| All 4 surfaces | 135 | 258 | +91.1 |
| Mesial | 18 | 29 | +61.1 |
| Distal | 10 | 32 | +220.0 |
| Gingival | 23 | 60 | +160.9 |
| Incisal | 84 | 137 | +63.1 |
The subjects were recruited over a 1-year period. Sixty-five patients who expressed an interest were approached to participate. Five did not want to participate for a variety of reasons after protocol procedures were explained and did not sign the consent form. Sixty patients participated in the study. Of the 29 patients, chosen randomly from the 60, who used the MI Paste Plus, 26 completed the study; of the 31 placebo patients, 24 completed the study. This amounted to 416 teeth and 1664 tooth surfaces studied for the MI Paste Plus patients, and 384 teeth and 1536 tooth surfaces studied for the placebo patients.
There was a decrease of 53.5% in the enamel decalcification index scores with MI Paste Plus usage from T1 to T4 ( Table III ). Moreover, the sums of the enamel decalcification index scores for all teeth in the MI Paste Plus group were 271 at T1 and 126 at T4 ( Table III ). The patients in the placebo group, on the other hand, showed a 91.1% increase in enamel decalcification index scores from T1 to T4 ( Table IV ). More specifically, the sums of the enamel decalcification index scores for all teeth in the placebo group were 135 at T1 and 258 at T4 ( Table IV ).
Consistent with the enamel decalcification index scores, MI Paste Plus was linked to a 44.8% decrease in scores over the same time period ( Table V ). Moreover, the sums of the international caries detection and assessment system scores for all teeth in the MI Paste Plus group were 145 at T1 and 80 at T4 ( Table V ). Also consistent with the enamel decalcification index scores, the placebo corresponded with an increase of 43.1% in the international caries detection and assessment system over the same time period ( Table V ). More specifically, the sums of the international caries detection and assessment system scores for all teeth in the placebo group were 116 at T1 and 166 at T4 ( Table V ).
| Product | Sum of ICDAS scores for all teeth | Change in ICDAS, T1-T4 (%) | |||
|---|---|---|---|---|---|
| T1 | T2 | T3 | T4 | ||
| MI Paste Plus | 145 | 129 | 95 | 80 | −44.8 |
| Placebo | 116 | 145 | 150 | 166 | +43.1 |
Stay updated, free dental videos. Join our Telegram channel

VIDEdental - Online dental courses


