Fig. 13.1
Tympanometric patterns associated with normal middle ear function (a), middle ear effusion (b), negative middle ear pressure (c), and reduced middle ear mobility (d) (Reprinted with permission from Roush and Grose (2006), p. 378)
Acute otitis media, when treated medically, is treated with antibiotics to cover for H. influenzae, S. pneumoniae, or M. catarrhalis. For most penicillin-resistant organisms, amoxicillin/clavulanate has proven effective (AAP 2004). When middle ear effusion persists for more than 3 months, as it often does in children with cleft palate, it is considered chronic. Tympanostomy tube placement is a common practice for those children, and improvements in hearing have been noted (Gould 1990) with relatively few complications (Curtin et al. 2009); however, multiple tube insertions have been associated with persistent conductive hearing loss (Goudy et al. 2006). Although it is unclear whether this is due to the placement of the tympanostomy tubes or to middle ear damage from the inflammation associated with otitis media, some clinicians have favored hearing aids over multiple tube placements (Maheshwar et al. 2002).
Complications from middle disease, although relatively rare, include perforation or retraction of the tympanic membrane, resulting in cholesteatoma. Cholesteatoma refers to a benign but locally erosive mass of squamous cells that often begins with a tympanic membrane retraction pocket or perforation. It is a potentially serious condition that warrants evaluation by an otolaryngologist and subsequent surgery.
Surgical repair of the palate has been shown to improve Eustachian tube function and decrease the frequency of otitis media and need for tympanostomy tubes (Bluestone 1971); however, it may take several years for Eustachian tube function to fully recover (Smith et al. 1994; Goudy et al. 2006). As with the non-cleft palate population, the likelihood of hearing loss decreases with age (Gordon et al. 1988); however, hearing loss persists in adulthood for some patients.
13.2 Assessment of Hearing
Newborn hearing screening is now a standard of care throughout the United States (JCIH 2007). Two technologies are used for newborn screening and both employ physiologic methods; that is, they involve physiologic measurement of auditory function obtained without the infant’s active participation. The first involves the measurement of otoacoustic emissions (OAEs) which are low-intensity sounds produced by the inner ear in response to acoustic stimuli (tones or clicks) and detected by a sensitive microphone placed in the ear canal. Successful recording of OAEs confirms a healthy inner ear (cochlea) and is consistent with normal or near-normal hearing sensitivity. Since the middle ear is involved in both the conduction of acoustic stimuli to the inner ear and reverse transmission of OAEs to the ear canal, their presence also confirms normal middle ear function. OAEs are “preneural” so their inclusion in the test battery makes it possible to evaluate the auditory system at the level of the inner ear without involving higher auditory centers. However, absent OAEs may be due to a variety of conditions ranging from middle ear dysfunction to profound cochlear hearing loss; other tests are needed to resolve their absence. OAE screening may be performed by a variety of professionals or support personnel, while diagnostic OAE testing is performed by an audiologist as part of a test battery. Another physiologic test used for both hearing screening and diagnosis of hearing loss is the auditory brainstem response (ABR). Like OAEs, ABRs are elicited by acoustic stimuli presented in the ear canal; however, ABRs are neurological responses obtained from surface electrodes attached to the head and provide information regarding the functional integrity of the auditory nerve and brainstem pathway. When used in a diagnostic test battery, audiologists use frequency-specific ABR testing to estimate hearing threshold levels.
Although OAE and ABR tests provide valuable information regarding the auditory system at the preneural, auditory nerve, and brainstem levels, they are not considered true tests of hearing in the perceptual sense. Comprehensive assessment of hearing requires behavioral tests, that is, procedures that involve observation of a listener’s response to sound. Methods used for behavioral assessment of hearing vary depending on the child’s age and developmental status. When infants reach a developmental age of approximately 6 months, most can be tested using an operant conditioning procedure known as visual reinforcement audiometry. By age 3–4 years, behavioral testing can be accomplished using conditioned play procedures, and by 5 years of age, typically developing children respond by raising their hand or pressing a response button.
To summarize, newborn hearing screening is conducted using physiologic measures: otoacoustic emissions or auditory brainstem responses, alone or in combination. Infants who do not pass the newborn hearing screening are referred to an audiologist for comprehensive assessment using these and other specialized procedures to determine if hearing impairment is present and, if so, to ascertain the type and degree of hearing loss. When infants reach a developmental age of approximately 6 months, they can be tested using behavioral methods. When permanent hearing loss is diagnosed, most children benefit from acoustic amplification (hearing aids). Those with severe-profound hearing loss are likely to benefit from a cochlear implant.
The initial goal of the audiologic assessment, whether conducted using physiologic or behavioral methods, is to obtain a frequency-specific estimate of the child’s hearing thresholds for each ear, based on detection levels for air- and bone-conducted test stimuli. Air conduction audiometry involves the presentation of pure tones from an earphone or insert receiver; bone conduction audiometry involves the presentation of signals through a bone vibrator placed behind the ear on the mastoid process. Testing is usually performed at octave intervals from 250 to 8,000 Hz for air conduction and 250–4,000 Hz for bone conduction. Thresholds are displayed on an audiogram which plots threshold levels in decibels hearing level (dB HL) as a function of frequency, using standard symbols (Fig. 13.2). The degree of hearing loss can be summarized by averaging the pure-tone air conduction thresholds in the mid-frequencies. Terms used to classify hearing levels include normal (0–15 dB HL), borderline normal (16–25 dB HL), mild (26–45 dB HL), moderate (46–75 dB HL), severe (76–0 dB HL), and profound hearing loss (>90+ dB HL). Borderline categories may be described using a combination of terms, such as moderate to severe (Roush and Grose 2006).
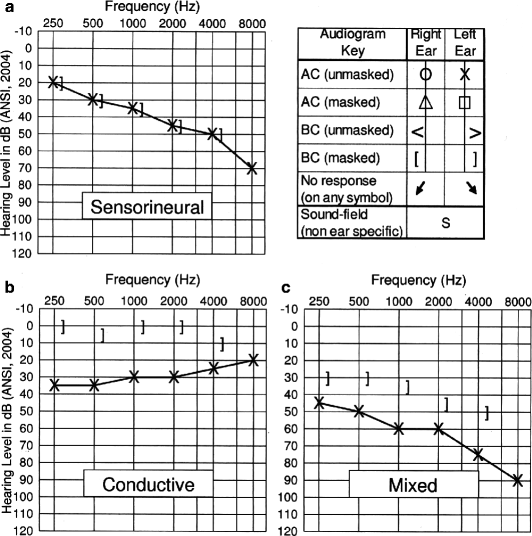

Fig. 13.2
Audiograms illustrating mild to moderate sensorineural hearing loss (a), mild conductive hearing loss (b), and moderate-to-severe mixed hearing loss (c) (Reprinted with permission from Kramer (2008), p. 153)
When pure-tone air conduction thresholds are abnormally elevated, bone conduction testing is performed to differentiate problems with sound transmission lateral to the inner ear. As shown in Fig. 13.2a, when air and bone conduction thresholds are equally elevated, the loss is described as sensorineural. Conductive hearing loss, illustrated in Fig. 13.2b, is characterized by normal or near-normal bone conduction thresholds with elevated air conduction thresholds. A mixed hearing loss, shown in Fig. 13.2c, is characterized by abnormal responses to both air conduction and bone conduction signals, with air conduction thresholds poorer than bone conduction thresholds.
13.3 Hearing Loss in Children with Cleft Palate and Craniofacial Anomalies
Stay updated, free dental videos. Join our Telegram channel

VIDEdental - Online dental courses


