Oral mucositis is a clinically important and sometimes dose-limiting complication of cancer therapy. Mucositis lesions can be painful, affect nutrition and quality of life, and have a significant economic impact. The pathogenesis of oral mucositis is multifactorial and complex. This review discusses the morbidity, economic impact, pathogenesis and clinical course of mucositis. Current clinical management of oral mucositis is largely focused on palliative measures such as pain management, nutritional support and maintenance of good oral hygiene. However, several promising therapeutic agents are in various stages of clinical development for the management of oral mucositis. These agents are discussed in the context of recently updated evidence-based clinical management guidelines.
Oral mucositis refers to erythematous and ulcerative lesions of the oral mucosa observed in patients with cancer being treated with chemotherapy, and/or with radiation therapy to fields involving the oral cavity. Lesions of oral mucositis are often very painful and compromise nutrition and oral hygiene as well as increase risk for local and systemic infection. Mucositis can also involve other areas of the alimentary tract; for example, gastrointestinal (GI) mucositis can manifest as diarrhea. Thus, mucositis is a highly significant and sometimes dose-limiting complication of cancer therapy .
Epidemiology of mucositis
Oral mucositis is a significant problem in patients undergoing chemotherapeutic management for solid tumors. In one study, it was reported that 303 of 599 patients (51%) receiving chemotherapy for solid tumors or lymphoma developed oral and/or GI mucositis . Oral mucositis developed in 22% of 1236 cycles of chemotherapy, GI mucositis in 7% of cycles, and both oral and GI mucositis in 8% of cycles. An even higher percentage (approximately 75% to 80%) of patients who receive high-dose chemotherapy before hematopoietic cell transplantation develop clinically significant oral mucositis .
Patients treated with radiation therapy for head and neck cancer typically receive an approximately 200 cGy daily dose of radiation, 5 days per week, for 5 to 7 continuous weeks. Almost all such patients will develop some degree of oral mucositis. In recent studies, severe oral mucositis occurred in 29% to 66% of all patients receiving radiation therapy for head and neck cancer . The incidence of oral mucositis was especially high in (1) patients with primary tumors in the oral cavity, oropharynx, or nasopharynx; (2) those who also received concomitant chemotherapy; (3) those who received a total dose over 5000 cGy; and (4) those who were treated with altered fractionation radiation schedules (eg, more than one radiation treatment per day).
Clinical significance of oral mucositis
Oral mucositis can be very painful and can significantly affect nutritional intake, mouth care, and quality of life . For patients receiving high-dose chemotherapy before hematopoietic cell transplantation, oral mucositis has been reported to be the single most debilitating complication of transplantation . Infections associated with the oral mucositis lesions can cause life-threatening systemic sepsis during periods of profound immunosuppression . Moderate to severe oral mucositis has been correlated with systemic infection and transplant-related mortality . In patients with hematologic malignancies receiving allogeneic hematopoietic cell transplantation, increased severity of oral mucositis was found to be associated significantly with an increased number of days requiring total parenteral nutrition and parenteral narcotic therapy, increased number of days with fever, incidence of significant infection, increased time in hospital, and increased total inpatient charges .
In patients receiving chemotherapy for solid tumors or lymphoma, the rate of infection during cycles with mucositis was more than twice that during cycles without mucositis and was directly proportional to the severity of mucositis . Infection-related deaths were also more common during cycles with both oral and GI mucositis. In addition, the average duration of hospitalization was significantly longer during chemotherapy cycles with mucositis. Importantly, a reduction in the next dose of chemotherapy was twice as common after cycles with mucositis than after cycles without mucositis . Thus, mucositis can be a dose-limiting toxicity of cancer chemotherapy with direct effects on patient survival.
The majority of patients receiving radiation therapy for head and neck cancer are unable to continue eating by mouth because of mucositis pain and often receive nutrition through a gastrostomy tube or intravenous line. It has been demonstrated that patients with oral mucositis are significantly more likely to have severe pain and a weight loss of 5% or more . In one study, approximately 16% of patients receiving radiation therapy for head and neck cancer were hospitalized because of mucositis . Further, 11% of the patients receiving radiation therapy for head and neck cancer had unplanned breaks in radiation therapy because of severe mucositis . Thus, oral mucositis is a major dose-limiting toxicity of radiation therapy to the head and neck region.
Clinical significance of oral mucositis
Oral mucositis can be very painful and can significantly affect nutritional intake, mouth care, and quality of life . For patients receiving high-dose chemotherapy before hematopoietic cell transplantation, oral mucositis has been reported to be the single most debilitating complication of transplantation . Infections associated with the oral mucositis lesions can cause life-threatening systemic sepsis during periods of profound immunosuppression . Moderate to severe oral mucositis has been correlated with systemic infection and transplant-related mortality . In patients with hematologic malignancies receiving allogeneic hematopoietic cell transplantation, increased severity of oral mucositis was found to be associated significantly with an increased number of days requiring total parenteral nutrition and parenteral narcotic therapy, increased number of days with fever, incidence of significant infection, increased time in hospital, and increased total inpatient charges .
In patients receiving chemotherapy for solid tumors or lymphoma, the rate of infection during cycles with mucositis was more than twice that during cycles without mucositis and was directly proportional to the severity of mucositis . Infection-related deaths were also more common during cycles with both oral and GI mucositis. In addition, the average duration of hospitalization was significantly longer during chemotherapy cycles with mucositis. Importantly, a reduction in the next dose of chemotherapy was twice as common after cycles with mucositis than after cycles without mucositis . Thus, mucositis can be a dose-limiting toxicity of cancer chemotherapy with direct effects on patient survival.
The majority of patients receiving radiation therapy for head and neck cancer are unable to continue eating by mouth because of mucositis pain and often receive nutrition through a gastrostomy tube or intravenous line. It has been demonstrated that patients with oral mucositis are significantly more likely to have severe pain and a weight loss of 5% or more . In one study, approximately 16% of patients receiving radiation therapy for head and neck cancer were hospitalized because of mucositis . Further, 11% of the patients receiving radiation therapy for head and neck cancer had unplanned breaks in radiation therapy because of severe mucositis . Thus, oral mucositis is a major dose-limiting toxicity of radiation therapy to the head and neck region.
Economic impact of mucositis
Chemotherapy patients who have significant oral mucositis require supportive care measures such as use of total parenteral nutrition, fluid replacement, and prophylaxis against infections. These can add substantially to the total cost of care. For example, in patients receiving chemotherapy for solid tumors or lymphoma, the estimated cost of hospitalization was $3893 per chemotherapy cycle without mucositis, $6277 per cycle with oral mucositis, and $9132 per cycle with both oral and GI mucositis . A single-point increase in peak mucositis scores in hematopoietic cell transplant patients is associated with one additional day of fever, a 2.1-fold increase in risk of significant infection, 2.7 additional days of total parenteral nutrition, 2.6 additional days of injectable narcotic therapy, 2.6 additional days in hospital, and a 3.9-fold increase in 100-day mortality risk, collectively contributing to over $25,000 in additional hospital charges . Radiation-induced oral mucositis also has a significant economic impact because of costs associated with pain management, liquid diet supplements, gastrostomy tube placement or total parenteral nutrition, management of secondary infections, and hospitalizations. In one study of patients receiving radiation therapy for head and neck cancer, oral mucositis was associated with an increase in costs ranging from $1700 to $6000 per patient, depending on the grade of oral mucositis (see the article by Elting elsewhere in this issue).
Pathogenesis of mucositis
Recent studies have indicated that the fundamental mechanisms involved in the pathogenesis of mucositis are much more complex than direct damage to epithelium alone . Mechanisms for radiation-induced and chemotherapy-induced mucositis are believed to be similar. The following five-stage model for the pathogenesis of mucositis is based on the evidence available to date ( Fig. 1 ):
- 1.
Initiation of tissue injury: Radiation and/or chemotherapy induce cellular damage resulting in death of the basal epithelial cells. The generation of reactive oxygen species (free radicals) by radiation or chemotherapy is also believed to exert a role in the initiation of mucosal injury. These small highly reactive molecules are by-products of oxygen metabolism and can cause significant cellular damage.
- 2.
Up-regulation of inflammation via generation of messenger signals: In addition to causing direct cell death, free radicals activate second messengers that transmit signals from receptors on the cellular surface to the inside of the cell. This leads to up-regulation of pro-inflammatory cytokines, tissue injury, and cell death.
- 3.
Signaling and amplification: Up-regulation of pro-inflammatory cytokines such as tumor necrosis factor- alpha (TNF-α), produced mainly by macrophages, causes injury to mucosal cells, and also activates molecular pathways that amplify mucosal injury.
- 4.
Ulceration and inflammation: There is a significant inflammatory cell infiltrate associated with the mucosal ulcerations, based in part on metabolic by-products of the colonizing oral microflora. Production of pro-inflammatory cytokines is also further up-regulated as a result of this secondary infection .
- 5.
Healing: This phase is characterized by epithelial proliferation as well as cellular and tissue differentiation , restoring the integrity of the epithelium.
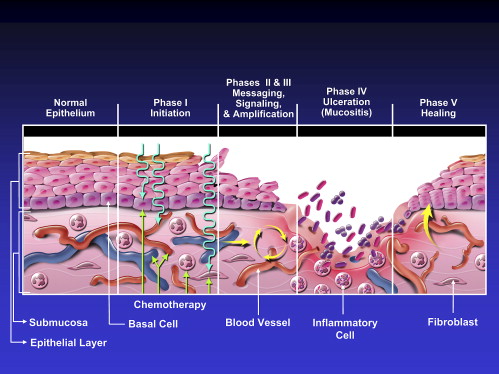
The degree and extent of oral mucositis that develops in any particular patient and site appears to depend on factors such as age, gender, underlying systemic disease, and race as well as tissue-specific factors (eg, epithelial types, local microbial environment, and function). The effects of patient age and gender on the development of oral mucositis are not clear. One study reported increased prevalence of mucositis in children , while other studies reported increased prevalence and/or severity in older patients . Similarly, there is conflicting evidence for the effects of gender on risk for mucositis with some studies reporting increased risk for mucositis in females , and others finding no gender effect .
Interactions of such factors, coupled with underlying genetic influences, are postulated to govern the risk, course, and severity of mucositis . For example, epidermal growth factor (EGF) in luminal secretions may affect the response of intestinal mucosa to chemotherapy although overexpression of EGF in a transgenic mouse model did not reduce intestinal mucositis . Recent studies have indicated that pathways associated with pro-inflammatory molecules including cyclooxygenase-2, nuclear factor-kappa B, and interleukin-6 are up-regulated in oral mucositis. Thus, these may provide potential therapeutic targets for new therapies .
Clinical course of oral mucositis
Oral mucositis initially presents as erythema of the oral mucosa, which then often progresses to erosion and ulceration. The ulcerations are typically covered by a white fibrinous pseudomembrane ( Figs. 2 and 3 ). The lesions typically heal within approximately 2 to 4 weeks after the last dose of stomatotoxic chemotherapy or radiation therapy. In immunosuppressed patients (eg, patients undergoing hematopoietic cell transplantation), resolution of oral mucositis usually coincides with granulocyte recovery; however, this temporal relationship may or may not be causal.
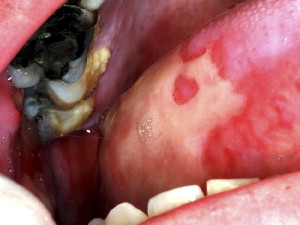
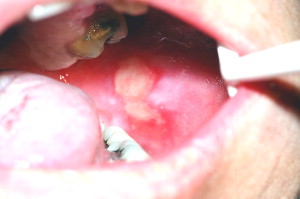
Several factors affect the clinical course of mucositis. In chemotherapy-induced oral mucositis, lesions are usually limited to nonkeratinized surfaces (ie, lateral and ventral tongue, buccal mucosa, and soft palate) . Ulcers typically arise within 2 weeks after initiation of chemotherapy. Selected agents such as antimetabolites and alkylating agents cause a higher incidence and severity of oral mucositis . In radiation-induced oral mucositis, lesions are limited to the tissues in the field of radiation, with nonkeratinized tissues affected more often. The clinical severity is directly proportional to the dose of radiation administered. Most patients who have received more than 5000 cGy to the oral mucosa will develop severe ulcerative oral mucositis .
The clinical course of oral mucositis may sometimes be complicated by local infection, particularly in immunosuppressed patients. Viral infections such as recrudescent herpes simplex virus (HSV) and fungal infections such as candidiasis can sometimes be superimposed on oral mucositis. Although HSV infections do not cause oral mucositis , they can complicate its diagnosis and management.
Measurement of oral mucositis
A wide variety of scales have been used to record the extent and severity of oral mucositis in clinical practice and research. The World Health Organization (WHO) scale is a simple, easy-to-use scale that is suitable for daily use in clinical practice. This scale combines both subjective and objective measures of oral mucositis ( Box 1 ). The National Cancer Institute (NCI) Common Terminology Criteria for Adverse Events (CTCAE) version 3.0 includes separate subjective and objective scales for mucositis ( Box 2 ) . The Oral Mucositis Assessment Scale (OMAS) is an objective scale, suitable for research purposes, that measures erythema and ulceration at nine different sites in the oral cavity. This scale has been validated in a multicenter trial with high interobserver reproducibility and strong correlation of objective mucositis scores with patient symptoms . The Eastern Cooperative Oncology Group (ECOG) common toxicity criteria are also used in oncology trials to document severity of oral mucositis .
-
Grade 0 = No oral mucositis
-
Grade 1 = Erythema and soreness
-
Grade 2 = Ulcers, able to eat solids
-
Grade 3 = Ulcers, requires liquid diet (due to mucositis)
-
Grade 4 = Ulcers, alimentation not possible (due to mucositis)
-
Oral mucositis (clinical examination)
-
Grade 1 = Erythema of the mucosa
-
Grade 2 = Patchy ulcerations or pseudomembranes
-
Grade 3 = Confluent ulcerations or pseudomembranes; bleeding with minor trauma
-
Grade 4 = Tissue necrosis; significant spontaneous bleeding; life-threatening consequences
-
Grade 5 = Death
-
Oral mucositis (functional/symptomatic)
-
Grade 1 = Minimal symptoms, normal diet
-
Grade 2 = Symptomatic but can eat and swallow modified diet
-
Grade 3 = Symptomatic and unable to adequately aliment or hydrate orally
-
Grade 4 = Symptoms associated with life-threatening consequences
-
Grade 5 = Death
Clinical management of oral mucositis
Management of oral mucositis has been largely palliative to date, although targeted therapeutic interventions are now being developed . Based on a comprehensive systematic review of the literature, the Mucositis Study Group of the Multinational Association for Supportive Care in Cancer and the International Society of Oral Oncology (MASCC/ISOO) has developed clinical practice guidelines for the management of mucositis (see the article by Fischer and Epstein elsewhere in this issue). Management of oral mucositis is divided into the following sections: pain control, nutritional support, oral decontamination, palliation of dry mouth, management of oral bleeding, and therapeutic interventions for oral mucositis.
Pain control
The primary symptom of oral mucositis is pain. This pain significantly affects nutritional intake, mouth care, and quality of life. Thus, management of mucositis pain is a primary component of any mucositis management strategy. Many centers use saline mouth rinses, ice chips, and topical mouthrinses containing an anesthetic such as 2% viscous lidocaine. The lidocaine may be mixed with equal volumes of diphenhydramine and a soothing covering agent such as Maalox (Novartis Consumer Health, Inc., Fremont, MI) or Kaopectate (Chattem, Inc., Chattanooga, TN) in equal volumes. Such topical anesthetic agents may provide short-term relief.
A number of other topical mucosal bioadherent agents are also available that are not anesthetics but are postulated to reduce pain by forming a protective coating over ulcerated mucosa. Of these, sucralfate is the most widely studied. The MASCC/ISOO guidelines recommend against the use of sucralfate in radiation-induced oral mucositis due to lack of efficacy. No recommendation has been made for the use of sucralfate in chemotherapy-induced oral mucositis due to lack of consistent results . In addition to the use of topical agents, most patients with severe mucositis require systemic analgesics, often including opioids, for satisfactory pain relief. The MASCC/ISOO guidelines recommend patient-controlled analgesia with morphine for patients undergoing hematopoietic cell transplantation .
Nutritional support
Nutritional intake can be severely compromised by the pain associated with severe oral mucositis. In addition, taste changes can also occur secondary to chemotherapy and/or radiation therapy . It is essential that nutritional intake and weight be monitored by a dietician or other professional working together with family caregivers. A soft diet and liquid diet supplements are more easily tolerated than a normal diet when oral mucositis is present. In patients expected to develop severe mucositis, a gastrostomy tube is sometimes placed prophylactically, although this varies considerably from center to center. In patients undergoing hematopoietic cell transplantation, total parenteral nutrition is usually given via an indwelling catheter such as a Hickman line.
Oral decontamination
Oral decontamination may result in significant positive outcomes in this population. First, it has been hypothesized that microbial colonization of oral mucositis lesions exacerbates the severity of oral mucositis and, therefore, decontamination may help to reduce mucositis. Indeed, multiple studies have demonstrated that maintenance of good oral hygiene can reduce the severity of oral mucositis . Patients who have undergone hematopoietic cell transplantation and who develop oral mucositis also have been found to be three times more likely to develop bacteremias resulting in increased length of hospital stays as compared with patients without mucositis . Therefore, oral decontamination may reduce mucositis that, in turn, may reduce bacteremia. Furthermore, oral decontamination can reduce infection of the oral cavity by opportunistic pathogens . Therefore, a second function of oral decontamination can be to reduce the risk of systemic sepsis from resident oral and/or opportunistic pathogens. This is especially true in patients who are immunosuppressed as a result of chemotherapy. The risk of systemic sepsis from oral mucositis has not been well studied although one study found that an intensive oral care protocol decreased risk of oral mucositis but not the percentage of patients with a documented septicemia . Patients receiving radiation therapy alone are less likely to develop sepsis of oral origin.
The MASCC/ISOO guidelines recommend use of a standardized oral care protocol including brushing with a soft toothbrush, flossing, and the use of nonmedicated rinses (eg, saline or sodium bicarbonate rinses). Patients and caregivers should be educated regarding the importance of effective oral hygiene . Alcohol-containing chlorhexidine mouthrinse may be difficult for patients to tolerate during clinical oral ulceration; thus, formulations without alcohol are used at some centers. Multiple studies have examined the role of chlorhexidine mouthrinse in oral mucositis but have not demonstrated significant efficacy in reducing severity of mucositis . Therefore, the MASCC/ISOO guidelines recommend against the use of chlorhexidine mouthrinse for prevention or treatment of oral mucositis.
Nystatin rinse has not been found to be effective in reducing the severity of chemotherapy-induced mucositis . On the other hand, a recent study indicated that systemic fluconazole versus no treatment significantly and dramatically reduced both candidal carriage and incidence of severe mucositis induced by radiation therapy (15% versus 45%) in patients with head and neck cancer . The MASCC/ISOO guidelines recommend against the routine use of antimicrobial lozenges or of acyclovir and its analogs to prevent oral mucositis . However, drugs such as acyclovir and valacyclovir have a well-established role in prophylaxis and treatment of lesions caused by HSV in this patient population (see the article by Lerman and colleagues elsewhere in this issue).
Palliation of dry mouth
Patients undergoing cancer therapy often develop transient or permanent xerostomia (subjective symptom of dryness) and hyposalivation (objective reduction in salivary flow). Hyposalivation can further aggravate inflamed tissues, increase risk for local infection, and make mastication difficult. Many patients also complain of a thickening of salivary secretions because of a decrease in the serous component of saliva. The following measures can be taken for palliation of a dry mouth:
- •
Sip water as needed to alleviate mouth dryness. Several supportive products including artificial saliva are available.
- •
Rinse with a solution of half a teaspoon of baking soda half a in 1 cup warm water several times a day to clean and lubricate the oral tissues and to buffer the oral environment.
- •
Chew sugarless gum to stimulate salivary flow.
- •
Use cholinergic agents as necessary.
Please also see the article by Fischer and Epstein elsewhere in this issue.
Management of bleeding
In patients who are thrombocytopenic as a result of high-dose chemotherapy (eg, hematopoietic cell transplant recipients), bleeding may occur from the ulcerations of oral mucositis. Local intraoral bleeding can usually be controlled with the use of topical hemostatic agents such as fibrin glue or gelatin sponge . Patients whose platelet counts fall below 20,000/ml may receive platelet transfusion because of the risk for spontaneous internal bleeding, which may have grave consequences especially in the central nervous system.
Therapeutic interventions
Several agents have been tested to reduce the severity of, or prevent, mucositis. These different classes of agents are discussed briefly in the context of the MASCC/ISOO guidelines where applicable.
Cryotherapy
It has been hypothesized that topical administration of ice chips to the oral cavity during administration of chemotherapy results in decreased delivery of the chemotherapeutic agent to the oral mucosa. This effect is presumably mediated through local vasoconstriction and reduced blood flow. Several studies have demonstrated that cryotherapy reduces the severity of oral mucositis in patients receiving bolus doses of chemotherapeutic agents . The MASCC/ISOO guidelines recommend the use of cryotherapy to reduce oral mucositis in patients receiving bolus doses of 5-fluorouracil, melphalan, and edatrexate . Ice chips are placed in the mouth, beginning 5 minutes before administration of chemotherapy and replenished as needed for up to 30 minutes. Cryotherapy is only useful for short bolus chemotherapeutic infusions, may not be well tolerated in some subjects and does not have a role in radiation-induced oral mucositis.
Growth factors
Reduction in the proliferative capacity of oral epithelial cells is thought to play a role in the pathogenesis of mucositis. Therefore, various growth factors that can increase epithelial cell proliferation have been studied for the management of oral mucositis. Recent evidence shows that intravenous (IV) recombinant human keratinocyte growth factor-1 (Palifermin, Amgen, Thousand Oaks, CA) significantly reduced incidence of WHO grades 3 and 4 oral mucositis in patients with hematologic malignancies (eg, lymphoma and multiple myeloma) receiving high-dose chemotherapy and total body irradiation before autologous hematopoietic cell transplantation . On that basis, the MASCC/ISOO guidelines recommend the use of recombinant human keratinocyte growth factor-1 in this specific population . Palifermin has also been approved by the US Food and Drug Administration (FDA) for patients with hematologic malignancies receiving myelotoxic therapies requiring hematopoietic cell support. Interestingly, a related compound, human keratinocyte growth factor-2 (Repifermin, Human Genome Sciences), was found to be ineffective in reducing the percentage of subjects who experienced severe mucositis . Intravenous human fibroblast growth factor-20 (Velafermin, Curagen Corp., Branford, CT) is currently in clinical development for reduction of mucositis secondary to high-dose chemotherapy in autologous hematopoietic cell transplant patients .
The safety of this class of growth factors has not been established in patients with nonhematologic malignancies. There is a theoretical concern that these growth factors may promote growth of tumor cells, which may have receptors for the respective growth factor. However, one recent study found no significant difference in survival between subjects with colorectal cancer receiving palifermin or placebo at a median follow-up duration of 14.5 months . Further studies are ongoing to confirm the safety of epithelial growth factors in the solid tumor setting including patients receiving radiation therapy for head and neck cancer.
Anti-inflammatory agents
Benzydamine hydrochloride is a nonsteroidal anti-inflammatory drug that inhibits proinflammatory cytokines including TNF-α. In one Phase III trial, benzydamine hydrochloride mouthrinse reduced the severity of mucositis in patients with head and neck cancer undergoing radiation therapy of cumulative doses up to 50-Gy radiation therapy . Based on this and previous studies, the MASCC/ISOO guidelines recommended use of this agent in patients receiving moderate-dose radiation therapy . However, this agent has not received approval for this use from the FDA; furthermore, most patients with head and neck cancer receive well over 50-Gy radiation therapy with concomitant chemotherapy. A more recent Phase III trial of this agent in radiation-induced oral mucositis in patients with head and neck cancer was halted based on negative results of an interim analysis.
Saforis (MGI Pharma) is a proprietary oral suspension of L-glutamine that enhances the uptake of this amino acid into epithelial cells. Glutamine may reduce mucosal injury by reducing the production of proinflammatory cytokines and cytokine-related apoptosis and may promote healing by increasing fibroblast and collagen synthesis . In a Phase III study, this topical agent reduced the incidence of clinically significant chemotherapy-induced oral mucositis as compared with placebo . By comparison, the MASCC/ISOO guidelines recommend that systemically administered glutamine not be used for the prevention of GI mucositis because of lack of efficacy .
Antioxidants
Amifostine (Ethylol, MedImmune, Gaithersburg, MD) is thought to act as a scavenger for harmful reactive oxygen species that are known to potentiate mucositis . However, because of insufficient evidence of benefit, a MASCC/ISOO guideline could not be established regarding the use of this agent in oral mucositis in chemotherapy or radiation therapy patients. The use of amifostine was recommended for the prevention of esophagitis in patients receiving chemo-radiation for non–small-cell lung cancer . RK-0202 (RxKinetix) consists of the antioxidant N-acetylcysteine in a proprietary matrix for topical application in the oral cavity. In a placebo-controlled phase II trial in patients with head and neck cancer, this agent significantly reduced the incidence of severe oral mucositis up to doses of 50-Gy radiation therapy .
Low-level laser therapy
Multiple studies have indicated that low-level laser therapy can reduce the severity of chemotherapy and radiation-induced oral mucositis , although the mechanism of such an effect is not understood. It has been speculated that low-level laser therapy may reduce levels of reactive oxygen species and/or proinflammatory cytokines that contribute to the pathogenesis of mucositis. Studies are difficult to compare because of varying laser types and parameters (such as wavelength). Nevertheless, based on the encouraging results to date, the MASCC/ISOO guidelines suggest the use of low-level laser therapy in chemotherapy-induced oral mucositis at centers able to support the necessary technology and training .
Stay updated, free dental videos. Join our Telegram channel

VIDEdental - Online dental courses


