In 2003 and 2004, the first reports of patients who developed necrosis of the jawbones while taking biophosphonates appeared in literature; most patients were on this drug for treatment of cancer and some osteoporosis. Since then, more than 500 cases have been identified and the number of these cases continues to grow. This article reviews the action of bisphosphonates, the condition called bisphosphonate-associated osteonecrosis of the jaws, strategies to minimize occurrence, and treatment of this condition.
In 2003 and 2004, the first reports of patients who developed necrosis of the jawbones while taking bisphosphonates appeared in the literature; most patients were on this drug for treatment of cancer and some for osteoporosis . Since then, more than 500 cases have been identified and the number of these cases continues to grow. This article reviews the action of bisphosphonates, the condition called bisphosphonate-associated osteonecrosis of the jaws , strategies to minimize occurrence, and treatment of this condition.
Nomenclature
A universally accepted term for this new condition has not been established, which has caused some degree of confusion. This complication has been referred to in the literature as BRONJ (bisphosphonate-related osteonecrosis of the jaw), BRON (bisphosphonate-related osteonecrosis), BON (bisphosphonate osteonecrosis), BAONJ (bisphosphonate-associated osteonecrosis of the jaw), and simply ONJ (osteonecrosis of the jaw). Based on the clear association between bisphosphonate therapy and jaw necrosis that has been established in numerous retrospective studies, the American Association of Oral and Maxillofacial Surgeons (AAOMS) has decided to adopt the term BRONJ for this entity , and this term will be used throughout this article.
Action of bisphosphonates
The actions and pharmacology of bisphosphonates have been well reviewed and only a summary is offered here . Bisphosphonates are analogs of inorganic pyrophosphates. Pyrophosphates are well-known in dentistry, because they are a component of tartar-control toothpaste and inhibit calcium precipitation . Unlike pyrophosphates, bisphosphonates contain a carbon rather than oxygen molecule. This P-C-P structure allows the molecule to bind to the hydroxyapatite crystals with high affinity, while the presence of nitrogen in one of the side chains confers markedly increased potency to the drug ( Fig. 1 ). The early forms of bisphosphonate, such as etidronate and clodronate, do not contain nitrogen and therefore are less potent; they are available only in the oral preparation. The later forms of bisphosphonates contain nitrogen and some are available as intravenous preparations, making them much more bioavailable ( Table 1 ). Pamidronate and alendronate contain nitrogen in an alkyl chain and are 10 to 100 times more potent than etidronate and clodronate. The most potent of the bisphosphonates (such as zoledronic acid and risedronate) contain nitrogen within a heterocyclic ring. The half-life of bisphosphonates is approximately 10 years, and therefore prolonged use of this drug causes substantial drug accumulation within the skeleton. The drug is tightly bound to the apatite crystals until it is released during osteoclastic-mediated bone resorption.
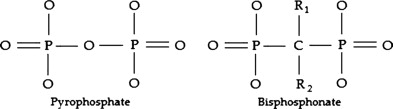
| Generic name | Brand name/delivery | Manufacturer | Indication | RP |
|---|---|---|---|---|
| Clodronate | Bonefos, PO | Hoffman/LaRoche | Not available in the United States | 50 |
| Pamidronate | Aredia, IV | Novartis | Bone cancer | 100 |
| Alendronate sodium 1000 | Fosamax, PO | Merck | Osteoporosis | 500–1000 |
| Risedronate | Actonel, PO | Proctor & Gamble | Osteoporosis | 5000 |
| Ibandronate | Boniva, PO/IV | Roche | Osteoporosis | 5000–10,000 |
| Zoledronic acid | Zometa, IV | Novartis | Bone cancer a | >10,000 |
a Intravenous zoledronic acid also has been approved for the treatment of osteoporosis (4–5 mg dose, once per year).
Antiresorptive activity
One of the most important and powerful effects of bisphosphonates is inhibition of osteoclastic activity, and herein lies one of its most important applications in clinical practice, both for managing osteoporosis and cancers in the skeletal system. When bone resorption occurs, bisphosphonates are released from the hydroxyapatite crystal and are taken up by osteoclasts. Metabolites of non-nitrogen containing bisphosphonates (such as etidronate and clodronate) are cytotoxic to the osteoclasts and lead to their death. Nitrogen-containing bisphosphonates, however, act by way of the mevalonate pathway (for cholesterol synthesis), inhibiting protein prenylation, a process essential for normal functioning of vital intracellular proteins, ultimately leading to osteoclast apoptosis (programmed cell death) . Bisphosphonates also inhibit differentiation of osteoclasts and stimulates osteoblasts to produce osteoclast-inhibiting factor . Therefore, the net result is reduced numbers of osteoclasts and reduced bone resorption. Because bone resorption is coupled to osteoblastic bone formation for remodeling, bone turnover (ie, resorption and deposition) becomes severely suppressed. However, the bone continues to mineralize and could become brittle and less elastic. In one case report, bisphosphonate taken at high doses led to an osteopetrotic-like state .
Tumoricidal activity
Many studies have shown that nitrogen-containing bisphosphonates also reduce the activity of cancer cells and control metastases . This process may be related to inhibition of protein prenylation leading to disruption of intracellular activity within the cancer cells . However, the alteration of the microenvironment itself, caused by reduced bone resorption alone, could also account for control of metastases . Bisphosphonates also reduce adhesion, invasion, and viability of cancer cells and may activate gamma delta T cells, which have tumoricidal activity .
Antiangiogenic activity
In vitro, zoledronic acid inhibits angiogenesis mediated through basic fibroblast growth factor and may induce apoptosis of endothelial cells . Antiangiogenic activity may also occur through lowering circulating levels of vascular endothelial growth factor and platelet-derived growth factor, both of which are proangiogenic .
Clinical applications
Bisphosphonates are widely used in the management of four main conditions: osteoporosis, Paget’s disease of bone, multiple myeloma, and cancers that have metastasized to the bones. This article focuses only on the oncologic aspects of bisphosphonate use.
Multiple myeloma is a malignancy of plasma cells that primarily affects the skeletal system. Patients develop multiple lytic bone lesions containing malignant plasma cells, and as a result may develop skeletal-related events (SRE), such as bone pain, fractures, necessity for surgery and radiation, and hypercalcemia. Patients also develop hypergammaglobulinemia from secretory activity of these cells, which can lead to renal failure. Clinical trials show that bisphosphonates significantly reduce these SREs, and pamidronate and zoledronic acid are widely used for treating multiple myeloma . Other agents for treating this disease include thalidomide, glucocorticoids such as dexamethasone, and proteasome inhibitors such as bortezomib, usually in combination . These agents also have antiangiogenic properties.
Similar SREs are noted in patients who have metastatic cancers (with metastatic breast and prostate cancers being the most common), and bisphosphonates also have been shown to significantly reduce SREs in these patients .
Early reports of bisphosphonate-associated osteonecrosis of the jaws
The earliest reports of bona fide cases of BRONJ occurred in 2003 , and have been followed by many more case reports and large case series . All cases have occurred in the maxilla or mandible except for a single case that occurred after ear surgery .
Etiopathogenesis
The exact mechanism for the development of BRONJ is unclear. The current hypothesis focuses on severe suppression of bone turnover, coupled with the unique conditions affecting the jaws and not other bones. These conditions include the following: (1) the jaw bones are separated from the oral environment from a very thin mucosa (only several mm thick for the most part) and this barrier is readily breached even with simple physiologic activities such as mastication, (2) the oral cavity is filled with bacteria and the jaws are often involved in infection through either the periodontal ligament or the pulp, (3) dentoalveolar surgery is a common procedure (eg, extractions, periodontal surgeries, apicoectomies) in which bone is exposed to a bacteria-rich environment, and (4) the rate of turnover of the jawbones is higher than that for the long bones .
One possible mechanism of action is that after a sufficient concentration of bisphosphonates has accumulated, the jawbones become hypodynamic and turn over at a low rate. When infection or surgery requires increased bone turnover for healing, this does not occur. How the bone actually becomes necrotic and exposed is unclear at this time. However, there is a condition that mimics BRONJ that occurs spontaneously, involves only a few millimeters of necrotic bone that heals without incident, and occurs in healthy adults. This benign sequestration of the lingual plate is a well-recognized phenomenon and its putative mechanism of action is trauma to the lingual periosteum, loss of vascular supply of bone supplied by that area of periosteum, death of bone with sequestration, and healing . The lingual plate/mylohyoid area is also a common site for spontaneously occurring BRONJ.
Risk factors for bisphosphonate-associated osteonecrosis of the jaws
Not all patients who use bisphosphonates develop BRONJ. The factors that affect its development are:
- 1)
The use of nitrogen-containing bisphosphonates, particularly zoledronic acid . Zoledronic acid used alone produced 9.5-fold and 4.5-fold greater risk compared with pamidronate used alone or with zoledronic acid, respectively . However, BRONJ has been seen in a patient who had myeloma who took only oral clodronate, a first-generation non–nitrogen-containing bisphosphonate .
- 2)
The cumulative dose of bisphosphonates. This factor also indirectly relates to the patient’s underlying condition, because patients who have cancer receive much higher doses of bisphosphonates than those who have osteoporosis. For example, a patient who has osteoporosis who requires intravenous bisphosphonate therapy receives 4 to 5 mg of zoledronic acid per year compared with a patient who has myeloma who receives 4 mg of zoledronic acid every 4 weeks. The median time of exposure to zoledronic acid with development of BRONJ ranged from 9 to 30 months and is significantly shorter compared with other bisphosphonate regimens . However, the exposure can be as little as 3 months. The risk for developing BRONJ increases over time, with a cumulative hazard of 1% within the first year of treatment with zoledronic acid, and 15% at 4 years .
- 3)
Dentoalveolar surgery. Approximately 60% of all cases are associated with either tooth extraction or other surgery (eg, periodontal and apical surgery and implant placement) . However, tooth extractions are the most common inciting factor . One study showed that the prevalence of BRONJ in patients who had cancer who did not undergo extractions was 1% compared with 7% to 9% in those who did . BRONJ has been reported in patients after surgical endodontic procedures .
- 4)
Trauma. Many cases occur on the lingual mandible (where the mucosa is especially fragile) and on tori .
Other factors that may contribute to the development of ONJ, but for which the evidence is less robust, include dental infections. By the time BRONJ develops, it is difficult to determine if the infection caused BRONJ or was merely a consequence of BRONJ.
Other factors, such as route of administration (intravenous preparations are more frequently associated with BRONJ) and age, are indirectly related to the factors noted earlier. For example, patients who have cancer affecting the skeleton (usually older patients) typically are exposed to the more potent bisphosphonates, such as zoledronic acid and pamidronate, both of which are only available as intravenous preparations, and are used more frequently.
Whether the antiangiogenic activity of bisphosphonates plays an important and direct role in BRONJ is unclear. Other comorbidities have not been entirely elucidated, but include concomitant use of antiangiogenic agents and vascular compromise. Thalidomide, which has antiangiogenic properties, has been shown to increase the risk 2.4-fold , although this has been disputed . One study showed that patients who had BRONJ were more likely to have associated diabetes mellitus, although most patients already had diabetes when their cancer was diagnosed . Another study showed that patients who developed BRONJ had hyperparathyroidism and mild hypocalcemia compared with controls .
Prevalence
The prevalence varies from 4% to 7% within any one center. Most of the studies were performed through retrospective chart reviews or telephone interviews .
Although most patients who developed BRONJ underwent dental extractions, the number of patients who had extractions or other dento-alveolar surgery and did not develop BRONJ is unclear.
Diagnosis and clinical presentation of bisphosphonate-associated osteonecrosis of the jaws
Standardization of diagnostic criteria for this new clinical entity is important to facilitate future clinical and epidemiologic research and help distinguish BRONJ from other intraoral osteonecrotic conditions exhibiting delayed healing. The AAOMS established a working definition for BRONJ that is fairly concise and specific . Patients may be considered to have BRONJ if they have all of the following characteristics: (1) current or previous treatment with a bisphosphonate, (2) exposed, necrotic bone in the maxillofacial region that has persisted for more than 8 weeks, and (3) no history of radiation therapy to the jaws. Patients who are considered at risk according to the AAOMS criteria have no evidence of exposed or necrotic bone but have been exposed to either intravenous or oral nitrogen-containing bisphosphonates for long periods.
The American Society for Bone and Mineral Research (ASBMR) has also made recommendations for a provisional case definition for confirmed and suspected cases of bisphosphonate associated osteonecrosis . A confirmed case is identical to the AAOMS definition for BRONJ. A suspected case fulfills all the criteria of a confirmed case, except that the necrotic bone has been present for less than 8 weeks. Suspected cases should be followed up to determine if they eventually meet the criteria of a confirmed case.
Diagnosis of BRONJ is primarily based on patient history and clinical examination. Patients present with exposed, necrotic bone varying in size from a few millimeters to several centimeters; only 60% of cases report pain, and some cases may remain asymptomatic for weeks, months, or years . These lesions frequently become symptomatic when surrounding tissues become inflamed or an infection develops. Signs and symptoms that may occur before the development of clinically detectable BRONJ include pain, tooth mobility, mucosal swelling, erythema, ulceration, and the development of sinus tracts. These somewhat nonspecific signs and symptoms are similar to those of banal odontogenic infections. Some patients may also present with complaints of altered sensation or paresthesia in the affected area as the neurovascular bundle becomes affected by inflammation or infection around the necrotic bone. Chronic maxillary sinusitis secondary to BRONJ with or without an oral-antral fistula may be the presenting symptom in patients who have maxillary bone involvement .
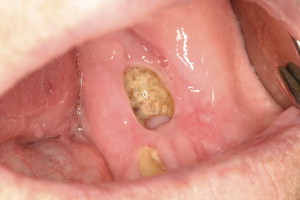
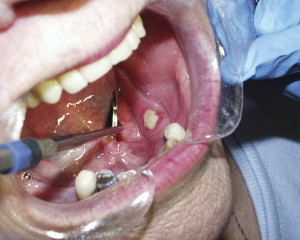
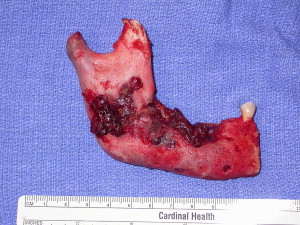
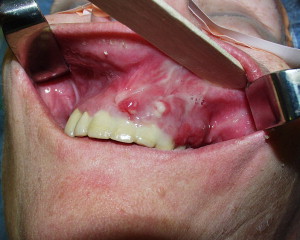
Lesions have been observed more commonly in the mandible than the maxilla (2:1 ratio) , and more commonly in areas with thin mucosa overlying bony prominences, such as tori, bony exostoses, and the mylohyoid ridge . Most cases occur at the site of prior dentoalveolar surgery ( Fig. 2 ). However, exposed bone has also been reported in patients who have no history of trauma or in edentulous regions of the jaw ( Fig. 3 ). The size of the affected area can be variable, and ranges from a nonhealing extraction site to exposure and necrosis of the entire jaw ( Fig. 4 ). The area of exposed bone may be surrounded by inflamed erythematous soft tissue. When infected, purulent discharge may be seen at the site of the exposed bone and intra- and extraoral fistulae ( Fig. 5 ). The AAOMS has proposed a clinical staging system based on the presence of symptoms and extent of disease ( Table 2 ) .
| Stage | Clinical presentation | Management |
|---|---|---|
| At risk | No exposed bone | Patient education |
| 1 | Asymptomatic exposed bone with little soft tissue inflammation | Patient education; antibacterial rinses a ; careful follow-up b |
| 2 | Exposed bone with pain, and usually with associated surrounding soft tissue inflammation or infection | Patient education; antibacterial rinses a ; antibiotics c ; superficial debridement of bone to dislodge loose fragments and smooth rough contours; careful follow-up b |
| 3 | Exposed bone with pain and usually with associated soft tissue inflammation or infection; may see osteolysis extending to the inferior border of mandible or pathologic fracture; may see extraoral fistula | Patient education; antibacterial rinses a ; antibiotics c ; palliative surgery; careful follow-up b |
Stay updated, free dental videos. Join our Telegram channel

VIDEdental - Online dental courses


