Chapter 69 Laryngomalacia
1 INTRODUCTION
Laryngomalacia is the term most widely used to describe the ‘inward collapse of supraglottic structures during inspiration’, as originated by Jackson and Jackson in 1942. The inward collapse of the epiglottis or arytenoids results in stridor that is typically inspiratory and high-pitched. Laryngomalacia is often cited as the most common congenital laryngeal anomaly and most frequent cause of stridor in infants. The stridor is often present at birth, and is usually noticed by 2 weeks of age. Laryngomalacia is usually positionally dependent and is worse at times of increased work of breathing. Older children and adults may be diagnosed with laryngomalacia occurring particularly during exercise. This brings into question the idea that laryngomalacia is only a congenital problem.
2 PATIENT SELECTION
The stridor of laryngomalacia is well described and the diagnosis can be easily made with fiberoptic laryngoscopy, but the pathogenesis is still unknown. The first proposed mechanism of pathogenesis was floppiness of the airway secondary to infantile cartilage abnormalities, but histological study did not support this.1 Others suggest that there is poor neuromuscular control with relative hypotonia of the supraglottic dilator muscles.1 The cause of the hypotonia is presumed to be central in nature but the pathophysiology is not understood.
Research of decades ago and again more recently has looked at pathways of chemosensory reflexes in the larynx causing apnea as a means of understanding laryngeal function.2,3 Others looking at the larynx more directly found that the laryngeal adductor reflex elicited by a calibrated puff of air to the larynx could be used as a diagnostic tool correlated with airway patency control, swallowing and apnea.4,5 Recent work by Thompson et al.6 found that children with laryngomalacia also have altered laryngeal sensation. The larynx of a child with laryngomalacia requires a more intense puff of air to activate the laryngeal adductor reflux than children without laryngomalacia. Thompson also found that those with severe laryngomalacia (children requiring surgical management) have a more elevated reflex level than children with mild to moderate laryngomalacia. The children with severe laryngomalacia were also relatively hypoxic with a mean SaO2 of 88.6% vs. 98–99% in children with moderate or mild laryngomalacia.6 The cause of the altered laryngeal sensation is currently under investigation and should shed light on the etiology of laryngomalacia.
There is a subset of children with laryngomalacia who have additional diagnoses7,8 but laryngomalacia is usually isolated and children with isolated laryngomalacia are developmentally normal. Many studies cite reflux disease as being at least a comorbid factor in explaining the symptoms of laryngomalacia.9–11 It is not clear whether the reflux is the primary problem or if reflux occurs secondary to the changes in intrathoracic pressure with the increased work of breathing that occurs with more significant laryngomalacia. A third option is to consider reflux as either primary or secondary.
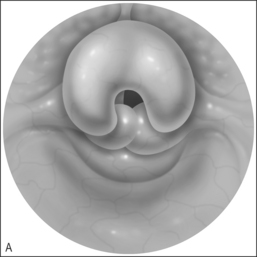
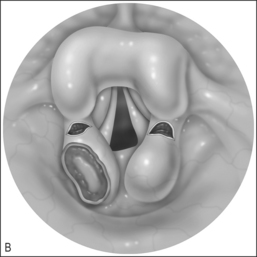
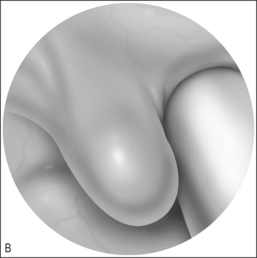
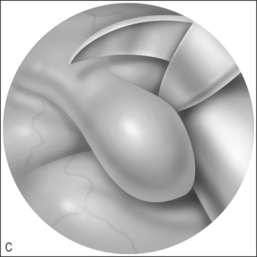
To that end, it may be helpful to consider the diagnosis of laryngomalacia as having two subsets: primary laryngomalacia (Fig. 69.1&), in which there is actual anatomic variation such as the shortening of the aryepiglottic folds12 or a more pronounced omega-shaped epiglottis, and secondary laryngomalacia (Fig. 69.2&) in which reflux disease is the inciting factor. In primary laryngomalacia, the anatomic anomaly narrows the laryngeal inlet. Venturi principles of airflow then describe the resulting ‘floppy airway’8 such as described in obstructive sleep apnea when the soft palate and tongue base are sucked against the posterior pharyngeal wall as a result of tonsils and/or adenoids narrowing the airway. Primary laryngomalacia may result in enough airway distress to induce secondary reflux which then exacerbates the edema and swelling of the tissues that are being sucked into the airway with each breath as seen on fiberoptic evaluation. Secondary laryngomalacia then is when the airway is anatomically normal to begin with but in the child with reflux disease, the airway becomes edematous which begins the process of narrowing and the resulting airway collapse with inspiration.
2.1 PREOPERATIVE EVALUATION
The preoperative evaluation of the patient with stridor begins with a history that includes how well the patient is eating, growing and developing. This gives a sense as to how significant the stridor is and what management options may be necessary. The diagnosis of laryngomalacia is made when viewing the larynx during inspiration and expiration and correlating the noise heard with infolding of the supraglottic structures. It is sometimes helpful to perform laryngoscopy with the patient in both sitting and lying positions. The evaluation of laryngeal dynamics is most directly made in the awake patient as general anesthesia may affect the dynamics of the airway.13 The size of the flexible laryngoscope may also influence the airway dynamics by obstructing the nasal cavity and thus increasing the force for inspiration so the smallest scope that affords good visualization should be chosen for each patient. Another consideration is the positioning of the infant as there can be differences in the airway dynamics if the child is in a sitting or lying position.14 Laryngoscopy in both positions may be considered particularly if what is seen in one position is not what was expected.
When performing laryngoscopy, the surgeon is evaluating the airway for edema, erythema, anatomic structure and movement. Rotation of the epiglottis posteriorly, the arytenoids anteriorly and areas of inward collapse of the supraglottic structure are identified. In addition to noting the static endpoints of inspiration and expiration, evaluation should include where the larynx is positioned in the airway and if the larynx rotates as a whole up toward the tongue base into a protective position away from the esophagus. Shaker described the esophagoglottal closure reflex that occurs when the airway is exposed to reflux.15 This reflex may account for the neuromuscular abnormalities and inco-ordinate swallowing seen in the infant with laryngomalacia. If the airway is sufficiently swollen, it may be impossible to determine if there is underlying anatomic anomaly or if the obstruction is secondary only to edema.
A medical trial with a proton pump inhibitor for mild to moderate airway erythema and edema in the child who is not growing well or having feeding difficulties may result in improvement or resolution of the stridor.10,16 If the child is then able to consistently feed and gain weight, surgery may be avoided, particularly for secondary laryngomalacia. For severe airway obstruction a burst of steroid for 2–5 days is often helpful to relieve acute symptoms and allow more precise evaluation of the underlying anatomy upon repeating the laryngoscopy 1–2 weeks later. If the child presents in airway distress before 1 month of age medical therapy is unlikely to succeed and supraglottoplasty is offered. Supraglottoplasty is the term used to describe the division or removal of any supraglottic tissue for the relief of airway obstruction in the patient with laryngomalacia.13 Approximately 10% of infants with laryngomalacia require surgical intervention. Infants requiring surgery are those with significant primary laryngomalacia and those with secondary laryngomalacia in whom reflux therapy has been inadequate to relieve airway obstruction or feeding difficulties.
Debate exists concerning the need for the evaluation of synchronous airway lesions with a full microlaryngo-scopy and bronchoscopy in every child with laryngomalacia. It has been shown that the majority of secondary airway problems are found in patients who have progressive symptoms unresponsive to medical therapy or observation. Microlaryngoscopy and bronchoscopy should be performed on any child being taken to the operating room for possible supraglottoplasty, but may not be necessary for every child with laryngomalacia.17
3 SURGICAL PROCEDURE
Topical lidocaine without epinephrine is applied to the supraglottic and laryngeal mucosa as a mucosal anesthetic and to reduce reflex laryngeal adduction induced by laryngeal stimulation. Microlaryngoscopy/bronchoscopy is performed to elucidate the anatomic areas of concern in each patient with laryngomalacia and to rule out any additional airway anomalies. With adequate control of active reflux disease, the patient who remains symptomatic usually has a specific anatomic anomaly such as shortened AE folds. Based on the areas of anatomic narrowing found on preoperative evaluation and the microlaryngoscopy and bronchoscopy performed at the beginning of the procedure, the surgical plan is made for division of AE folds only or also for removal of unilateral inward collapsing mucosa.18,19 Bilateral mucosal excision may result in supraglottic stenosis.20 While this is a rare complication, stenosis of the supraglottic structure is a very difficult complication to treat. Often a tracheostomy is required, at least temporarily. Unilateral supraglottoplasty is highly successful and avoids the risk of circumferential stenosis. The opposite side may require supraglottoplasty at a future date 15%19 to 17%18 of the time. Bilateral supraglottoplasty also carries a reoperation rate of 5.1%.19 Therefore, unilateral supraglottoplasty is recommended.
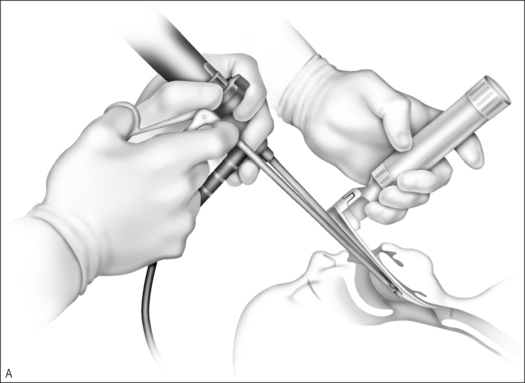
Once the surgical plan has been made, the larynx is then exposed. Options for laryngeal exposure include suspension laryngoscopy with a laryngoscope holder and a microscope for visualization or hand-held suspension laryngoscopy and the endoscope for visualization. Suitable laryngoscopes for traditional suspension include the Cherry Jako, Jako-Cherry, Benjamin, Lindholm or Weerda operating laryngoscopes. The latter laryngoscopes provide superior binocular vision and a broad inlet to facilitate instrument manipulation.17 Hand-held laryngoscopy is accomplished using the anesthesiology intubating laryngoscope with a straight blade. A Wisconsin or Wis-Hipple blade is ideal as the larger posterior opening allows easier access for the endoscope and instrumentation needed to perform supraglottoplasty. The laryngoscope tip may be placed in the vallecula or against the laryngeal surface of the epiglottis depending on the portion of the larynx needing exposure and manipulation. Tissue can be divided or resected using microlaryngeal instruments, the microdebrider,21 or the carbon dioxide laser.
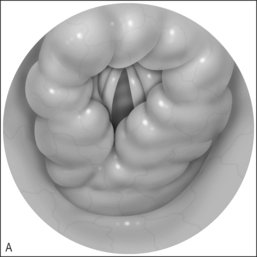
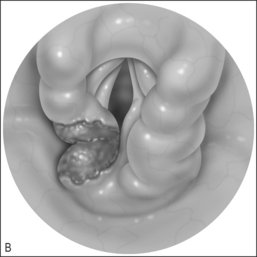
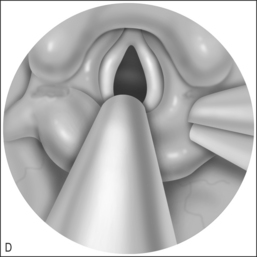
Consistent success has been experienced using the Wisconsin anesthesia blade in the valleculae to expose the larynx. Typically only the epiglottic tip and the posterior edge of the cricoid are seen. The 4 mm Hopkins rod telescope can maneuver between the epiglottis and the arytenoids. If the patient is breathing spontaneously, bilateral respiratory movement of the vocal folds can rule out vocal cord paresis as this may be the first good look at the vocal folds. Flexible laryngoscopy preoperatively may see only the arytenoids moving and no view of the true vocal folds as the epiglottis is tilted posteriorly covering the folds. Once microlaryngo-scopy and bronschoscopy are complete and any additional airway lesions or malacia are noted, the patient is intubated. It is not uncommon that a half size smaller than expected endotracheal tube is all that will fit through the constricted supraglottic opening until the aryepiglottic folds have been released. Once the aryepiglottic folds are released, there is a large air leak around the small tube. In severe aryepiglottic shortening, the patient may only be able to be intubated by inverting the epiglottis into the trachea. The epiglottis is then teased out of the airway using the endoscope at the epiglottic edge or by pressing gently on the pediole of the epiglottis. With the assistant then holding the Wisconsin blade (Fig. 69.3) the aryepiglottic fold is divided using a microlaryngeal scissor, first on one side and then, after repositioning around the endotracheal tube, the other. The endotracheal tube can be used to lever open the narrowed laryngeal inlet by using the endoscope and pushing down on the endotracheal tube to expose the aryepiglottic fold for division. The endoscope is used for the dual purpose of exposure of the ary-epiglottic fold and the visualization of the fold to be divided. The fold is cut until the larynx springs open. Typically this is just after a small blood vessel is encountered. Bleeding is controlled with oxymetazoline on a 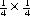 inch surgical sponge. At the beginning of the procedure the arytenoids and the epiglottis are parallel. After dividing the aryepiglottic folds, the epiglottis and arytenoids are nearly perpendicular. The patient is then extubated at the end of the procedure if possible. It is rare that mucosa and submucosal tissue need to be removed from the arytenoids if good preoperative medical control of the edema is accomplished with proton pump inhibitors and/or steroids. Mucosal resection, only if required as determined in the preoperative evaluation, is accomplished using cup forceps and scissors (Fig. 69.4&).
inch surgical sponge. At the beginning of the procedure the arytenoids and the epiglottis are parallel. After dividing the aryepiglottic folds, the epiglottis and arytenoids are nearly perpendicular. The patient is then extubated at the end of the procedure if possible. It is rare that mucosa and submucosal tissue need to be removed from the arytenoids if good preoperative medical control of the edema is accomplished with proton pump inhibitors and/or steroids. Mucosal resection, only if required as determined in the preoperative evaluation, is accomplished using cup forceps and scissors (Fig. 69.4&).
< div class='tao-gold-member'>
Stay updated, free dental videos. Join our Telegram channel

VIDEdental - Online dental courses