Introduction
Mechanical loading induces remodeling of the periodontal ligament and the alveolar bone and is mediated by cytokines and chemokines. In this study, we investigated the kinetics of interleukin-6 and chemokine ligands 2 and 3 levels in periodontal ligaments subjected to orthodontic forces.
Methods
We used 64 premolars in this split-mouth design study. The experimental group consisted of premolars subjected to a force of 0.980 N in the apical direction for 3 hours, 15 hours, 3 days, 12 days, or 21 days with a 0.017 × 0.025-in beta-titanium alloy cantilever. The contralateral teeth, without orthodontic appliances, were used as controls. The premolars were extracted for orthodontic reasons, and the periodontal ligaments were scraped for analysis of cytokine levels by ELISA.
Results
Compared with the control group, an increase in chemokine ligand 2 was observed on days 3 and 12, and increases in interleukin-6 and chemokine ligand 3 were observed on day 12 in the experimental group.
Conclusions
Our data demonstrated differential expressions of interleukin-6 and chemokine ligands 2 and 3 in periodontal ligaments after mechanical loading; this might reflect the distinct roles of these molecules in the bone remodeling process.
Orthodontic tooth movement is a combination of force-induced periodontal ligament (PDL) and alveolar bone remodeling. Mechanical stimuli exerted on a tooth cause vascular changes that lead to an aseptic and transient inflammatory response in the periodontal tissues. Inflammatory mediators are released and trigger biologic processes associated with alveolar bone remodeling such as bone resorption and new bone deposition.
Cytokines are key mediators involved in bone remodeling under physiologic and mechanical loading-induced conditions. Interleukin-6 (IL-6) regulates the remodeling process by directly interacting with bone cells. Orthodontic forces result in an increase of IL-6 expression in periodontal tissues. Moreover, chemotactic cytokines (chemokines) are important signals for the trafficking, development, activity, and survival of bone cells. These molecules are expressed in periodontal tissues subjected to orthodontic forces.
Studies regarding the patterns of cytokine and chemokine expression during orthodontic tooth movement have shown heterogeneity in their methods. In animal studies, periodontal tissue samples were analyzed under varying conditions. In studies with human subjects, samples from gingival crevicular fluid and periodontal tissues have been obtained at different times by using distinct experimental protocols. PDL samples have been used to quantify mRNA levels of inflammatory molecules after palatal expansion or have been tested in vitro under hypoxic treatment, loading of static compressive force, stretching-induced mechanical stress, or the influence of proinflammatory cytokines.
Although the analysis of gingival crevicular fluid is noninvasive, it provides results that represent indirect measurements of changes in the PDL. Thus, it might not be a specific indicator of periodontal remodeling in pressure or tension areas. The side independency of cytokine levels in gingival crevicular fluid is probably a result of continuous circulation of the gingival crevicular fluid in the PDL. Otherwise, we believe that the use of the PDL is a better representation of its environment. In this setting, PDL evaluation during orthodontic tooth movement is an important tool for clarifying the cellular and molecular responses to mechanical loading. This knowledge would be useful for orthodontic treatment because these molecules could be used as diagnostic markers and potential targets for therapeutic intervention. To our knowledge, no study has demonstrated the kinetics of inflammatory mediators during orthodontic tooth movement with PDL samples in a split-mouth design. The aim of this study was to determine the kinetics of IL-6 and the chemokine ligands 2 and 3 (CCL2 and CCL3, formerly known as monocyte chemotactic protein-1 and macrophage inflammatory protein 1-alpha, respectively) expression in the PDL during orthodontic treatment.
Material and methods
Eighteen patients (9 male, 9 female), aged 11 to 40 years (median, 13.5 ± 6.96 years), seen in the Department of Pediatric Dentistry and Orthodontics, Faculty of Dentistry, Universidade Federal de Minas Gerais, Belo Horizonte, Minas Gerais, Brazil, were selected to participate in this study. Based on their clinical examinations and orthodontic records, these patients required extraction of the first or second premolars for orthodontic reasons. The inclusion criteria were as follows: (1) healthy patients with no evidence of type 1 or type 2 diabetes mellitus or osteoporosis; (2) patients who had not taken systemic antibiotics, or anti-inflammatory or hormonal drugs for 6 months before the study; (3) patients who required tooth extractions before treatment with fixed appliances; and (4) patients with good periodontal health and no radiographic evidence of periodontal bone loss. This study was approved by the institutional ethics committee (protocol number 372/07). Informed consent was obtained from each participant and their guardians when the subject was less than 18 years of age.
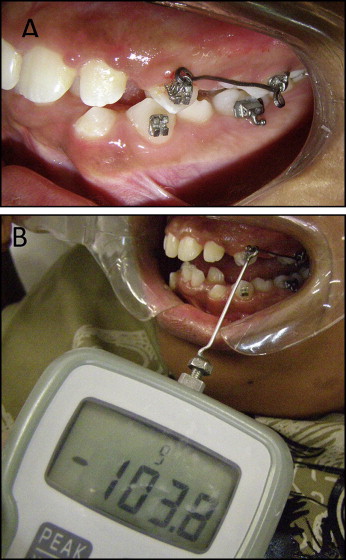
The experimental group consisted of extracted mandibular or maxillary premolars that had previously received orthodontic mechanical loading. The contralateral teeth from the same arch without orthodontic appliances were used as the controls. In the experimental group, an orthodontic appliance consisting of 0.022 × 0.028-in light Roth tubes and brackets (Morelli Orthodontics, Sorocaba, São Paulo, Brazil) was bonded with Transbond XT (3M Unitek, Monrovia, Calif). A 0.017 × 0.025-in beta-titanium alloy cantilever and a 0.010-in metallic ligature (Morelli Orthodontics) were placed between the premolar and the first molar on the same side ( Fig 1 , A ) by an orthodontist (D.F.M.). A force in the apical direction was applied to the premolar. The force magnitude was 0.980 N, measured by a digital tensiometer (model FGV-1X; Nidec-Shimpo, Itasca, Ill) that was perpendicular to the cantilever ( Fig 1 , B ). No other forces were applied to the teeth before or during this phase. The experimental teeth were randomly selected. If a patient had 4 premolars to be extracted, the pairs of teeth were allocated to 2 time points. The patients were instructed about proper oral hygiene.
The teeth were extracted at the following times: 3 hours, 15 hours, 3 days, 12 days, or 21 days. The PDL of each extracted tooth was taken from the whole root surface. Before the extraction, the force was measured again. The PDL of an extracted tooth was immediately scraped by using a 13/14 Gracey curette (Maximus, Contagem, Minas Gerais, Brazil). The sample was placed in a sterile tube and kept frozen at −80°C for further analysis. Afterward, the PDL samples were weighed and homogenized in phosphate-buffered saline solution (0.4 mmol/L of sodium chloride and 10 mmol/L of sodium phosphate [NaPO 4 ]) containing protease inhibitors (0.1 mmol/L of phenylmethylsulfonyl fluoride [PMSF], 0.1 mmol/L of benzethonium chloride, 10 mmol/L of ethylenediamine tetraacetic acid [EDTA], and 0.01 mg/mL of aprotinin A) and 0.05% Tween-20 at 1 mg/mL. The mixture was centrifuged (10,000 rpm) for 10 minutes at 4°C. The supernatant was then collected and assayed with an enzyme-linked immunosorbent assay (ELISA). The concentrations of IL-6, CCL2, and CCL3 were evaluated by using commercially available kits according to the manufacturer’s instructions (R&D Systems, Minneapolis, Minn). The results were expressed as picograms of cytokine per 100 mg of tissue.
Statistical analysis
The Shapiro-Wilks test was used to assess the quantitative variables. There was no normality of cytokines ( P <0.05); thus, nonparametric tests were used. The Mann-Whitney test was performed to verify the influence of sex on the cytokines. The Kruskal-Wallis test was used to compare cytokine levels and types of teeth. The Spearman correlation was used to assess the association between age and cytokines. The Wilcoxon test was used to assess the influence of cytokines on the experiment at each time point. The analysis was performed for each time point separately. Thus, although there was more than 1 pair of tooth per patient, it is possible to consider the sample units independent. The level of statistical significance was set at P <0.05. All statistical evaluations were performed with SPSS software (version 19.0; SPSS, Chicago, Ill).
Results
A total of 64 premolars were obtained (34 maxillary first premolars, 28 mandibular first premolars, and 2 mandibular second premolars). A mean of 6.4 pairs of teeth was allocated at each time point. The demographic description of the participants is described in the Table . The appliances were well tolerated. The initially applied force magnitude of 0.980 N was gradually reduced to the median of 0.892 ± 0.097 N before the extraction of the experimental teeth. Sex, type of tooth, age of the participants, and experimental force had no influence on IL-6, CCL-2, or CCL-3 concentrations at any time point ( P >0.05).
| Time point | Patient | Sex | Age (y) | Control tooth | Experimental tooth | Initial force (N) | Final force(N) |
|---|---|---|---|---|---|---|---|
| 3 hours | 1 | F | 12 | 14 | 24 | 0.980 | 0.980 |
| 2 | F | 14 | 14 | 24 | 0.980 | 0.980 | |
| 3 | M | 23 | 24 | 14 | 0.980 | 0.980 | |
| 4 | F | 17 | 14 | 14 | 0.980 | 0.980 | |
| 5 | F | 22 | 14 | 24 | 0.980 | 0.980 | |
| 6 | M | 14 | 24 | 14 | 0.980 | 0.980 | |
| 7 | F | 12 | 44 | 34 | 0.980 | 0.980 | |
| n = 7 | Median 14 ± 4.57 | Median 0.980 ± 0 | |||||
| 15 hours | 6 | M | 4 | 34 | 44 | 0.980 | 0.921 |
| 7 | F | 12 | 24 | 14 | 0.980 | 0.980 | |
| 8 | M | 16 | 14 | 24 | 0.980 | 0.892 | |
| 9 | M | 12 | 44 | 34 | 0.980 | 0.976 | |
| 10 | F | 12 | 34 | 44 | 0.980 | 0.980 | |
| 11 | M | 17 | 14 | 24 | 0.980 | 0.967 | |
| n = 6 | Median 13 ± 2.22 | Median 0.972 ± 0.037 | |||||
| 3 days | 3 | M | 23 | 34 | 44 | 0.980 | 0.961 |
| 10 | F | 12 | 14 | 24 | 0.980 | 0.686 | |
| 12 | F | 13 | 24 | 14 | 0.980 | 0.960 | |
| 13 | M | 18 | 34 | 44 | 0.980 | 0.860 | |
| 15 | F | 40 | 24 | 14 | 0.980 | 0.872 | |
| 16 | M | 11 | 24 | 14 | 0.980 | 0.787 | |
| n = 6 | Median 15.5 ± 11.04 | Median 0.866 ± 0.105 | |||||
| 12 days | 9 | M | 12 | 14 | 24 | 0.980 | 0.768 |
| 13 | M | 18 | 24 | 14 | 0.980 | 0.885 | |
| 14 | M | 13 | 14 | 24 | 0.980 | 0.785 | |
| 15 | F | 40 | 34 | 44 | 0.980 | 0.892 | |
| 17 | F | 11 | 24 | 14 | 0.980 | 0.902 | |
| 18 | M | 12 | 34 | 44 | 0.980 | 0.762 | |
| n = 6 | Median 12.5 ± 11.21 | Median 0.835 ± 0.067 | |||||
| 21 days | 1 | F | 12 | 44 | 34 | 0.980 | 0.790 |
| 2 | F | 14 | 45 | 35 | 0.980 | 0.690 | |
| 4 | F | 17 | 44 | 34 | 0.980 | 0.835 | |
| 8 | M | 16 | 34 | 44 | 0.980 | 0.778 | |
| 11 | M | 17 | 44 | 34 | 0.980 | 0.760 | |
| 12 | F | 13 | 44 | 34 | 0.980 | 0.745 | |
| 17 | F | 11 | 34 | 44 | 0.980 | 0.877 | |
| n = 6 | Median 14 ± 2.42 | Median 0.778 ± 0.061 | |||||
| Total | n = 18 | M = 9 F = 9 |
Median 13.5 ± 6.96 | Median 0.892 ± 0.097 |
The concentrations of IL-6, CCL2, and CCL3 are shown in Figure 2 . After 3 hours of force application, there were no significant differences between the experimental and control groups for any of the evaluated molecules ( P >0.05). Although there was no significant difference at 15 hours, the results showed a tendency toward an increase of IL-6 levels ( P = 0.068). On day 3, the expression of CCL2 was greater in the experimental group than in its control group ( P = 0.028). On day 12, IL-6 ( P = 0.046), CCL2 ( P = 0.028), and CCL3 ( P = 0.046) levels were augmented in the experimental group. On day 21, a reduction of these inflammatory mediators was observed; therefore, no difference was detected ( P >0.05). In the experimental group, correlations of IL-6 with CCL2 and CCL3 (Spearman’s correlation coefficient/Rs = 0.405, P = 0.021; Spearman’s correlation coefficient/Rs = 0.382, P = 0.031, respectively), and CCL2 with CCL3 (Spearman’s correlation coefficient/Rs = 0.426, P = 0.015), were observed.




