Ca/P molar ratio
Compounds and their typical abbreviations
Chemical formula
Solubility at 25 °C, −log(Ks)
Solubility at 25 °C, g/L
pH stability range in aqueous solutions at
0.5
Monocalcium phosphate monohydrate (MCPM)
Ca(H2PO4)2·H2O
1.14
∼18
0.0–2.0
0.5
Monocalcium phosphate anhydrous (MCPA or MCP)
Ca(H2PO4)2
1.14
∼17
c
1.0
Dicalcium phosphate dihydrate (DCPD), mineral brushite
CaHPO4·2H2O
6.59
∼0.088
2.0–6.0
1.0
Dicalcium phosphate anhydrous (DCPA or DCP), mineral monetite
CaHPO4
6.90
∼0.048
c
1.33
Octacalcium phosphate (OCP)
Ca8(HPO4)2 (PO4)4·5H2O
96.6
∼0.0081
5.5–7.0
1.5
α-Tricalcium phosphate (α-TCP)
α-Ca3(PO4)2
25.5
∼0.0025
a
1.5
β-Tricalcium phosphate (β-TCP)
β-Ca3(PO4)2
28.9
∼0.0005
a
1.2–2.2
Amorphous calcium phosphates (ACP)
CaxHy(PO4)z· nH2O, n = 3–4.5; 15–20 %
b
b
∼5–12d
1.5–1.67
Calcium-deficient hydroxyapatite (CDHA or Ca-def HA)e
Ca10−x(HPO4)x (PO4)6−x (OH)2−x (0<x<1)
∼85
∼0.0094
6.5–9.5
1.67
Hydroxyapatite (HA, Hap or OHAp)
Ca10(PO4)6(OH)2
116.8
∼0.0003
9.5–12
1.67
Fluorapatite (FA or FAp)
Ca10(PO4)6F2
120.0
∼0.0002
7–12
1.67
Oxyapatite (OA, OAp or OXA)f
Ca10(PO4)6O
∼69
∼0.087
a
2.0
Tetracalcium phosphate (TTCP or TetCP), mineral hilgenstockite
Ca4(PO4)2O
38–44
∼0.0007
a
As follows from the designation, all calcium orthophosphates contain both calcium (Ca, atomic number 20) and phosphorus (P, atomic number 15) as the major constituencies. The history of both chemical elements is long. Namely, according to Wikipedia, the free encyclopedia, calcium (from Latin calx, genitive calcis, meaning “lime”) compounds were known as early as the first century, when the ancient Romans prepared lime as calcium oxide [3]. However, calcium sulfate (also known as plaster of Paris or lime plaster) had been known much earlier: three statues were discovered in a buried pit at’Ain Ghazal in Jordan; those were sculpted with lime plaster over armatures of reeds and twine. They were made in the pre-pottery Neolithic period, around 7200 BC. However, calcium in a pure state was not isolated until 1808, when the famous British chemist and inventor Sir Humphry Davy (1778–1829) electrolyzed a mixture of lime and mercuric oxide [4, 5].
Phosphorus is a bit younger. The discovery of this element in a pure state (its name given from Greek mythology, Φωσφόρος meaning “light bearer” (Latin: Lucifer), referring to the “Morning Star,” the planet Venus) is credited to the German merchant and alchemist Hennig Brand (ca. 1630–ca. 1710) in 1669, although other alchemists might have discovered phosphorus around the same time. Brand experimented with urine, which contained considerable quantities of dissolved phosphates from the normal metabolism [6]. As the matter stands, the earliest research publication containing the word “phosphorus” in the title belongs to M. Krafft from Holland and was printed in 1677 in French Journal des Sçavans [7], which was the earliest academic journal published in Europe. In 1680, the famous British natural philosopher, chemist, physicist, and inventor Robert Boyle (1627–1691) also deposited a paper on phosphorus, but it was published only in 1693, shortly after his death [8]. However, it was the famous French chemist Antoine Laurent Lavoisier (1743–1794) who recognized phosphorus as a chemical element in 1777. It turns out that phosphorus was the first element discovered since antiquity [6].
15.2 General Definitions, Major Problems, and Limitations
To begin with, one must define the subject and outline the problems. According to Wikipedia: “History (from Greek 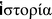 – historia, meaning “inquiry, knowledge acquired by investigation”) is the discovery, collection, organization, and presentation of information about past events. It is a field of research which uses a narrative to examine and analyse the sequence of events, and it sometimes attempts to investigate objectively the patterns of cause and effect that determine events. The task of historical discourse is to identify the sources which can most usefully contribute to the production of accurate accounts of past” [9].
– historia, meaning “inquiry, knowledge acquired by investigation”) is the discovery, collection, organization, and presentation of information about past events. It is a field of research which uses a narrative to examine and analyse the sequence of events, and it sometimes attempts to investigate objectively the patterns of cause and effect that determine events. The task of historical discourse is to identify the sources which can most usefully contribute to the production of accurate accounts of past” [9].
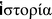 – historia, meaning “inquiry, knowledge acquired by investigation”) is the discovery, collection, organization, and presentation of information about past events. It is a field of research which uses a narrative to examine and analyse the sequence of events, and it sometimes attempts to investigate objectively the patterns of cause and effect that determine events. The task of historical discourse is to identify the sources which can most usefully contribute to the production of accurate accounts of past” [9].
– historia, meaning “inquiry, knowledge acquired by investigation”) is the discovery, collection, organization, and presentation of information about past events. It is a field of research which uses a narrative to examine and analyse the sequence of events, and it sometimes attempts to investigate objectively the patterns of cause and effect that determine events. The task of historical discourse is to identify the sources which can most usefully contribute to the production of accurate accounts of past” [9].Unfortunately, the requirement “to identify the sources” meets serious problems, due to the substantial difficulties with accessing to the scientific literature published in the first half of the nineteenth century and before. In addition, that time, both the scientific concepts and the presentation ways were rather different from the modern ones, while the chemical formulae had not been introduced yet. More to the point, scientific journals were rare and just a few of them are currently available in the electronic form. To complicate things further, numerous old journals have been discontinued, merged, and/or renamed, which strongly complicates accessing to the papers published in those journals. Luckily, Google Inc. has scanned a great number of old books as a part of its project to make the world’s books discoverable online. This timely project by Google combined with the power of the modern electronic databases of scientific publications allows reconstructing the major historical milestones on calcium orthophosphates, which was often impossible for earlier review writers. For example, a paper of 1994 by Driskell entitled “Early history of calcium phosphate materials and coatings” [10] started with the classical publication of 1920 by Albee assisted by Morrison [11]. In 1999, Shackelford published a paper, “Bioceramics – an historical perspective” [12], in which the same publication by Albee assisted by Morrison [11] was mentioned as the earliest reference. The same is valid for the historical papers by Hulbert et al. [13, 14] and Shepperd [15]. Thus, it might create a false impression that calcium phosphates were unknown before 1920. Certainly, this is not the case; nevertheless, the precise sequence of the scientific events that happened in the first half of the nineteenth century still remains poorly restorable, while the correct historical time scale of even earlier scientific events remains almost irrecoverable. This is mainly due to a lack of the citation practice existing in the scientific literature published at that time. Besides, even nowadays, for the entire eighteenth century and approximately the first quarter of the nineteenth century, only scientific books digitalized by Google are easily accessible. Just a few journal papers devoted to the subject published before ∼1820s have been found. Nevertheless, in no case this means that they do not exist; presumably, either they have not been scanned yet (especially, this might be valid for the discontinued journals) or an access to the scanned versions is restricted to a limited amount of subscribers only. Therefore, the historical narrative of the findings and discoveries of the eighteenth century is based dominantly on the available books, and the annals of the nineteenth century are based on both the available books and a limited number of journal papers, while the chronicle of the twentieth century is based on the journal publications only. Here one should also stress that starting from the second half of the nineteenth century, the detailed description of every publication on the subject becomes impossible due to permanently increasing amount of them. Furthermore, since the end of the 1910s, even mentioning all and sundry publications on the subject becomes impossible either (the quantity with moderate novelty breeds ignoring). Therefore, for the twentieth century, the milestone studies have been included only.
15.3 Investigations and Knowledge at the “Pre-calcium Orthophosphate” Time
Prior examination of issues let me briefly report on the earliest scientific publications on bones, teeth, and other types of calcified tissues of mammals, which are the main sources of calcium orthophosphates of the biological origin. Since in those early days the major scientific language was Latin, I am forced to confine myself by the English-language publications only.
According to the second earliest but the world’s longest-running scientific journal Philosophical Transactions (published in UK), the priority in journal publications on investigations of bones and teeth belongs to the famous Dutch tradesman and scientist Antonie Philips van Leeuwenhoek (1632–1723) from Delft. He is best known for his work on the improvement of a microscope and is often considered as “the father of microbiology.” Therefore, within 1674–1695, van Leeuwenhoek published several letters devoted to microscopic investigations of bones and teeth [16–20]. Namely, he wrote (please note the old-fashioned using a long, medial, or descending letter “ſ,” which is a form of the minuscule letter “s” formerly used where “s” occurred in the middle or at the beginning of a word) [16]: “I thought likewiſe, I ſaw then alſo, that that Bone conſiſted of united Globuls. Afterwards, I viewed the Shinbone of a Calf, in which I found ſeveral little holes, paſſing from without inwards; and I then imagined, that this Bone had divers ſmall pipes going longways” (p. 125). Today these small pipes are known as Haversian canals, named after the British physician Clopton Havers (1657–1702), who did the pioneering research on the microstructure of bones and teeth [21]. Let me continue citing [16]: “But I have ſince obſerved the Tooth of a Cow and I found it made up of tranſparent Globuls which I can ſee very perfectly. The ſame I have obſerved in Ivory or Elephants-Teeth. And having ſeen this ſeveral times, I doubt no longer but that all white Bones do conſiſt of tranſparent Globuls” (p. 125). Obviously, this was the first recognition of the fact that bones and teeth of mammals consist of small units (currently we call them crystals) with a transparent nature. Indeed, single crystals of all known calcium orthophosphates (Table 15.1) are transparent.
Simultaneously, other researchers tried to measure the physical properties and establish the chemical composition of various calcified tissues (both normal and pathological) of humans. For example, let me cite a summary of the experiments performed by the British physician and chemist Frederick Slare (1648–1727) from a publication of 1684 [22]: “Several Stones of the Bladder and Kidnies were diſtill’d, all afforded volatile urinous Salts; which Ferment upon any Acids. Bones were diſtill’d and found to be of agreeable Principles. Calculi examined Hydroſtatically, were found in proportion to their Bulk of water as 5 to 4. We weigh’d Bones Hydroſtatically and found them twice as heavy as their Bulk of water. Bones not eaſily wrought on by common Acids, only by nitrous ones and that without Ebullition.” (p. 532). Therefore, scientists dissolved biologically formed calcium orthophosphates in acids as far back as 1684.
Shortly before a discovery of the calcium orthophosphate nature of bones (the next section), Hérissant (presumably, it is referred to either Jean Thomas Hérissant or his son Louis Antoine Prosper Hérissant) and the Swiss anatomist, physiologist, naturalist, and poet Albrecht von Haller (1708–1777) examined a phenomenon that even the weakest acids had the power of softening bones. Namely, according to Fourcroy [23], that study was reported in 1758 and Hérissant “thought he had found in the nitric acid (with which he ſoftened the bones which he ſoaked in it, by diſſolving their earth, which he believed to be cretaceous, without affecting their membraneous cellular part) a means of inſulating the two conſtituent matters of theſe organs. Haller has verified this ſoftening of the bones even by the acetous acid and lemon juice; he ſuſpected that in the ſoftening of the bones by the effect of diſeaſes, there exiſted an acid which thus corroded them. All the chemiſts have afterwards obſerved that a ſolution of bones in an acid precipitated by alkali affords a ſeemingly earthy matter, which does not poſſeſs the property of becoming quick-lime by calcination.” (p. 388).
To conclude this part, one can see that the aforementioned few examples clearly demonstrate a scientific importance of the “pre-calcium orthophosphate” epoch, which, ideally, should be investigated in more details. Undoubtedly, one must appreciate the important observations and discoveries made by those early researchers-predecessors.
15.4 Investigations and Knowledge on Calcium Orthophosphates
15.4.1 In the Eighteenth Century
According to the available literature, the history of calcium orthophosphates began in 1769 and was associated with names of the famous Swedish chemist and metallurgist Johan Gottlieb Gahn (1745–1818) and the famous German-Swedish pharmaceutical chemist Carl Wilhelm Scheele (1742–1786). Namely, in 1881, Roscoe and Schorlemmer [24] published the following statement: “Gahn, in 1769, discovered the existence of calcium phosphate in bones, but it was not until this fact was published by Scheele in 1771 that phosphorus was obtained from bone-ash, which has from that time invariably served for its preparation” (p. 458). Furthermore, let me cite a publication of 1777 [25] (again, pay attention to the old-fashioned using a letter “ſ” instead of “s”): “I have only been informed of this diſcovery, by the Gazette, Salutaire de Bouillon, October, 1775. It is there ſaid, that Mr. Henry Gahn, a phyſician at Stockholm, has communicated a proceſs for extracting from bones the ſaline matter in question; and that Mr. Scheele had aſcertained, that the earth of animals was compoſed of a calcareous ſubſtance united with the phoſphoric acid. This diſcovery, continues the author of the article of the Gazette, belongs to Mr. Gahn, and has been confirmed by later experiments.” (p. 383). Presumably, this citation might be considered as one of the earliest mentioning on calcium phosphates in the history.
The earliest accessible description on how Scheele discovered the chemical composition of bones was given by Fourcroy in 1804 [23]: “After having, like Heriſſant, diſſolved bones in the nitric acid, he filtrated the liquor, and aſcertained that, though always acid, even when ſaturated with all that it could diſſolve of bone, it precipitated ſulphate of lime by the addition of concentrated ſulphuric acid; and that after this precipitation, the ſolution drawn off clear, and evaporated in a retort, afforded volatilized nitric acid, and left phoſphoric acid, which was fuſed into glaſs by the action of a ſufficient heat. He concluded from this experiment that the nitric acid diſſolved the phoſphate of lime, the baſe of the bones; that the ſolution in this acid was a mixture of calcareous nitrate and of phoſphoric acid; that when the firſt was decompoſed by the ſulphuric acid, and the precipitated ſulphate of lime ſeparated from it, there remained only a mixture of the two acids, the phoſphoric and the nitric, and that this latter mixture, when heated, diſengaged volatile nitric acid, whilſt the fixed phoſphoric acid remained at the bottom of the retort.” (pp. 388–389). Even more than 200 years afterward, we can add nothing to improve this description. This is an example of how the genius could do!
Simultaneously, the presence of orthophosphates was discovered in blood serum, which follows from a publication of 1770 [26]: “The ſerum conſiſts chemically of a coagulable matter, and water in which common ſal ammoniac and phoſphoric ammoniac, and generally common ſalt, and frequently ſelenites, and, fixed ammoniac, are diſſolved” (p. 309). Obviously, “phoſphoric ammoniac” means ammonium orthophosphate.
Further, according to Shepperd [15], the famous German chemist Martin Heinrich Klaproth (1743–1817) and the famous French chemist Joseph-Louis Proust (1754–1826) also contributed to calcium orthophosphates. Unfortunately, Shepperd has not provided any references to the publications by those great chemists. Nevertheless, a search performed among the Google books has given some results. For example, a short extract from a letter by M. Klaproth was published in 1788 [27], which started with the following words: “Monsieur, Je viens d’analyſer l’apatit de M. Verner (I). J’en ai retiré de la terre calcaire & de l’acide phoſphorique” (p. 313). In addition, in a French book of 1790 [28], one can find a subchapter entitled “Apatite. Phosphate de Chaux” which is started from the following sentence: “C’est Mr. Klaproth, qui à découvert le premier cette combinaison de la terre calcaire avec l’acide phosphorique” (p. 363). Further, “Mr. Proust dans une Lettre à Mr. d’Arcet a donné l’Analyse de cette pierre, qui est une combinaison de la terre calcaire avec l’acide phosphorique” (p. 366). Thus, by that time, apatite was already recognized as a calcium phosphate, which is also confirmed by a German book of 1789 [29], in which “Apatit” was called “die Phosphorsaure mit Kalkerde” (p. 2).
More to the point, according to a study, submitted on December 20, 1785 [30], the production process of orthophosphoric acid by decomposition of calcined bones in sulfuric acid was already known. This is how it was described by Lavoisier in 1790 [31]: “The bones of adult animals being calcined to whiteneſs, are pounded, and paſſed through a fine ſilk ſiewe; pour upon the fine powder a quantity of dilute ſulphuric acid, leſs than is ſufficient for diſſolving the whole. This acid unites with the calcareous earth of the bones into a ſulphat of lime, and the phoſphoric acid remains free in the liquor.” (p. 205). Further, the production process of white phosphorus has been described [31]: “The liquid is decanted off, and the reſiduum waſhed with boiling water; this water which has been uſed to waſh out the adhering acid is joined with what was before decanted off, and the whole is gradually evaporated; the diſſolved ſulphat of lime criſtallizes in form of ſilky threads, which are removed, and by continuing the evaporation we procure the phoſphoric acid under the appearance of a white pellucid glaſs. When this is powdered, and mixed with one third its weight of charcoal, we procure very pure phoſphorus by ſublimation.” (p. 206).
In the last decade of the eighteenth century, very extensive investigations on calcium orthophosphates were performed by two famous French chemists Antoine François, comte de Fourcroy (1755–1809) and Nicolas Louis Vauquelin (1763–1829), who, among other accomplishments, discovered an existence of acidic calcium orthophosphates, currently known as MCPM, MCPA, DCPD, and DCPA (Table 15.1). For example, let me cite a Fourcroy’s book of 1789 [32]: “Phoſphoric acid, poured into lime-water, precipitates from it a ſcarce ſoluble ſalt, which does not efferveſce with acids, – is decompoſed by mineral acids, but proof againſt the attacks of cauſtic alkalis. This ſalt is of the ſame nature with the baſe of bones. An exceſs of phoſphoric acid renders calcareous phoſphate ſoluble in water; but magneſia, lime, cauſtic fixed alkalis, and even ammoniac, by carrying off from it the exceſs of acid, precipitate the neutral ſalt. Calcareous phoſphate is not decompoſable by caustic alkalis, but carbonates of potaſh and ſoda decompoſe it. The ſolid matter of bones conſiſts of phoſphate ſuperſaturated with lime.” (p. 248). Here we read the correct chemical description of CDHA precipitation from H3PO4 and Ca(OH)2, followed by CDHA transformation into soluble MCPM in the presence of an excess of H3PO4, which is again transformed into insoluble CDHA by addition of basic compounds MgO, CaO, KOH, NaOH, and NH4OH. Moreover, the last sentence means that the amount of CaO in bones exceeds the stoichiometry of orthophosphates; therefore, that was the earliest indirect observation of the apatitic (Ca/P > 1.5) composition of bones.
Furthermore, in his later book of 1804 [33], Fourcroy wrote: “A FEW years ago this ſalt, which I here denominate the acid phoſphate of lime, was unknown. Scheele very properly remarked that the ſaline earth of bones was diſſolved by an acid in human urine, but he did not obſerve that this union between phoſphoric acid and the bony phoſphate, makes a kind of permanent and particular ſalt different from the latter. It was in the year 1795, the third year of the Republic, that I diſcovered it, with Citizen Vauquelin, in a connected ſeries of experiments upon bony matters; wherein we proved that the calcareous phoſphate which conſtitutes the ſolid baſe, is only in part decompoſable by acids, and that the portion of phoſphoric acid which is ſeparated, retains in ſolution phoſphate of lime, which it then defends from all ſubſequent alteration by other acids.” (p. 347). The aforementioned is confirmed by an encyclopedia published in 1816 [34]: “Super-Phosphat of Lime was diſcovered in 1795, by Fourcroy and Vauquelin. It had indeed been often formed before, but chemiſts had neglected to examine it” (p. 458). Thus, the term “superphosphate” has been known since, at least, 1795, and initially it meant calcium phosphates containing an excess (or super quantity) of phosphoric acid. Besides, in the same publication, Fourcroy mentioned other contributors to early studies on calcium orthophosphates [33]: “Citizens Nicholas of Nancy, Pelletrier, Berniard, Bullion, in France; Weſtrumb, and ſeveral chemiſts in Germany; Bonvioſin, at Turin; Tenant, Pearſon, and ſome others in England, multiplied their inquiries and experiments” (p. 337). Thus, one can notice that in the end of the eighteenth century, the subject of calcium orthophosphates was extensively investigated by a number of researchers in several countries of Western Europe.
Finally, it is important to cite a page from an agricultural chemistry book, published in 1795 [35]: “PHOSPHAT OF LIME Is contained in animal matters, such as bones, urine, shells, &c. &c. in some sorts of limestone, and in vegetable substances, particularly in the gluten, or vegeto-animal matter of wheat or other grain. It is a saline compound very insoluble. There is reason to believe, a very considerable proportion of this nearly insoluble salt is contained in most fertile soils, especially those that have been long under cultivation. It is not to be decomposed by pure alkalis; but this may be effected by mild vegetable and mineral alkalis, on the principle of the double electrive attractions; in which case, carbonate of lime (or chalk) will be precipitated, and phosphoric acid will join with the alkali, and form phosphat of potash, or phosphat of soda, according to the alkali applied. These alkaline phosphates will be found to promote vegetation in a very great degree: the substances of which they are composed, viz. alkaline salts and phosphoric acid, are found in the ashes of most vegetables.” (p. 75)
To conclude the 1700s, one should recognize that the correct basic knowledge on calcium orthophosphates became available by the end of the eighteenth century.
15.4.2 In the Nineteenth Century
For the nineteenth century, both scientific books and a limited amount of journal publications are available; however, for the first half of the nineteenth century, the quantity of journal publications was very small. Nevertheless, according to the available literature, one can claim on a real explosion of the scientific knowledge on calcium orthophosphates in the nineteenth century. For example, in 1804, Fourcroy stated that the presence of water in acidic calcium orthophosphates was already known (again, pay attention to the old-fashioned using a letter “ſ” instead of “s”) [33]: “The acid phoſphate of lime contains a remarkable quantity of water: the cryſtallization immediately ſoftens and becomes liquefied by the fire” (p. 349). The chemical compositions of both neutral and acidic calcium orthophosphates were described as follows [33]: “100 parts of phoſphate of lime contain, according to the analyſis of Citizens Fourcroy and Vauquelin,
|
Phoſphoric acid
|
41
|
|
Lime
|
59” (p. 346),
|
and “An accurate analyſis of this ſalt affords the following proportions of component parts,
|
Lime
|
45
|
|
Phoſphoric acid
|
54” (p. 351).
|
Unfortunately, it is impossible to establish the real meaning of all numerical values. Nevertheless, if we consider the first compound as the stoichiometric HA (Ca/P = 1.67), simple calculations using the proportion rules will give Ca/P = 0.96 for the acidic phosphate of lime, and if we consider the first compound as TCP (Ca/P = 1.50), similar calculations will give Ca/P = 0.86 for the acidic phosphate of lime. In both cases, the calculated Ca/P ratios appear to be close to those in DCPD and DCPA. Taking into consideration that the “acid phoſphate of lime contains a remarkable quantity of water,” one can conclude that Fourcroy prepared DCPD by 1804. Finally, one more citation from Fourcroy’s book is as follows [33]: “The phoſphate of lime is extremely difficult to fuse; nevertheleſs, by ſtrong fire, ſuch as that of a glaſs-house, it ſoftens and acquires a ſemi-tranſparence like the grain of porcelain” (p. 341). Thus, calcium phosphate ceramics were prepared by 1804.
Knowledge on acidic calcium orthophosphates was further developed in 1805 [36], “Acid Phosphate of Lime is formed either by taking away a part of the base by sulphuric, nitric, or muriatic acid, or by superadding phosphoric acid to the last described phosphate” (p. 195), and in 1806 [37], “In this way were distinguished among the salts two combinations, one neutral, and one with an excess of acid; and these were supposed to be determinate, as in sulphate and super-sulphate of potassa, or the phosphate and super-phosphate of lime” (p. 38). In 1807, a detailed preparation technique of superphosphate of lime was described as follows [38]: “This ſuper-phosphat of lime is prepared with moſt accuracy by digesting phoſphat of lime in phoſphoric acid till it is ſaturated. This ſolution yields the ſalt by evaporation in the form of ſmall ſhining seales or filaments which have almost a gelatinous conſiſtence, and a very ſour taſte” (p. 222). Obviously, this was the earliest description of MCPM preparation; however, an admixture of MCPA was possible due to a partial dehydration at the evaporation stage.
More precise information on acidic calcium orthophosphates became available by 1819 [39]: “XI. Biphosphate of Lime. This salt may be formed by digesting phosphate of lime in phosphoric acid, dissolved in hot water” (p. 327). “XII. Quadriphosphate of Lime. Glassy Phosphoric Acid of the Apothecaries. This salt may be formed by digesting, for some time, finely powdered phosphate of lime in a quantity of sulphuric acid, sufficient to saturate all the lime of the phosphate, and afterwards diluting the mixture with a sufficient quantity of water, and filtering. Sulphate of lime remains on the filter, and a liquid quadriphosphate passes through.” (pp. 327–328). Presumably, aqueous solutions of DCPD and MCPM, respectively, were prepared; therefore, one can claim that MCPM and DCPD were differentiated by 1819. Furthermore, “XIII. Subphosphate of Lime. This salt occurs native under the names of apatite or asparagus stone” (p. 328). Since the prefix “sub-” means “incomplete,” one can claim that a shortage of orthophosphate ions in apatites (if compared with phosphate of lime) was confirmed.
Various preparation processes of a pure calcium orthophosphate currently known as CDHA were developed by 1807 [38]: “Phosphate of Lime may be obtained quite pure either by ſaturating the pure acid with marble and then evaporating; or by waſhing bone-aſh with hot water to diſſolve out any ſoluble ſalt which it may contain; then adding a little acetous or dilute muriatic acid till the efferveſcence ceaſes to engage the carbonat of lime which it contains, and again thoroughly edulcorating. Phoſphat of lime is produced alſo by double decompoſition, on dropping a ſolution of any phoſphated alkali into any ſoluble calcareous ſolution that has not any great exceſs of acid. Vauquelin alſo found that on boiling phoſphat of ſoda with wet and newly precipitated carbonat of lime, the ſame reſult took place, and carbonate of ſoda with phoſphate of lime were produced.” (p. 221). One can see that some of these processes are still used to produce chemically pure CDHA. In the fourth edition of Encyclopædia Britannica (1810), applications of calcium orthophosphates were described as follows [40]: “The phoſphate of lime is of great importance in chemiſtry, for the purpoſe of extracting phoſphoric acid, to be decompoſed to obtain phoſphorus. It is also employed for making cupels, for poliſhing metals and precious ſtones, and for removing ſpots of greaſу from linen, paper, and ſilk. It is uſed in medicine as a remedy for rickets, to correct the ſuppoſed effects of acids in ſoftening the bones.” (p. 585). Although it is slightly beyond the subject, one should mention that calcium orthophosphite (Ca3(PO3)2) with trivalent P was also mentioned in that edition of Encyclopædia Britannica, as “phoſphite of lime” (p. 586).
In 1819, the major properties of CDHA were described as follows [39]: “4. Phosphate of lime is a white insoluble powder, destitute of taste, and unaltered by exposure to air. It is soluble in hydrochloric (muriatic) and nitric acids, and may be precipitated from solution in them by means of ammonia. When exposed to a very violent heat, it undergoes a kind of fusion, and is converted into a white semi-transparent porcelain. 5. According to an analysis by Berzelius, calculated in numbers, in which the equivalent number for lime is assumed, phosphate of lime is composed of
|
Phosphoric acid
|
34·3
|
|
Lime
|
29·0
|
one atom; so that it appears that the composition of this salt cannot be reconciled with the atomic theory.” (p. 327). Here, one can see the earliest mentioning of the terms “atom” and “atomic theory.”
In 1823, W. Henry described the following compounds containing calcium and phosphorus [41]: phosphuret of lime obtained by passing phosphorus over red-hot lime (p. 565), which was, obviously, calcium phosphide (Ca3P2). It “has the remarkable property of decomposing water and the water afterwards contains phosphite, or hypophosphite, not phosphate of lime.* Drop a small piece of it into a wine-glass of water, and in a short time bubbles of phosphuretted hydrogen gas will be produced; which, rising to the surface, will take fire, and explode” (p. 566), followed by references to two publications written by the famous French chemist and physicist Joseph Louis Gay-Lussac (1778–1850), to whom this discovery was credited. Obviously, the “phosphuretted hydrogen gas” was a mixture of PH3, P2H4, and more complicated PH compounds. Then, W. Henry described phosphate of lime [41], which “is constituted, according to Mr. Dalton’s experiments, of 49 acid + 51 lime, proportions which authorize us to consider phosphate of lime as consisting of 1 atom of acid = 28, + 1 atom of base = 28, the compound atom being 56” (pp. 566–567). Here, the term “atom” was used again. In addition, this is an evidence that the famous English chemist, meteorologist, and physicist John Dalton (1766–1844), who is best known for his pioneering work in the development of modern atomic theory, as well as his research into color blindness (sometimes referred to as daltonism, in his honor), also contributed to calcium orthophosphates. Afterward, other calcium orthophosphates were mentioned as follows [41]: “Bi-phosphate of lime may be formed by digesting phosphate of lime with a quantity of phosphoric acid equivalent to that already engaged in the salt” (p. 567). Obviously, it was DCPD. “Tri-phosphate of lime. – This salt, according to Mr. Dalton, perhaps formed by adding pure phosphoric acid to lime water, until a commencement of precipitation appears, when a drop or two of acid must clear the solution. If the solution be evaporated to dryness at a moderate heat, and then dissolved in water again, simple phosphate of lime remains, and a quadri-phosphate exists in the solution.” (p. 567). Unfortunately, this description does not allow to identify calcium orthophosphates. They might be DCPD (in solution) and DCPA (evaporated to dryness at a moderate heat) or OCP (in solution) and either ACP or CDHA (evaporated to dryness at a moderate heat): “Quadri-phosphate of lime. – If 100 parts of phosphate of lime be digested for 24 hours with 87 parts of sulphuric acid, diluted with a sufficient quantity of water, and be then filtered, the liquid which passes through contains the whole of the phosphoric acid, with only one-fourth of the lime which existed in the original salt … The dissolved salt is therefore a compound of 1 atom of lime = 28 + 4 atoms of acid = 112. When evaporated it forms, on cooling, pearly scales, which have an acid taste, and dissolve readily in water, giving a solution of the sp. gr. 1,44.” (p. 567). Obviously, this was MCPM with a possible admixture of MCPA formed after evaporation. Although the meaning of the statement “1 atom of lime = 28 + 4 atoms of acid = 112” remains unclear, one may guess that the numerical values 28 and 112 should be a kind of molar masses. Indeed, 28 is 50 % of the molar mass of CaO, i.e., this is a gram equivalent (synonym: an equivalent weight). According to Wikipedia, the equivalent weights were established by 1777 [42]; therefore, this suggestion sounds reasonable. Finally, W. Henry [41] mentioned “Octo-phosphate of lime. – Mr. Dalton is of opinion that a compound of 8 atoms of acid and 1 atom of lime is the true result of the process described under the last head; and that a compound of as many as 12 atoms of acid with one of lime may exist, forming a dodecaphosphate” (p. 567). Such superacidic calcium orthophosphates are currently unknown; moreover, they were excluded from the books published in 1828 [43] and 1829 [44]. Presumably, chemists quickly found a mistake. In addition, in the latter book, one can read the following [44]: “The biphosphate of lime may be prepared by adding one atom of phosphoric acid to one atom of the phosphate of lime” (p. 252). Here, the term “an atom” undoubtedly means either “a molecule” or “a mole,” i.e., the correct chemical understanding of the stoichiometry of DCPD preparation from H3PO4 and TCP was achieved by 1829.
By 1827, the German mineralogist Gustav Rose (1798–1873) established the correct understanding of the chemical composition of apatites. To demonstrate this, let me cite a publication of 1859, coauthored by the famous French chemist Henri Étienne Sainte-Claire Deville (1818–1881) [45]: “The singular composition of apatite, first determined by M. Gustav Rose in 1827, shows that it is a definite compound of chloride and fluoride of calcium with phosphate of lime” (p. 128). In 1832, a chemical term “tribasic phosphate of lime” (p. 90), which fully corresponds to both α-TCP and β-TCP, was mentioned for the first time [46].
Coincidently, the famous German chemist Eilhard Mitscherlich (1794–1863), who was a learner and a friend of J.J. Berzelius and who today was best remembered for his law of isomorphism (1819), also worked in this area. To prove this, let me cite an introduction from a paper of 1833, written by the Scottish chemist Thomas Graham (1805–1869), clearly demonstrating the established knowledge on phosphates and arsenates by that time [47]: “No classes of salts have more liberally rewarded investigation than the arseniates and the phosphates. Witness the discovery of the extraordinary phosphates of lime by Berzelius; the observation of the identity of form of the corresponding arseniates and phosphates by Mitscherlich, and the doctrine of isomorphism to which that observation led; the discovery by the same chemist of two biphosphates of soda, agreeing in composition but differing in form; and lastly, the discovery of the pyrophosphates by my friend and townsman Mr. Clark. Much, however, still remains to be done to complete the history of these interesting salts.” (p. 253). In addition, various modifications of phosphoric acid, currently known as orthophosphoric, pyrophosphoric, and metaphosphoric acids, were already known. On p. 281 of his paper, Graham called them as “a terphosphate, a biphosphate, and phosphate of water,” respectively [47].
Concerning chemical formulae of calcium orthophosphates, the earliest ones were discovered in the aforementioned paper by Graham [47] as 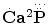 and
and 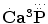 (p. 265). Another type of the chemical formulae with struck out phosphorus was published in 1844 by Mitscherlich [48] as 2.Ca2P and 5.Ca2P (p. 69a). Furthermore, a combination of both styles with dots above the element’s symbols as in Ref. [47] and striking out phosphorus as in Ref. [48] (additionally, chlorine was written as Cl and fluorine was written as F) was also used [49]. Even such a complicated way of writing with dots, striking out and mathematical fractions as
(p. 265). Another type of the chemical formulae with struck out phosphorus was published in 1844 by Mitscherlich [48] as 2.Ca2P and 5.Ca2P (p. 69a). Furthermore, a combination of both styles with dots above the element’s symbols as in Ref. [47] and striking out phosphorus as in Ref. [48] (additionally, chlorine was written as Cl and fluorine was written as F) was also used [49]. Even such a complicated way of writing with dots, striking out and mathematical fractions as
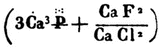
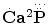 and
and 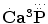 (p. 265). Another type of the chemical formulae with struck out phosphorus was published in 1844 by Mitscherlich [48] as 2.Ca2P and 5.Ca2P (p. 69a). Furthermore, a combination of both styles with dots above the element’s symbols as in Ref. [47] and striking out phosphorus as in Ref. [48] (additionally, chlorine was written as Cl and fluorine was written as F) was also used [49]. Even such a complicated way of writing with dots, striking out and mathematical fractions as
(p. 265). Another type of the chemical formulae with struck out phosphorus was published in 1844 by Mitscherlich [48] as 2.Ca2P and 5.Ca2P (p. 69a). Furthermore, a combination of both styles with dots above the element’s symbols as in Ref. [47] and striking out phosphorus as in Ref. [48] (additionally, chlorine was written as Cl and fluorine was written as F) was also used [49]. Even such a complicated way of writing with dots, striking out and mathematical fractions as
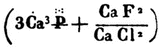
was used on page 399 of Ref. [50] to describe the chemical composition of apatite. One can see that initially neither hydrogen nor oxygen was included into the chemical formulae of calcium orthophosphates. Nevertheless, this topic kept developing. For example, the correct but just strange-looking chemical formula of apatite as 3Ca3O3,P2O5 + CaCl F might be found on page 29 of a book published in 1841 [51]. However, in another book of 1841 [52], the numerical subscripts were replaced by superscripts; therefore, the chemical formula for bone phosphate of lime was written as 8CaO + 3P2O5, that for triphosphate of lime as 3CaO + P2O5, that for diphosphate of lime as 2CaO + P2O5 + 1 eq. basic water, and one for phosphate of lime as CaO + P2O5 + 2 eq. basic water (p. 304). Obviously, the term “basic water” means acidic orthophosphates; therefore, these formulae were the first correct ones for CDHA, TCP, DCPD/DCPA, and MCPM/MCPA, respectively (hydrate water was not mentioned yet). Similar way of writing was used in some later books, such as a publication of 1854 [53], in which the chemical formula of “calcis triphosphas. – triphosphate of lime.” was written as 3CaO,PO5 (p. 625) and the chemical formulae of other calcium orthophosphates were written as 2CaO,PO5 (p. 626) and 8CaO,3PO5 (p. 627), as well as a publication of 1859 [45]: “Apatite has the composition 3(PO5, 3CaO){Cl F}Ca” (p. 128). In the latter formula, anions were placed ahead of cations. In addition, in 1843 and 1845, John Percy published identical papers [54, 55] (the earliest example of redundant publications?), in which he described formation of “a new hydrated phosphate of lime” with a chemical formula 2CaO + PO5 + 6HO, with “1 equiv. water being basic and 5 constitutional.” Obviously, the “basic water” means existence of HPO4 −, which is typical for DCPD, OCP, and CDHA, while the “constitutional water” is the hydrate water. Since only OCP contains a combination of HPO4 − with 5 molecules of hydrate water (Table 15.1), presumably, John Percy prepared OCP.
With a few exceptions [47, 54, 55], the aforementioned information on apatites and other calcium orthophosphates was taken from various books. In addition, a limited amount of journal publications of the first half of the nineteenth century is also available. Namely, research papers on the subject were published by the famous Swedish chemist Jöns Jacob Berzelius (1779–1848) [56, 57], as well as by G.O. Rees [58], M. Baruel [59], the English physician and chemist Henry Bence Jones (1813–1873) [60–62], J.D. Smith [63], and J.L. Lassaigne [64]. Among them, a paper of 1845 by Jones [60] should be noted. Namely, let me cite a figure caption: “Amorphous deposit in alkaline urine. Deposit on boiling phosphate of soda with chloride of calcium, or with sulphate of magnesia” (p. 349). This statement is astonishing because it might be considered as the earliest mentioning on ACP [65], 50 years before discovering X-rays in 1895 by the famous German scientist Wilhelm Conrad Röntgen (1845–1923)! Furthermore, Jones published the earliest-found pictures of precipitated crystals and some of them belonged to calcium orthophosphates (Fig. 15.1). Next early pictures of calcium orthophosphate precipitates from urine might be found in publications of 1859 [66] and 1860 [67].
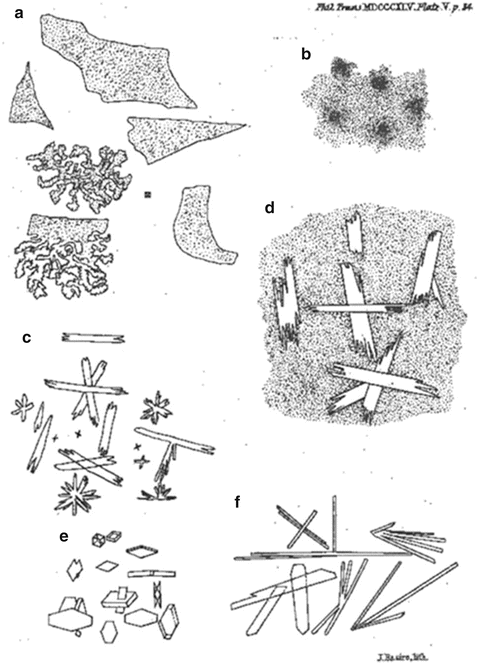

Fig. 15.1
The earliest available pictures of precipitates, some of which contain calcium orthophosphates “(a) Iridescent pellicles on some alkaline urine; (b) Amorphous deposit in alkaline urine. Deposit on boiling phosphate of soda with chloride of calcium, or with sulphate of magnesia; (c) Chloride of calcium with acid phosphate of soda, or with common phosphate of soda, after long standing; (d) Phosphate of soda with little chloride of calcium. Bone-earth phosphate; (e) On boiling phosphate of soda with sulphate of magnesia and little biphosphate of soda; (f) Phosphate of soda with sulphate of magnesia; after long standing” (p. 349). Namely, the precipitates on (b) are ACP (in the case, calcium chloride was used) and the crystals on (c) and (d) are either DCPD or OCP (Reprinted from Ref. [60])
In the end of the 1840s, the earliest-found solubility experiments of calcium orthophosphates were performed by Lassaigne [64]. Those experiments were carried out in water, saturated with carbon dioxide. The following data were obtained by 1847: “1. Pure phosphate of lime, obtained by the double decomposition of a calcareous salt and alkaline phosphate; the solubility was 0·000750. 2. Fresh bone, a piece of nearly two inches long, 46/100 dths of an inch wide, and 4/100 dths thick; solubility 0·000166. 3. Bone disinterred after about twenty years’ repose in a cemetery, the subsoil of which was sandy; solubility 0·000300.” [64]. One can see that the solubility value of pure phosphate of lime (presumably, it was either precipitated ACP or CDHA) was found to be higher than those of bones. In 1849, it was stated that “M.J.L. Lassaigne, at the meeting of the Academy of Sciences of Paris, of the 15th January, presented a memoir upon this subject, showing by experiments that the phosphate and carbonate of lime are introduced into plants in solution in water containing carbonic acid, which had before been shown as to the phosphate by M. Dumas, and has long been known as to the carbonate.” [68]. Thus, a higher solubility of both calcium orthophosphates and calcium carbonate in very weak acids was a well-known fact. The solubility data were updated in a publication of 1855 [69]: “Apatite dissolves in 393,000 parts of water saturated with carbonic acid; Artificial neutral phosphate, freshly precipitated, in 1,503 parts; Artificial basic phosphate, freshly precipitated, in 1,102 parts” (p. 27). Furthermore, “apatite requires 356 times as much liquid for its solution, as the same salt artificially prepared” (p. 28). Obviously, the “artificial basic phosphate, freshly precipitated,” was ACP, and, indeed, the differences in solubility values between ACP and the stoichiometric well-crystallized HA might be that big. The results of the successive solubility experiments were published in 1866 [70], 1868 [71], 1871 [72, 73], and 1883 [74].
The second half of the nineteenth century started with “Chemistry” by Wilson, published in 1850 [75]. The following parts from that book are worth citing: “797. Phosphates of Lime. – There are many compounds of lime and phosphoric acid, owing to the peculiarity of that acid in relation to the number of equivalents of base it combines with at once. The most interesting phosphate of lime is that which occurs in bones, and is distinguished as the bone-earth phosphate, 3CaO,PO5.” (p. 219). Thus, various types of calcium orthophosphates were known already. However, the preparation technique sounds unusual to the modern readers: “The phosphorus combines in part with the oxygen of the lime, CaO, to form phosphoric acid, and this with undecomposed lime, to form phosphate of lime, CaO,PO5. At the same time another portion of the phosphorus combines with the calcium of the lime, forming phosphuret of calcium, CaP” (p. 164). Thus, the compositional differences between the chemical formulae of the oxygen-containing calcium phosphates and the oxygen-free calcium phosphides were clearly established by 1850. The earliest-found thesis on calcium orthophosphates was defended in 1853 [76]. In a publication of 1859, various preparation techniques of apatites were described as follows [45]: “M. Daubrée has prepared apatite by passing chloride of phosphorus over lime; M. Mauross and M. Briegleb, in continuation of the remarkable researchers made in the laboratory of Professor Wöhler, have reproduced apatite in more distinct and more beautiful forms by taking advantage of the double decomposition of alkaline phosphates and chloride of calcium; M. Forchhammer, by acting on phosphate of lime with chloride of sodium, has obtained very beautiful specimens of the same mineral species.” (pp. 129–130). Here, the readers’ attention was also paid to the names of additional early contributors to calcium orthophosphates.
In “Chemistry” by Brande and Taylor, published in 1863 [77], one can find the following statements: “Common Phosphate of Lime; Tribasic Phosphate of Lime; Bone Phosphate; (3(CaO),PO5). This salt occurs abundantly in bone-ash, and is found as a mineral product” (p. 331). Furthermore, “Native phosphate of lime (bone phosphate) occurs in apatite, moroxite, phosphorite, and asparagus stone; its primitive form is a six-sided prism: it also occurs in some volcanic products” (p. 332). Thus, a similarity between the inorganic phase of bones and calcium orthophosphate rocks of natural origin (apatite and phosphorites) was known by 1863. “When a solution of bone-earth in hydrochloric or nitric acid is boiled to expel all carbonic acid, and decomposed by caustic ammonia, the bone-phosphate separates in the form of a bulky precipitate, which, when perfectly dried, is white and amorphous” (p. 331). This seems to be the second earliest mentioning on ACP. Next citation, “The substance known under the name of coprolites, and which appear to be the excrements of fossil reptiles, also abound in phosphate of lime” (p. 332), means that by 1863, researchers were aware of this fact.
Further historical description is based on the journal publications only. Namely, C. Morfit [78]; Robert Warington (there were two chemists with this name, presumably, a father (1807–1867) and a son (1838–1907)), who performed the earliest well-documented systematic studies of the outstanding quality [70, 72, 79, 80]; and R. Fresenius [81] also worked with calcium orthophosphates. Interestingly, C. Morfit wrote the chemical symbol of phosphorus as Ph, and, thus, he wrote the chemical formula of tribasic phosphate as 3CaO, PhO5 [78]. Furthermore, let me cite a paragraph from a Warington’s paper of 1866 [70]: “Mitscherlich tells us, that when chloride of calcium is added to ordinary disodic phosphate, the latter being maintained in excess, the precipitate formed is tricalcic phosphate, while the solution becomes acid from the production of monosodic phosphate. Berzelius, on the contrary, states, that the precipitate formed under these conditions is not tricalcic phosphate, but the octocalcic triphosphate, which lie has elsewhere described. All experimenters agree, that when the operation is reversed, and disodic phosphate is poured into an excess of chloride of calcium, the precipitate is neither tricalcic nor octocalcic, but dicalcic phosphate.” (pp. 296–297). Thus, TCP, DCP, and OCP have been differentiated from each other by 1866, while, in fact, at least, 20 years earlier because Berzelius died in 1848. Furthermore, the researchers were already aware of the fact that the type of a precipitated calcium orthophosphate depended on a sequence of the mixing reagents.
Among the available publications written by two Robert Waringtons [70, 72, 79, 80], Ref. [70] by Robert Warington Jr. deserves both a special attention and extensive citations. For example, to prove that OCP indeed was already known by 1866, let me make another citation from Ref. [70]: “Octocalcic phosphate can only be produced by the simultaneous formation of monosodic phosphate:
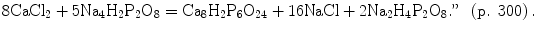
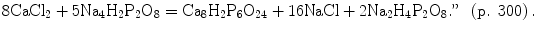
One can see a balanced chemical equation, identical to the modern ones. It is hard to believe that it was published in 1866! It is interesting to note that only 22 years appeared to be enough to perform a transition from the primitive chemical formulae without oxygen, such as 2.Ca2P and 5.Ca2P [48], to the modern chemical equations. More to the point, the chemical formulae of hydrated forms of calcium orthophosphates were known by 1866: “8·73 grs. of the vacuum-dried salt, lost on ignition 12·30 grains, or 26·35 per cent.; the formula Ca2H2P2O8·4H2O, demands 26·16 per cent. of water” (p. 299). Needless to explain, “Ca2H2P2O8·4H2O” represents two molecules of DCPD (see Table 15.1). Furthermore, “It is interesting to observe that while disodic phosphate is of an alkaline nature, dicalcic phosphate possesses faint acid properties” (p. 300). More to the point, the form and shape of DCPD crystals were described as well: “The crystalline form of the dicalcic tetrahydrated phosphate has been examined by Professor Church. He describes the crystals as thin rhomboïdal plates, of which the diagonally opposite acute angles are sometimes truncated, hexagonal forms being thus produced. This truncation seems to be occasionally hemihedral, and then may proceed up to the diagonal between obtuse angles; from this change triangular forms arise. Other modifications are also met with.” (pp. 300–301). Another interesting conclusion might be found here: “We may then safely affirm that whenever dicalcic phosphate, octocalcic triphosphate, or any phosphate of intermediate composition, is precipitated from solution by ammonia, the salt obtained will be the octocalcic triphosphate; a tricalcic phosphate cannot be obtained in this manner. The following is probably a type of the reaction:
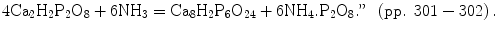
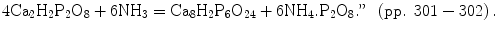
This seems to be the earliest mentioning on the fact that TCP cannot be precipitated from the aqueous solutions (currently we know that ACP or CDHA are precipitated instead). In addition, the following citation from the same publication “It is quite possible that precipitated tricalcic phosphate may possess somewhat different solubilities, when prepared by different methods; this difference can, however, scarcely be great” (p. 304) has two important consequences: (1) by 1866, this fact was not quite clear yet; (2) it indirectly points to variability in the Ca/P ratio for the precipitated ACP or CDHA, which is the major reason of different solubilities.
The latest available publication by Warington of 1873 [80] was devoted to the hydrolysis of a freshly precipitated TCP (i.e., either ACP or CDHA) to the stoichiometric HA under continuous (up to 50 h) boiling in distilled water. From the results of numerous chemical analyses, the author concluded that during boiling an aqueous suspension of the TCP was slowly transformed to a suspension of 3Ca3P2O8. CaOH2O (i.e., HA – see Table 15.1) and soluble acidic calcium orthophosphates. The following conclusion was made: “Since it appears that all phosphates of calcium less basic than apatite are unstable under the continued action of pure water, it seems probable that a more exact examination of natural phosphates would show that many phosphates now regarded as tricalcic are in fact of a more basic nature” (p. 989). Thus, the apatitic nature of the majority of natural calcium orthophosphates has been predicted in 1873. The next available journal article on the TCP hydrolysis was published in 1929 only [82].
Now it would be logical to cite a publication of 1906 by Wells [83]: “The apparent constancy of the proportion of carbonate and phosphate of calcium in bones made an impression on Hoppe-Seyler in 1862, and we find him speculating on the possibility of the components of the two salts being joined together to form one giant molecule: 3(Ca3(Po4)2)–CaCo3, which he imagined might be united in some such way” (p. 522) – see Fig. 15.2. Further, Wells mentioned: “This formula is interesting chiefly from the historical standpoint, but it serves to emphasize the tendency of these salts to appear in nearly constant proportions in the animal body, a fact possibly of some importance” (p. 523). Obviously, the atomic arrangement shown in Fig. 15.2 represents the earliest structural drawing of a single molecule of an ion-substituted calcium orthophosphate, currently known as carbonate apatite. An attentive reader will notice two different types of calcium (currently known as Ca(1) and Ca(2)) in that structure. Besides, that time, apatites were considered as combined compounds, which results from this citation of 1879 [84]: “Calcium phosphate, combined with calcium chloride or calcium fluoride, occurs in the well-known minerals, apatite and osteolite” (p. 188). One might guess that, in the nineteenth century, the atomic arrangement of single molecule of carbonate apatite (Fig. 15.2) could have inspired researchers to compose similar drawings for the single molecules of FA, HA, and/or chlorapatite; unfortunately, I have succeeded to find nothing on this matter.
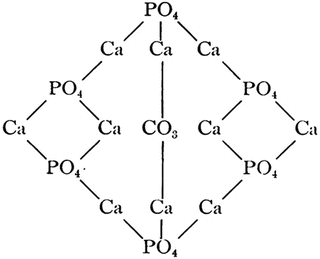

Fig. 15.2
The first available structure of a bone mineral, currently known as carbonate apatite (Reprinted from Ref. [83])
Chemical equations, describing various interactions between calcium phosphates and other chemicals, have been known since, at least, 1863. For example, the afore-cited production processes of both orthophosphoric acid and white phosphorus from the Lavoisier book [31], in 1863, were written using chemical equations [77]: “When bone-phosphate is digested in dilute sulphuric acid, it is resolved into sulphate of lime and (if a sufficiency of sulphuric acid be used) phosphoric acid:
![$$ 3\left(\mathrm{CaO}\right),{\mathrm{PO}}_5+3{\mathrm{SO}}_3=3\left[\mathrm{CaO},{\mathrm{SO}}_3\right]+{\mathrm{PO}}_5.\mbox{''}\kern0.5em \left(\mathrm{p}.\kern0.5em 331\right). $$](/wp-content/uploads/1008/A312282_1_En_15_Chapter_Equc.gif)
![$$ 3\left(\mathrm{CaO}\right),{\mathrm{PO}}_5+3{\mathrm{SO}}_3=3\left[\mathrm{CaO},{\mathrm{SO}}_3\right]+{\mathrm{PO}}_5.\mbox{''}\kern0.5em \left(\mathrm{p}.\kern0.5em 331\right). $$](/wp-content/uploads/1008/A312282_1_En_15_Chapter_Equc.gif)
One can see that hydrogen was not included into the chemical formulae yet. Nevertheless, already in 1866, it was included by Robert Warington Jr. [70] (see above). Furthermore, chemical formulae of various types of phosphates (namely, metaphosphate and acid phosphate), as well as differences in their solubility, were known by 1881 [24]: “In order to prepare phosphorus, the bone-ash is first mixed with so much dilute sulphuric acid as to form the acid phosphate:
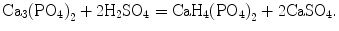
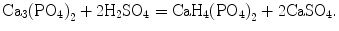
Stay updated, free dental videos. Join our Telegram channel

VIDEdental - Online dental courses


