Dentin composition
Apatite crystals (mineral)
Organic matrix (type I collagen and non-collagenous proteins)
Water (free and bound)
50 %
30 %
20 %
7.1.2 Extracellular Organic Matrix
The intertubular dentin matrix is mainly composed of type I collagen fibrils which are associated to non-collagenous proteins and proteoglycans, forming a three-dimensional organic network associated in a mineral phase constituted mostly by hydroxyapatite crystallites.
Dentin Composition
Ninety percent of the sound dentin is composed by type I collagen fibrils and 10 % appear as non-collagenous proteins (NCPs). They are located in dentin as components of the matrix extracellular molecules (ECM). NCPs include the family of SIBLINGs, which are phosphorylated proteins implicated in dentin mineralization (osteopontin, dentin sialoprotein (DSP) 1, bone sialoprotein (BSP), MEPE). Other molecules characterized as non-phosphorylated ECM proteins (osteocalcin, osteonectin) act as promotor or inhibitors of mineralization. Some molecules take origin in the serum (α2HS glycoproteins, albumin). Proteoglycans or SLRPs, including decorin, biglycan, lumican, and fibromodulin, play role in the formation of dentin complexes. Phospholipids, a series of small proteases, and ECM enzymes are also located within the ECM (Goldberg et al. 2011).
During the carious decay, NCP are the main targets for acidic or enzymes degradation. Type I collagen is more resistant. Collagenases, gelatinases, and some MMPs (matrix metalloproteinases) contribute to collagen degradation. The cross-links between collagen fibrils better resist bacterial attacks. All the molecules listed in the previous paragraphs provide substrates implicated in dentin degradation during the carious decay. For example, this is namely the case for MMP2, an enzyme modulating BSP, and detected by immunohistochemistry in association with the full-length molecule associated to the caries-affected dentinal tubules (Boushell et al. 2011). However, the degradation is not uniform and depends specifically on the dentin layer and the site of exposure to bacteria.
7.2 Size of the Hydroxyapatite Crystallites in the Intertubular Dentin
The size of needlelike apatite crystals is approximately 5 nm × 30 nm × 100 nm [length and width of the sound dentin intertubular crystals (109.6 ± 19.9 nm and 6.2 ± 1.2 nm)]. It contains less calcium (calcium deficient) and more carbonate (carbonate-rich hydroxyapatite) and therefore is more soluble than stoichiometric apatite (Zavgorodnuy et al. 2008). In sound dentin, the length of crystallites is 109 nm ± 19.9 nm, whereas in the transparent zone, crystals located in the middle dentin display 51 ± 13.0 nm as mean length and near the DEJ after demineralization 37.8 ± 20.1 nm (Zavgorodnuy et al. 2008).
7.3 White and Brown Spots
When the carious enamel lesion reaches the dentinoenamel junction (DEJ), a brownish discoloration in dentin suggests that the dentin respond with the formation of translucent dentin. There is no spreading of dentin caries along the DEJ but penetration of the lesion may be observed within dentin, at least in non-cavitated occlusal lesions with an apparently intact enamel surface (Bjorndal and Thylstrup 1995). Lateral spread along the DEJ was found in lesions with micro-cavitation or cavitation, therefore when soft and infected dentin was present (Ekstrand et al. 1998).
In the carious dentin, five zones have been identified (Newbrun 1983; Yamada et al. 1983). A simplified classification of the different carious layers allows distinguishing between the caries-infected dentin layers, which cannot be remineralized, located in the outer caries layer, and the caries-affected layers. These layers are both superficial and partially demineralized, but they may also be remineralizable. The inner caries layer, which is sclerotic (transparent), displays a subtransparent highly mineralizable sclerotic layer, located between the carious lesion and the normal dentin (Mazzoni et al. 2015).
Table 7.2 summarizes the different zones and events occurring during the development of carious events (Fig. 7.1):
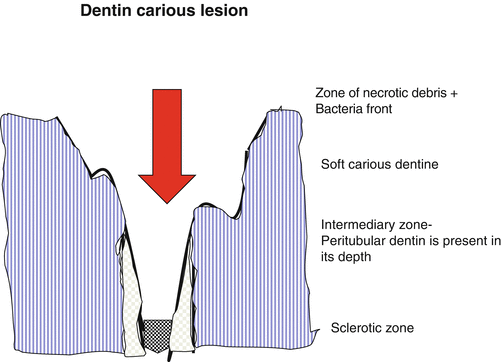
Table 7.2
|
Zone of degradation: food debris
(Vegetal and animal: meat, muscle). Degradation front of demineralized dentin
Outer carious dentin infected (Fig. 7.2)
|
Zone of bacterial invasion
(Soft carious dentin)
Inner carious dentin
(Fig. 7.3)
|
Zone of demineralization
(Infected soft carious dentin) enlarged tubules
(Affected tubules + mineral reprecipitations) intratubular non-apatitic precipitation
Whitlockite, calcium phosphate, non-occluding the lumen of tubules
(Fig. 7.4)
|
Zone of dentin sclerosis (transparent dentin):
Intratubular needlelike apatitic ghostlike mineral occluding the lumens of tubules
|
Zone of fatty degeneration
|
|
Carious cavity: degraded dentin unremineralizable
|
Carious cavity:
>Enlarged tubules > intertubular dentin demineralized
>No peritubular dentin
Discolored layer
|
Carious cavity:
Infected: active bacteria inside tubules.
Affected: gradient of peritubular dentin + increase of intertubular dentin mineralization
Transparent layer
|
Sclerotic dentin Occlusion of the lumens of tubules by:
Reprecipitation in non-apatitic and hydroxyapatite mineral forms
Subtransparent layer
|
“Sound” dentin with lipidic inclusions. Reactionary dentin
(Fig. 7.7)
|

Fig. 7.1




Schematic diagram of a dentin carious lesion. Beneath the zone of necrotic debris, a soft carious dentin is found, followed by the intermediary zone. Peritubular dentin has been dissolved, but reappears in the depth of the lesion. The sclerotic dentin where tubules are occluded by intratubular mineralization formed the sclerotic zone
Stay updated, free dental videos. Join our Telegram channel

VIDEdental - Online dental courses


