Introduction
The aim of this study was to investigate the effect of parathyroid hormone injection on experimental tooth movement in rats.
Methods
Sixty male Wistar rats were randomly divided into 2 groups. Their maxillary right first molars were moved mesially with nickel-titanium closed-coil springs. The experimental group received daily subcutaneous injections of parathyroid hormone at a dose of 4 μg per 100 g of body weight for 12 days, and the control group received vehicle injections. The results were evaluated by intraoral measurements and by hematoxylin and eosin, tartrate-resistant acid phosphatase, and immunohistochemistry staining.
Results
The tooth movement and osteoclast numbers were significantly increased in the parathyroid hormone group compared with the control group. The expressions of receptor activator of nuclear factor kappa B ligand and insulin-like growth factor-I were significantly stimulated in the parathyroid hormone group.
Conclusions
The data suggest that short-term parathyroid hormone injection might be a potential method for accelerating orthodontic tooth movement by increasing the alveolar bone turnover rate.
During orthodontic treatment, appliances usually cause inconveniences in patients’ daily lives and hinder proper oral hygiene, increasing the risk of dental caries and periodontal disease. Also, orthodontists are usually confronted with difficult situations of ineffective tooth movement, such as uprighting of tipping molars, or space closure in patients with high alveolar bone density. Therefore, it is a primary concern for orthodontists to develop a method of accelerating tooth movement to make orthodontic treatment more effective. Since orthodontic tooth movement is achieved through alveolar bone remodeling, medicines stimulating bone metabolism might promote tooth movement. As an important regulator of bone metabolism, parathyroid hormone could be a potentially valuable agent for facilitating tooth movement.
Parathyroid hormone, as a major regulator of calcium and phosphate homeostasis, has gained particular attention for its paradoxical effects on bone metabolism. Relative studies have confirmed that parathyroid hormone could stimulate both osteoclast-mediated bone resorption and osteoblast-mediated bone formation, therefore accelerating the bone turnover rate. The ultimate effect of parathyroid hormone on bone remodeling is determined by the administration protocol. Continuous infusion of parathyroid hormone results in a catabolic effect, whereas intermittent injection leads to an anabolic effect. Actually, intermittent low-dose parathyroid hormone analogs have been widely administered in the clinical treatment of osteoporosis.
Previous studies have investigated the effect of different administration patterns of parathyroid hormone on orthodontic tooth movement rate and periodontal repair. The results suggested that systemic continuous infusion or local chronic application of parathyroid hormone could accelerate tooth movement in rats through enhancement of alveolar bone resorption, whereas long-term intermittent injection of parathyroid hormone facilitated periodontal repair of bone or root resorption after orthodontic tooth movement through activation of osteoblastic cells. However, the effect of intermittent injection of parathyroid hormone on the tooth movement rate has not been clarified yet. Salazar et al evaluated the tooth movement in ovariectomized rats treated with intermittent parathyroid hormone injections and found that they did not hinder tooth movement; on the contrary, those rats showed greater tooth movements than did the control group. The authors mentioned that unlike other osteoporosis treatment drugs such as bisphosphonates, calcitonin, and calcium with vitamin D, which diminish bone resorption, parathyroid hormones stimulated osteoblast function with no interference in osteoclast activity and facilitated bone remodeling. This could explain the acceleration effect of intermittent parathyroid hormone.
The precise mechanism underlying the effect of intermittent parathyroid hormone on bone metabolism has not been clarified yet. However, it is verified that under intermittent parathyroid hormone administration, both osteoblast and osteoclast activities are stimulated. Insulin-like growth factor-I (IGF-I) and receptor activator of nuclear factor kappa B ligand (RANKL)/osteoprotegerin system are essential to the biologic processes, respectively. The receptors of parathyroid hormone are only expressed on the cell membrane of osteoblasts. After the binding of parathyroid hormone molecules to their receptors, the osteoblasts are stimulated to produce more IGF-I via a cyclic adenosine monophosphate (cAMP)-dependent mechanism, which functions as an autocrine/paracrine factor and activates its adaptor molecule insulin-receptor substrate-1 in osteoblast precursors in bone marrow, and causes osteoblast proliferation, differentiation, and function. On the other hand, osteoblasts stimulated by parathyroid hormone molecules also express more RANKL on the cell membrane, which binds to the receptor activator of nuclear factor kappa B (RANK) on the cell membrane of osteoclastic precursors through cell-to-cell contact and stimulates osteoclast proliferation, differentiation, and activation. Osteoprotegerin, the decoy receptor of RANKL, is secreted by osteoblasts and inhibits the binding of RANKL and RANK. The RANKL/osteoprotegerin ratio is an indirect reflection of the actual functional RANKL molecules and the activity of osteoclastogenesis. The osteoprotegerin expression level is not significantly changed by parathyroid hormone administration; therefore, the resultant RANKL/osteoprotegerin ratio is remarkably increased.
The anabolic effect of intermittent parathyroid hormone in the clinical treatment of osteoporosis involves not only osteoblastic bone formation, but also osteoclastic bone resorption. The ultimate increase of bone density is achieved through the “anabolic window,” which means that bone formation is greater than bone resorption during the first 6 to 18 months. Actually, some researchers suppose that active osteoclastic resorption is necessary for the effect of the parathyroid hormone on bone formation in a remodeling system. After intermittent parathyroid hormone administration, RANKL expression is greatly enhanced, resulting in an increase in osteoclastic resorptive activity. In turn, the resorptive activity increases the release of osteogenic growth factors from bone matrix and osteoclasts, and it stimulates bone remodeling.
Based on the previous literature, intermittent parathyroid hormone administration stimulates both osteoclast and osteoblast activity, and it promotes bone formation via enhanced bone remodeling. We hypothesize that intermittent parathyroid hormone could also facilitate orthodontic tooth movement through stimulation of alveolar bone remodeling. The purpose of this study was to investigate whether intermittent parathyroid hormone administration can accelerate orthodontic tooth movement and its impact on the expression of the RANKL/osteoprotegerin system and IGF-I in the periodontal area.
Material and methods
All experimental procedures were approved by the animal experiment ethics committee of the State Key Laboratory of Oral Diseases of Sichuan University in China. Sixty 8-week-old male Wistar rats weighing 200 ± 10 g were obtained from the university’s experimental animal center. The animals were randomly divided into 2 groups of 30 animals each: the parathyroid hormone group and the control group. They were kept in plastic cages with a standard 12-hour light-and-dark cycle and fed a soft diet with water ad libitum. During the experiment, their weights were recorded every day.
An orthodontic elastic closed-coil spring (Grikin Advanced Materials, Beijing, China) was fixed between the maxillary right first molar and the incisors with ultraviolet curable resin (MB 4403; 3M Unitek, Monrovia, Calif) under anesthesia with intraperitoneal injection of 2% ketamine hydrochloride at 2 mL per kilogram of body weight. The maxillary first molars were moved mesially with a force of 40 g ( Fig 1 ). The appliances were activated immediately upon insertion, and the fit was checked daily. No reactivation was performed during the experimental period.
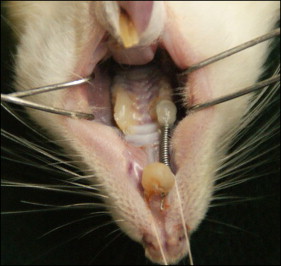
Starting 1 day before the installation of the orthodontic appliance, the animals in the experimental group received a daily injection of recombinant human parathyroid hormone (1-34) (PTHP-002; Chinese Peptide, Hangzhou, China) at a dosage of 4 μg per 100 g of body weight dissolved in phosphate-buffered saline (PBS) at a concentration of 1 μg per milliliter. The animals in the control group received the same volume of vehicle (PBS). The protocol of parathyroid hormone (1-34) injection was decided according to previous studies.
Six animals from each group were killed on day 0 (the day before orthodontic force application) and on days 3, 6, 9, and 12. After that, the maxilla of each animal was dissected and divided into halves and prepared for light microscopic observation. The distance of tooth movement was determined by measuring the separation between the first and second maxillary molars using a vernier caliper with an accuracy of 0.02 mm.
The right half of the maxilla of each animal was fixed in 4% paraformaldehyde in 0.1 mol/L of PBS for 24 hours and decalcified in neutral 10% ethylene diamine tetra-acetic acid at room temperature for at least 2 months. This solution was changed every other day. A methyl red-ammonia-ammonium oxalate solution test was carried out to determine whether decalcification was completed. After dehydration and paraffin embedding, 5-μm serial sections in a mesiodistal direction parallel to the long axis of the distal root of the first molar were cut on a microtome (HM 355S; Microm International, Walldorf, Germany) and mounted on glass slides. Selected sections were stained with hematoxylin and eosin and tartrate-resistant acid phosphatase (TRAP).
Tissue sections were placed in a tris-hydroxy methyl aminomethane buffered saline solution (TBS) at pH 7.4 for 10 minutes. Thereafter, endogenous peroxidase activity was blocked in methanol/hydrogen peroxide for 10 minutes in the dark. Subsequently, sections were rinsed and then preincubated with TBS containing 4% bovine serum albumin (TBS/BSA) for 20 minutes to prevent unspecific background staining. Then, sections were incubated with a polyclonal primary antibody of rabbit origin raised against peptide mapping at the carboxy terminus of the protein (ZSGB-BIO, Beijing, China) in a 1:200 working solution (osteoprotegerin) or 1:50 working solution (RANKL) of TBS/BSA at 4°C overnight in a humidified chamber. The slides were rinsed again and incubated for 30 minutes with a rabbit anti-goat immunoglobulin diluted 1:100 in TBS/BSA (ZSGB-BIO) as secondary antibody. Incubation was stopped in TBS before the peroxidase-antiperoxidase complex was administered for 30 minutes. After another rinse, the tissue sections were stained in a 3,3′-diaminobenzidine solution for about 5 minutes, rinsed, and then counterstained with Mayer’s hematoxylin, dehydrated, and cover-slipped for light microscopic analysis. To prove the specificity of the immunoreactions, negative controls were carried out by omitting the primary antibody or using nonimmune immunoglobulin G instead, omitting both the primary and secondary antibody, and using TBS/BSA instead.
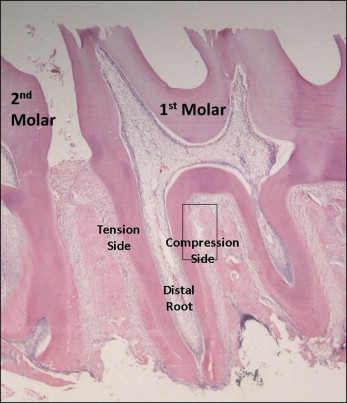
The light microscopic images were captured per specimen on the former compression side of the distal root of the maxillary first molar at the mesiocoronal area at magnification of 100 times, and near the bifurcation area at the mesiocoronal region at magnification of 400 times ( Fig 2 ). Among the serial sections in the mesiodistal direction from each specimen, several central sections were selected for histomorphometric and immunohistochemical measurements: 3 sections for each measurement. The number of TRAP positive cells in the periodontal area was counted and expressed as cell numbers per millimeter of root length. The immunoreactivity of molecules was evaluated using an automatic image analysis system (HPIAS-1000, version 6.0; Media Cybernetics, Silver Spring, Md), and the immunoreactive intensity was converted to gray-scale values. All measurements were performed by 2 operators (H.H., R.L.), and any disagreement was resolved through discussion or assessment by a third investigator (J.C.). Both operators were blinded to the treatment allocation.
Statistical analysis
The data were processed with SPSS software (version 11.5; SPSS, Chicago, Ill). The results were expressed as means and standard deviations. For histologic and immunohistochemical data, the results for each group were the means and standard deviations of the 6 measurements, each as an average of the 3 sections from a specimen. The data of tooth movement distance, osteoclast numbers, osteoprotegerin, and RANKL immunoreactivity between groups were evaluated by 1-way analysis of variance. The significance level was set at P <0.05.
Results
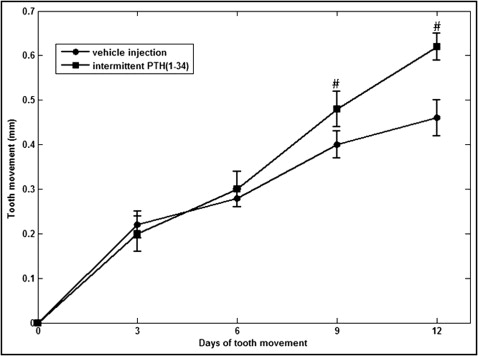
During the early phase of the experiment, there was no significant difference of total tooth movement between the parathyroid hormone group and the control group, with approximate averages of 0.20 ± 0.04 and 0.22 ± 0.03 mm (for the parathyroid hormone and control groups, respectively) on day 3, and 0.30 ± 0.04 and 0.28 ± 0.02 mm (for the parathyroid hormone and control groups, respectively) on day 6. However, during the late phase, the differences between the 2 groups became significant. On day 9, the total tooth movement amounts were 0.48 ± 0.04 and 0.40 ± 0.03 mm on average for the parathyroid hormone and control groups, respectively ( P <0.05). On day 12, the differences between the 2 groups became greater, and the total tooth movement amounts were 0.72 ± 0.03 and 0.52 ± 0.04 mm on average for the parathyroid hormone and control groups, respectively ( P <0.05) ( Fig 3 ).
Multinucleated clastic cells appeared on day 3 and increased on days 6 and 9. These cells formed the resorption lacunae around themselves on the surface of the alveolar bone. The resorption was more obvious in the mesiocoronal and distoapical regions of the distal root, indicating tipping movement of the first molar. In the parathyroid hormone group, there were more functional multinucleated clastic cells on the surface of alveolar bone on days 6 and 9, and the resorption lacunae were larger ( Fig 4 ).
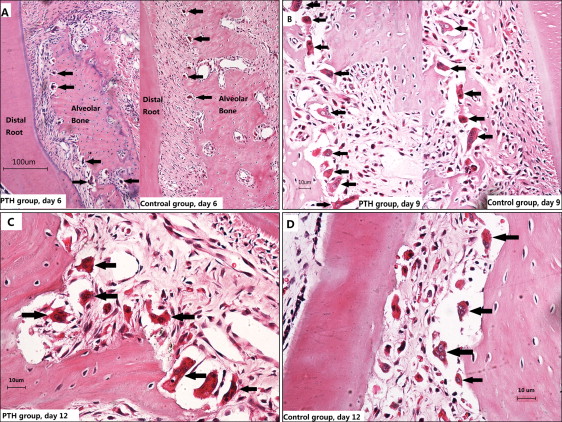
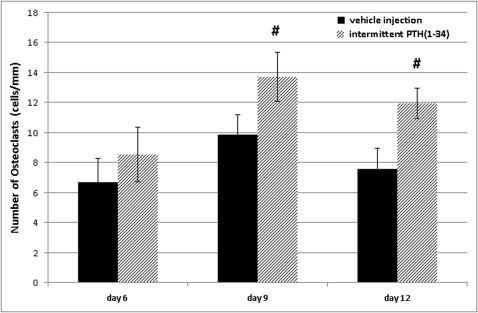
The multinucleated osteoclasts and a few mononucleated osteoclast precursors in the periodontal ligament area were all stained positive for TRAP. Most of the TRAP positive cells were located in the resorption lacunae on the alveolar bone surface. Only a few mononuclear TRAP positive cells were located near vessels in the periodontal ligament, resembling hematopoietic osteoclast precursors. On day 3, a few osteoclasts appeared on the surface of the alveolar bone, but the resorption was not active, and few resorption lacunae were formed. On day 6, the number of osteoclasts increased remarkably, and the average cell numbers per millimeter of root length were 8.55 ± 1.83 and 6.72 ± 1.58 for the parathyroid hormone and control groups, respectively ( P >0.05). On day 9, the osteoclast numbers increased to average cell numbers per millimeter of root length of 13.72 ± 1.62 and 9.85 ± 1.34 for the parathyroid hormone and control groups, respectively, with a significant difference between the groups ( P <0.05). On day 12, the average osteoclast numbers per millimeter were 11.96 ± 1.02 and 7.56 ± 1.42 for the parathyroid hormone and control groups, respectively, and the difference between the groups remained significant ( P <0.05). It seems that the difference between the parathyroid hormone and the control groups became larger over time ( Figs 5 and 6 ).