(1)
Department of Pediatric Dentistry, Selcuk University, Faculty of Dentistry, Konya, Turkey
Abstract
Stem cell technology is developing at a rapid pace. Every piece of seemingly unrelated, floating data is being conglomerated to broaden current knowledge and move closer to the inevitable shift in paradigms of current fundamental cell biology systems. The question of what a stem cell actually is may no longer require further discussion; however, there are still some differences in opinion between groups in terms of their functional roles.
Stem cell technology is developing at a rapid pace. Every piece of seemingly unrelated, floating data is being conglomerated to broaden current knowledge and move closer to the inevitable shift in paradigms of current fundamental cell biology systems. The question of what a stem cell actually is may no longer require further discussion; however, there are still some differences in opinion between groups in terms of their functional roles.
4.1 Evidence for Dental Pulp Stem Cells: Emergence of a New Generation of Odontoblast-Like Cells
The dentin–pulp complex has certain intrinsic healing capacities. Reparative dentin is formed in response to external factors effecting the pulp, thus stimulating the dentinogenic progenitors that are responsible for dentin repair, contained in dental pulp (Rutherford et al. 1993, 1994; Mao et al. 2006; Yildirim et al. 2011b). The mechanisms for reparative dentinogenesis have been discussed extensively in the literature and recent studies have further enriched our understanding of dental pulp and dentin regeneration (Schroder 1985; Mullane et al. 2008; Huang 2009; Kim et al. 2010a; Mao et al. 2012). However, the relationship between different characteristics of dental pulp, substantial infection or trauma of dental pulp, and intrinsic healing of dental pulp is not well understood.
Odontoblasts, as post-mitotic cells, remain in a tooth for its lifetime, secreting secondary dentin, though their synthesizing functions decrease in time (Arana-Chavez and Massa 2004). Moreover, odontoblasts are also responsible for the secretion of a tertiary dentin in response to caries or any other mechanical or chemical problems.
Primary odontoblasts secrete dentin as secondary dentin at a very slow rate. Conditions such as tooth decay, attrition and incidental or intentional trauma and microleakage between restoration and dentin stimulate the odontoblasts and their defense and/or repair responses. Several types of injurious agents can stimulate odontoblasts: bacteria and their toxins (lipopolysaccharides and other bacterial toxins), TNFα (via p38 phosphorylation of the MAPK pathway), and the liberation of cytokines and growth factors during demineralization of the dentin matrix by caries (McLachlan et al. 2003; Durand et al. 2006; Paula-Silva et al. 2009). Once stimulated, odontoblasts enter an active state and secrete the reactionary dentin (Smith et al. 1995). However, if the nature, magnitude, or duration of the problem causes irreversible damage or the dental pulp is exposed to the oral cavity by trauma or dental procedures, primary odontoblasts die. Since the odontoblasts are post-mitotic terminally differentiated cells, they cannot proliferate to replace irreversibly injured odontoblasts. Therefore, pulpal regenerative processes take place to initiate reparative dentinogenesis and pulp progenitor/stem cells differentiate into odontoblast-like cells, which secrete reparative dentin. In this way, reactionary and reparative dentins are the constituents of tertiary dentin. While reactionary dentin is laid down by the surviving preexisting odontoblasts, reparative dentin can be produced by recruited secondary odontoblast-like cells (Smith et al. 1995).
4.2 Origin of Progenitor Cells
The suggestion that the replacement of irreversibly injured odontoblasts by predetermined odontoblastoid cells that do not replicate their DNA after induction (Höhl 1896) was rejected by auto-radiographic studies using tritiated thymidine (3HTdR or [3H]-thymidine) (Feit et al. 1970; Fitzgerald 1979). Metabolic incorporation of [3H]-thymidine into cellular DNA is still a widely used protocol to monitor rates of DNA synthesis and cell proliferation (Hu et al. 2002). By this mean, the recruitment and induction of cell populations and DNA replication cycles before differentiation into functioning odontoblasts have been shown by several researchers (Yamamura et al. 1980; Yamamura 1985; Fitzgerald et al. 1990; Tziafas and Kolokuris 1990).
Yamamura et al. suggested that:
Injury caused by partial pulpotomy capped with pure calcium hydroxide leads either to dedifferentiation of mesenchymal cells in the deep pulp into undifferentiated mesenchymal cells, which then migrate toward the wound, undergo all phases of division and subsequently redifferentiate into new odontoblasts, or to direct induction and differentiation of existing undifferentiated mesenchymal cells (Yamamura 1985).
Fitzgerald et al. showed that after pulp exposure in primate teeth, there was a significant cellular influx from pulp proper to the wound site and repair of the wound by secreting a dentin-like matrix as odontoblasts do (Fitzgerald et al. 1990). They suggested that those cells might be derived from a population of pulpal cells and perivascular tissues that reside in the deeper sites of the pulp. Additional to labeled odontoblast-like cells, they have also observed unlabeled odontoblast-like cells at the exposure site. Since “no label” indicates “no DNA replication,” the authors rationally exclude the possibility of G2-blocked, predetermined cells as a source of those unlabeled odontoblast-like cells. Instead, they suggested that those unlabeled odontoblast-like cells had replicated their DNA before the tritiated thymidine injection in those cells. Therefore, the authors concluded there is either the presence of a synchronous induction of cells that then undergo synchronous differentiation, or a synchronous induction of heterogeneous population of cells (Fitzgerald et al. 1990).
These seminal papers might give clear ideas about the presence of dental stem cells in pulp proper and obviously tritiated thymidine labeling was a smart tracer for their niche. Currently, perivascular cells (pericytes), undifferentiated mesenchymal/mesectodermal cells, fibroblasts or Höhl’s cells of sub-odontoblastic layer are proposed dental pulp progenitors. However, controversy still exists about the origin and precise identification of those progenitor or stem cells, because of the usage of different terminology (progenitor versus stem cells) and different experimental conditions that the data derived (in vivo versus in vitro). The niche concept will be discussed further in the related section.
4.3 Dental Pulp Stem Cells (DPSC)
Adult stem cells are undifferentiated cells, which divide to replenish dying cells and regenerate damaged tissues, and found in numerous tissues throughout the body (Anderson et al. 2001). To date, other than bone marrow stem cells, mesenchymal stem cells (MSC) have been identified in a variety of tissues, such as adipose tissue, peripheral blood, spleen, brain, synovial fluid, dermis, muscle, dental pulp, umbilical cord, skin, cornea, retina, liver, pancreas, and intestines (Prockop 1997; Rodriguez et al. 2005). However, there are significant differences in their proliferation and differentiation abilities, and in harvesting procedures among these MSC (Guilak et al. 2004). One of the major obstacles in stem cell research is the difficulty in isolating them due to the lack of universally accepted markers for these cells.
The well-known regenerative potential of dental pulp has indicated the existence of stem cells for decades. Finally, in 2000 Gronthos et al. isolated a population of cells that are highly proliferative, and display a hierarchy for cellular differentiation and multipotentiality (Gronthos et al. 2000). Since dental pulp is derived from mesectoderm or ectomesenchyme, it has been shown that the clonogenicity, self-renewal and proliferation capacities, expression pattern of specific markers, and multipotency of DPSC display comparative equivalence with NC cells (Sasaki et al. 2008; Kerkis and Caplan 2012).
In general DPSC show the common properties of adult stem cells. In 2007, The International Society for Cellular Therapy (ISCT) agreed that an MSC should adhere to plastic in standard culture conditions, express (≥95 %) CD105, CD73, CD90 and not express (≤2 %+) CD45, CD34, CD14 or CD11b, CD79α or CD19, HLA-DR, and finally should give at least three differentiated lineages: osteoblastic, adipogenic, and chondroblastic (which needs to be demonstrated by staining of in vitro differentiated cell cultures) (Dominici et al. 2006). Reports from the last 30 years show many examples for the “new” stem cell lineages that completely satisfy these criteria. Although DPSC have been characterized by multi-potent differentiation, the expression of some stem cell markers, dentin regeneration in vivo, and colony-forming ability in culture, they also contain heterogeneous populations of cells from the pulp (Gronthos et al. 2000, 2002; Liu et al. 2006).
DPSC can
-
Promote the proliferation and differentiation of neural cells in the hippocampus of mice (Huang et al. 2008a)
-
Prevent the progression of liver fibrosis and contribute to the restoration of liver function in rats (Ikeda et al. 2008)
-
Reduce the area of myocardial infarction, improve ventricular function, and induce the revascularization by intra-cardiac injection (Gandia et al. 2008)
-
Reconstruct the corneal epithelium in a model of total limbal stem cell deficiency (Gomes et al. 2010)
-
Form bone when combined with platelet-rich plasma or hydroxyapatite (Yamada et al. 2010)
-
Engraft and stimulate angiogenesis and vasculogenesis in models of hind limb ischemia in mice (Iohara et al. 2008)
-
Contribute to developing the embryos of mice (Siqueira da Fonseca et al. 2009)
-
Migrate, engraft, and display myogenic potential when injected into dogs (golden retriever) with muscular dystrophy (Kerkis et al. 2008)
-
Generate tooth structures (Yildirim et al. 2011a)
4.4 DPSC from Deciduous Teeth Pulp
Since exfoliation of deciduous teeth is a physiological phenomenon and every child has 20 deciduous teeth, collecting them for isolation of adult stem cells from their pulpal remnants offers an easy, noninvasive, and ethically free method. On the other hand, dental pulp of permanent teeth from elderly patient has certain limitations for stem cell isolations, since the presence of subpopulations of stem cells may be greatly restricted in aged pulp cells. Because of the deleterious effects of old niches on young cells and acquired alterations in self-antigens, stem cell regenerative capacity within aged niches has been questioned (Hasler and Zouali 2005; Carlson and Conboy 2007; O’Connor et al. 2009).
As a matter of fact, stem cells from human exfoliated deciduous tooth pulp has been isolated and named as SHED (Stem cells from Human Exfoliated Deciduous teeth) in 2003 (Miura et al. 2003). They are highly proliferative postnatal stem cells capable of differentiating into odontoblasts, adipocytes, neural cells, and osteo-inductive cells (Miura et al. 2003). SHED was shown to be capable of generating a tissue that closely resembles human dental pulp (Cordeiro et al. 2008). It has been reported recently that SHED contains a higher proportion of the side population than DPSC (Wang et al. 2010). Moreover SHED have the potential to differentiate into functional vascular endothelial cells (Cordeiro et al. 2008; Sakai et al. 2010), can promote bone formation in immunocompromised mice (Miura et al. 2003), and give better osteogenic response to retinoic acid treatment than periodontal ligament stem cells (Chadipiralla et al. 2010).
Although some minor discrepancies regarding cell surface antigen profiles, and differentiation potentials into osteogenic, adipogenic, and chondrogenic lineages have been reported so far (Huang et al. 2009; Nakamura et al. 2009; Govindasamy et al. 2010; Kerkis and Caplan 2012; Wang et al. 2012), the main differences between SHED and DPSC are in their proliferation rate and colony-forming properties, which are higher in SHED than DPSC (in parallel, the doubling time of SHED is shorter than DPSC) (Nakamura et al. 2009; Govindasamy et al. 2010; Wang et al. 2012). The other difference is the expression of pluripotency markers, which are again higher and more abundant in SHED. Many researchers have reported that SHED is a more primitive, pluripotent population of cells than DPSC, bone marrow-MSC and umbilical cord stromal stem cells (UCSC) (Kerkis et al. 2006; Nakamura et al. 2009; Govindasamy et al. 2010; Chen et al. 2012). On the other hand, it has been proposed that DPSC have a higher propensity toward neural lineage, expressing significantly higher rates than SHED (Govindasamy et al. 2010).
4.4.1 The Degree of Resorption of the Teeth Related to Stem Cell Isolation
The exfoliation of deciduous teeth is a physiologic phenomenon. Histological changes associated with the process of shedding in human beings have been well documented, and it has been shown that pulp tissue structure maintains its integrity till the late phase of exfoliation (Sahara et al. 1993; Sasaki 2003). Although there is an increase in the numbers of inflammatory cells in the active resorption area around the root(s) as well as mononuclear cells in the subodontoblastic area, regular odontoblasts still exist along the dentin walls (Fig. 4.1).
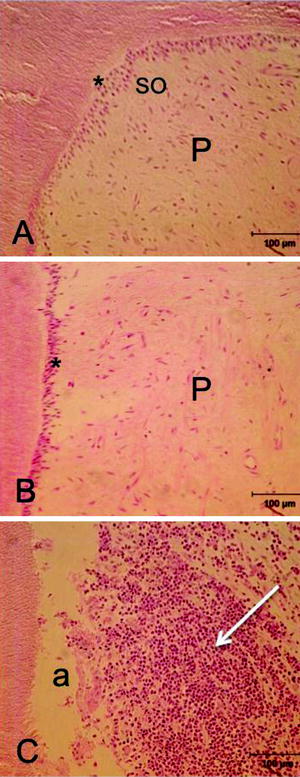

Fig. 4.1
The histological sections show healthy pulp (P) and firm layers of odontoblasts (asterisk) and subodontoblasts in coronal (a) and servical (b) portions, although numerous inflammatory cells invaded the resorbed root (c) (a artificial detachment, H&E, bars 100 μm)
When roots are resorbed completely, odontoclasts appear in the coronal pulp (Fig. 4.2a, b) and they start to resorb predentin. In that phase coronal pulp contains many mononuclear and inflammatory cells (Fig. 4.2c). TRAP-positive mononuclear cells (presumed to be preodontoclasts) (Sahara 2001) can be seen on predentin where they fused to form TRAP-positive multinucleate odontoclasts that resorbed the root (Fig. 4.3).
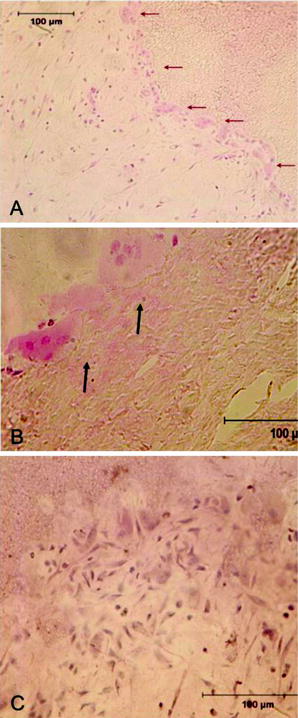
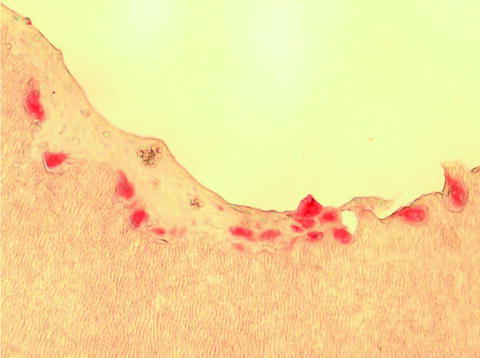

Fig. 4.2
In the final phase of resorption odontoclasts occur in the coronal dental pulp and start to resorb predentin (arrows in a and b). In that phase coronal pulp has more heterogeneous population including many mononuclear cells (H&E, bars 100 μm) (c)

Fig. 4.3
Dark TRAP-positive multinucleate odontoclasts that reside in the lacunae on predentin (TRAP staining, 400×)
When the resorption is completed, only minute amounts of epithelium and periodontal ligament can retain the tooth in the dental arch and resorbed sharp edges may cause pain during biting and eating. In the routine practice of pediatric dentistry we rarely extract resorbed deciduous teeth, unless the patient wants relief from the pain. In that case, with little regional or only topical anesthesia, the resorbed tooth can be separated from the periodontal ligament easily, and hence be extracted. Likewise, deciduous teeth are shed with little bleeding naturally, since the final shedding would occur by the tearing of the narrow tissue bridges by migrated gingival epithelium under the crown. In this final stage of resorption, there are only crowns, which their pulp chambers invaded by the oral epithelium (Fig. 4.4) (Sahara et al. 1993).
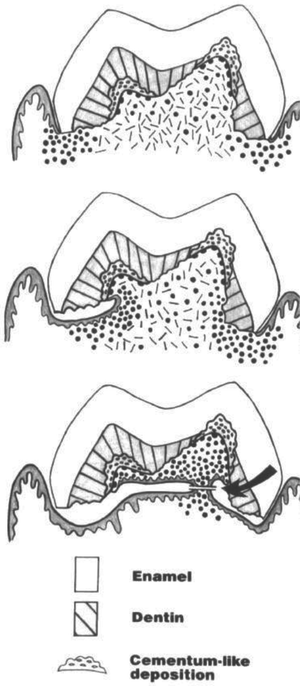

Fig. 4.4
Schematic representation of migration of the dento-gingival junction (DGJ) epithelium and gingival epithelium in the process of exfoliation of a human deciduous tooth. After the roots are completely resorbed, the DGJ epithelium gradually migrates toward the resorbed surface, and finally reaches the surface of the pulp chamber wall. Accompanying this migration, the gingival epithelium also proliferates and migrates toward the resorbed regions, and eventually is present under the crown. Therefore, the migrated gingival epithelium makes narrow tissue necks under the crown. The final shedding would occur by the tearing of these narrow tissue bridges (arrow) (reproduced from Sahara et al. 1993 with permission of the publisher)
Stem cell properties of deciduous dental pulp cells have been compared with the cells obtained from exfoliated and orthodontically extracted no-resorption teeth (Bernardi et al. 2011), and it has been reported that it was not possible to establish cell cultures from the teeth that did not show any visible resorption. Zhu et al. broadened the classification of resorbing teeth samples as stable, middle, and final stages and compared those samples with the non-resorbed deciduous teeth. They found that the stem cells obtained from the final resorption stage group were equal to SHED, whereas stem cells obtained from middle stage group exhibited higher proliferation potential than those in the final stage group, yet much higher than those of the stem cells from stable resorption group (Zhu et al. 2012).
Exciting possibilities exist despite the available pulp tissue in the exfoliated deciduous tooth being extremely small for stem cell isolation. The remnants of the pulp reside only in the pulp chamber as seen by dashed lines on the crown in Fig. 4.5, and it may occupy approximately 9–13 mm3 in an upper incisor (if we consider the pulp chamber as being cylindrical with a radius of approximately 1 mm and a height between 3 and 4 mm). Since the pulp is a loose connective tissue, the weight of the tissue can be estimated. In order to compare stem cell properties of two different stromal tissues (human umbilical cord stroma and deciduous dental pulp), we isolated mesenchymal cells from 15 cm-long cords and four exfoliated deciduous teeth. At the end of P0 (14 days) overall yield (106 cells) was the same in T25 cm2 flasks for both cell populations (Oktar et al. 2011).
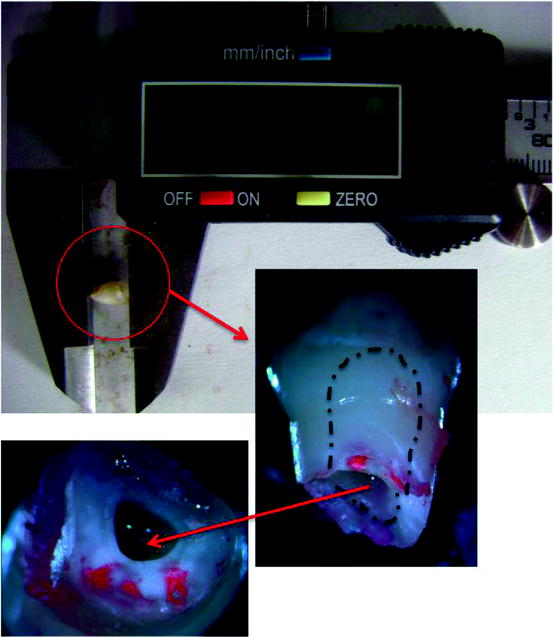

Fig. 4.5
An exfoliated deciduous tooth can be measured approximately as 7.00 mm including the crown. Pulp tissue resides in the coronal part. Dashed lines show the dimensions of pulp chamber
Continuing the discussion about the increase of inflammatory molecules during resorption (Angelova et al. 2004; Simsek and Duruturk 2005), it has been suggested that cellular influx could promote the release of cytokines acting in the activating stem cells during resorption and the increased proliferation and mineralization potential of SHED would be a response to active resorption (Bernardi et al. 2011). It has been hypothesized that SHED may promote osteoclastogenesis, during the active resorption process via several cytokines including the RANKL/OPG duo, and this function may subside in the final stage, thereby protecting the teeth from excessive resorption (Yildirim et al. 2008; Bernardi et al. 2011; Zhu et al. 2012).
4.5 Niche(s) in Dental Pulp
In healthy tissues, stem cells are found in their specific niche environment. The stem cell niche concept is coming from earlier work on the relationship between spleen colony forming cells and hematopoietic stem cells. Schofield proposed in 1978 that the niche is a physiologically supportive microenvironment for stem cells (Schofield 1978). Voog and Jones have defined the stem cell niche as follows:
-
Stem cell niches are discrete and dynamic functional domains that influence stem cell behavior to govern tissue homeostasis under diverse physiological (development and aging) and pathological (injury and disease) conditions. The niche must be flexible in order to coordinate stem cell behavior with homeostasis and repair; however, the plasticity of a niche may be co-opted in cancer and chronic disease.
-
The idea that specialized environments within tissues can preserve proliferative potential and block maturation of adult stem cells was the first description of the stem cell niche hypothesis. Implicit in this model is the prediction that removal of stem cells from the niche results in loss of stem cell identity, self-renewal capacity, and the onset of differentiation. As such, the niche would provide a mechanism to precisely balance the production of stem cells and progenitor cells to maintain tissue homeostasis. Therefore, a stem cell niche is not defined solely by the presence of stem cells but also by the ability to regulate stem cell behavior (Voog and Jones 2010).
Stay updated, free dental videos. Join our Telegram channel

VIDEdental - Online dental courses


