Fig 8.1.
Light micrograph of a ground section of an adult human canine. Enamel (E), dentin (D) and cementum (C) at cervical tooth situation
Cementum is a specialized connective tissue covering the outermost layer of calcified matrix on root surface, with a primary role to connect the periodontal ligament to the root surface. Cementum has not been largely encountered as a substrate from the bonding point of view, despite the fact that cervical microleakage has been early recognized to contribute to high incidence of secondary caries and fail of restorations [1, 2]. Very few studies have evaluated cementum-bonding restorations and the techniques used in present clinical practice in terms of their effectiveness and predictability [2 – 6].
A systematic approach to bonding at the cervical margins of class-II and class- V restorations requires understanding of anatomy and histo-physiology of the root dental structures.
This chapter reviews the main structural, compositional, and functional aspects of cementum, and its primary evaluation as a bonding substrate.
Developmental and Structural Aspects
Cementum Varieties and Their Locations
Three varieties of cementum cover the root of the human tooth: acellular afibrillar cementum; acellular extrinsic fiber cementum, and cellular mixed-stratified cementum. Their location and structural characteristics are associated with their different functional identity [7 – 10].
Acellular afibrillar cementum. Acellular afibrillar cementum is located mainly around the cemento-enamel junction covering further small enamel areas. Its distribution pattern varies from tooth to tooth and along the cemento-enamel junction of the same tooth [9]. By light and electron microscopy acellular afibrillar cementum represents a non-homogenous matrix composed of multiple calcified layers without collagen fibers [11].
Acellular extrinsic fiber cementum. The acellular extrinsic fiber cementum constitutes the majority of the cementum. It represents a thin layer of mineralized tissue located along the coronal two-thirds of the root surface (Fig. 8.2a, b). In anterior teeth acellular extrinsic fiber cementum is further extended to the apical root third. The formation of this type of cementum (Fig. 8.2 c) is a continuous process characterized by an extremely slow rate of new matrix deposition [12]; thus, it reaches a thickness of approximately 15 μm during the prefunctional stage of root development, and a total thickness of approximately 50 μm during the functional stage throughout the life of the tooth [13, 14]. The periodic deposition of this type of cementum is seen in light microscopy as multiple layers of matrix interspersed by growth lines.

Fig 8.2
a-e. Light micrographs of the main types of cementum. a Demineralized section of adult dog molar. Acellular extrinsic fiber cementum. b Ground section of adult human premolar. Acellular extrinsic fiber cementum. Note the dentino-cemental junction. c Demineralized section of developing dog molar. Cuboidal-shaped cementoblasts forming acellular extrinsic fiber cementum. d Apical situation from the section seen in a. Cellular mixed stratified cementum. e Apical situation from the section seen in d. Cellular mixed stratified cementum with numerous cementocyte lacunae
Cellular mixed stratified cementum. Cellular mixed stratified cementum is composed of multiple interposed layers of acellular extrinsic fiber cementum and cellular intrinsic fiber cementum (Fig. 8.2d, e), which is mainly deposited on the apical root surface area and in the furcation areas. In fact, pure cellular intrinsic fiber cementum is only deposited as a part of the reparative process following root resorption [15]. The appositional growth of cellular cementum is much faster that acellular cementum, reaching a mean thickness of more than 200 μm [16]. In adult teeth, impacted teeth, and in teeth without antagonists, extremely thick layers of cellular cementum could be found around the apical third of their roots [9, 17].
The primary role of acellular extrinsic fiber cementum is to attach periodontal ligament fibers to the root surface, supporting tooth mobility and function. Cellular cementum has important functional role in maintaining the occlusal relationship of the tooth and repairing the resorbed root surface areas. Furthermore, acellular afibrillar cementum seems to have only adaptive function.
Cementum Formation
Cellular and molecular mechanisms, which regulate formation of periodontal tissues, including cementum, have not been fully understood. It is well known that dental follicle cells of ectomesenchymal origin are capable of forming cementum when properly stimulated [18 – 20]. As to the nature of the cementum-forming cells (cementoblasts), there is a widely accepted opinion that they represent a population of periodontal cells [9, 21], although some authors suggest that epithelial cells may also contribute to the process of cementum formation [22 – 24].
The differentiation of dental follicle cells into cementoblasts is closely related to root dentin formation at a band extending 200-300 μm coronally to the developing root edge [9]. Progenitor cells migrate towards the newly deposited, but not yet mineralized, dentin matrix, as the Hertwig’s root sheath breaks down. Numerous cytoplasmic processes are interposed between the dentinal collagen fibers, forming the dentino-cemental junction [14]. A layer of new matrix (precementum or cementoid) is deposited onto the external surface of the dentin by the surface root lining cells in a polar pattern. Recently, Nociti et al. [20] reported that the levels of inorganic pyrophosphate within the extracellular matrix of periodontal ligament is one of the crucial factors that regulate both initiation and maturation of cementum. Early mineralization of precementum into acellular extrinsic fiber cementum by deposition of hydroxyapatite crystals initially between and then within the collagen fibers, before the complete mineralization of the mantle root dentin, is usually found [25, 26]. At the apical third, highly active cementum-forming cells [12] deposit matrix in a non-polar bone-like pattern (cellular cementum). Cells entrapped into cellular cementum are called cementocytes. In mature teeth acellular cementum is not covered by precementum, a 3- to 5-μm-width layer which is usually seen along the cellular cementum [27, 28].
The formation of cementum is characterized by apposition throughout the functional stage of the tooth life. Since the collagen fibers of the periodontal ligament undergo remodeling, acellular cementum formation providing attachment to Sharpey’s fibers is a continuous process. New cementoblasts seem to be continuously recruited from the periodontal ligament pool to replace destroyed cementoblasts or cementoblasts that have reached the end of their life span [9]; however, it is not generally agreed that a specialized cell type exists in the periodontal ligament for the continuous cementum formation. Whether cementoblasts-forming acellular extrinsic fiber cementum are phenotypically different from the periodontal fibroblasts, or further cementoblasts-forming cellular cementum are specific cells or “osteoblast variants,” is still not known [30].
The root surface covered by the acellular extrinsic fiber cementum is an area at risk of carious demineralization or invasion by plaque bacteria. Regeneration of cervical dental structures is of clinical importance, and it seems challenging to devise new therapeutic methods enhancing cementum regeneration in affected root surfaces. More particularly, with regard to restorative strategies, novel biomaterials aiming to induce controlled formation of acellular extrinsic fiber cementum might be designed.
Structural Characteristics of Acellular Extrinsic Fiber Cementum
Acellular extrinsic fiber cementum is not a homogenously calcified tissue (Fig. 8.3). Cementum is characterized by amorphous mineralized microstructures and a dense network of unmineralized Sharpey’s fibers, oriented perpendicularly to the root dentin. At the onset of cementum formation these fibers are extremely short, whereas in mature teeth they are continuous with the long periodontal ligament fibers. These fiber types are clearly distinguishable from a small number of intrinsic collagen fibers, which are also found in acellular cementum. Hydroxyapatite crystals, perpendicularly deposited to the root surface, between or within the collagen fibers, form crystals of 1-4 μm in length over a long period of time [27, 31]. In light microscopy acellular extrinsic fiber cementum is characterized by the presence of incremental lines, oriented parallel to the root surface.
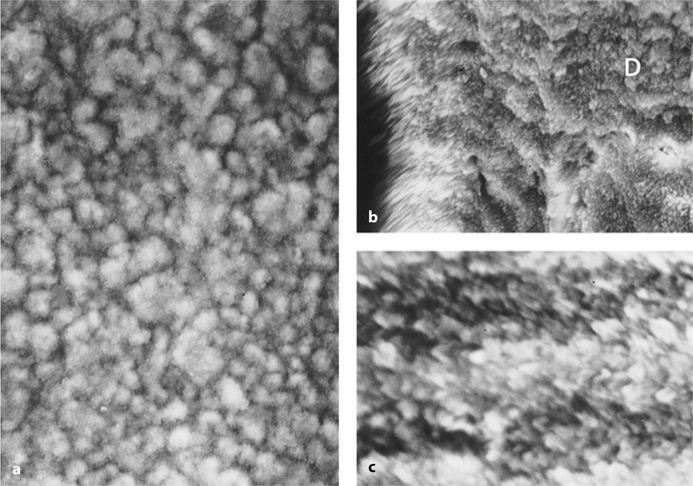
Fig 8.3a-c.
Scanning electron micrographs of adult human acellular extrinsic fiber cementum at cervical tooth situation. a Root surface. The non-homogenously calcified structure of cementum. b Longitudinal section from the same root area. The uncalcified core of Sharpey’s fiber oriented perpendicularly to the root dentin (D). c Higher magnification of b. Atypical organization of apatite crystals
The Cemento-Enamel and Dentino-Cemental Junctions
The junction between cementum and enamel exhibits three normal variations:
1.
Cementum overlaps the cervical border of enamel in more than 60% of the human teeth.
2.
Cementum meets directly the enamel in approximately 30% of the human teeth.
3.
In < 10%, a gap (exposing the dentin surface) exists between enamel and cementum.
The dentino-cemental junction is developed as a result of the multiple events occurring during the early stages of root formation, i.e., root dentinogenesis, cementum formation, epithelial root sheath disorganization, and periodontal ligament formation. Cementum-forming cells differentiating along the newly deposited predentin produce collagen fibers which are integrated between the collagen fibers of the predentin matrix [12]. Two structures have been associated with the dentino-cemental junction:
1.
The intermediate cementum. Some authors have reported the presence of a glycoprotein-rich matrix, or an enamel-like material at the interface between root dentin and cementum. This 10- to 20-μm-thick layer, characterized as hyaline zone in rodent teeth, is formed before cementum deposition, and it has been associated with cementum attachment to root dentin. Its existence in human teeth has been reported only for the apical portion of the root [32]. The presence of this collagen-free matrix at the junction between acellular cementum and root dentin and its origin is controversial [33, 34]. Thomas [12] stated that the junction is formed as a slow accretion of mineral on the root dentin surface, rather than the accumulation of minerals within a preformed collagenous matrix.
2.
Tomes’ granular layer. This layer has been described as a number of dark granules in the periphery of the root dentin underlying the cementum in ground sections with transmitted light. No collagenous matrix was found in this granular layer. Most authors believe that it represents a structure caused by the looping of the dentinal tubules in that region [35]. While peripheral dentin in the crown contains multiply branched dentinal tubules, the peripheral root dentin is atubular. Only in the subjacent granular area are dentinal tubules localized, but in a somewhat chaotic manner [36].
Biochemistry of Mature Cementum
Cementum represents the less characterized mineralized tissue. Similarly to the other connective calcified tissues, approximately 40% of extracellular organic matrix and 50-60% of hydroxyapatite crystals are composed of water [35]. In general, the two major types of cementum (acellular extrinsic fiber cementum and cellular mixed stratified cementum) are similar to bone, with three crucial exceptions:
Extracellular Matrix
The extracellular matrix of human cementum consists mainly of collagen type I (90% of the organic matrix) and collagen type III (5%) according to the classic study by Cristoffersen and Landis [40]. In all connective tissues collagen molecules play important structural roles, providing the functional tissue scaffold.
Furthermore, a number of non-collagenous cell attachment proteins, with only partially characterized properties, have been detected in cementum [29]:
1.
Fibronectin, osteopontin, and osteocalcin with possible role in cementum development and regeneration.
2.
Vitronectin, an adhesion molecule found in high concentration in serum with unknown function.
3.
Cementum attachment protein (and possibly other yet unidentified adhesion molecules) and cementum growth factor are tissue-specific molecules, although some homology to known bone proteins has been reported [38, 39]. Cementum attachment protein binds with fibronectin and hydroxyapatite but not with collagen [41]. It supports the attachment of mesenchymal cells, but not of epithelial cells [41].
4.
Enamel proteins have been detected in cementoblasts, but their presence in cementum extracellular matrix has not yet been demonstrated [9].
5.
Alkaline phosphatase, playing a role in precipitation of calcium phosphate salts, is at least partially found in a collagen-bound form [42].
Inorganic Component
The inorganic component of cementum is the same as in other calcified tissues, bone, dentin, and enamel. Chemical analysis of the cementum inorganic phase has shown that the primary mineral component is hydroxyapatite [Ca10(PO4)6(OH)2] containing amounts of amorphous calcium phosphate. In transmission electron micrographs hydroxyapatite crystals are found mainly between the collagen fibrils, normally arranged with their c-axis parallel to the long axis of collagen fibrils [27].
The crystallinity of the cemental inorganic component is lower than the other calcified tissues [9]. As a result, cementum is decalcified more easily, while it has a greater affinity for adsorption of environmental ions (i.e., fluoride). In general, cementum of adult mature teeth has a higher fluoride content in comparison with the other calcified tissues [43 – 45]. The Mg content in cementum is about half that in dentin. There is a gradual increase of Mg present in the cementum from periphery to deeper layers [43, 46]. Trace elemental concentrations of Cu, Zn, and Na were further detected by electron microprobe analysis in human root cementum of healthy and periodontally involved teeth [47].
Cementum in Health and Disease
Root Permeability
The physiology of root-calcified tissue system has not been studied systematically. With regard to permeability of cementum in health and disease, contrary data have been reported. In general, the cemental matrix is porous allowing only permeation of water and inorganic ions under physiological tissue conditions. Nevertheless, the structure of diseased root surface is also permeable to saliva organic components and plaque bacterial byproducts.
In periodontally involved teeth, plaque bacterial products have been detected in the 10- to 12-μm-deep surface layer [48]. Furthermore, Lygre et al. [49] found bacterial lipopolysaccharide at a distance of 70 μm from the surface of the periodontally diseased roots. Penetration of substances derived from saliva to the exposed root cementum might be attributed mainly to numerous cracks and fractures, which are developed at the non-supported areas with cells and fiber root surface [9]. Invasion of bacteria into cemental matrix has been demonstrated in chronic periodontal pathosis [48, 50]. Infected cementum has been characterized as a reservoir of periodontopathogenic bacteria [50].
Age Changes in Cementum
Cementum grows appositionally throughout life at a linear rate [53], although the variations in width of incremental lines indicate that the rate of cementum deposition varies from period to period. Generally, cementum increases in thickness by different growth patterns among types of cementum being formed. Its thickness varies greatly with tooth group, tooth surface area, and cervical/apical root positions [54]. Cementum, like bone, is a dynamic tissue, capable of responding to occlusal forces and physiological tooth movement. The greatest amount of cementum is deposited apically, constricting the apical foramen, and in the furcations of multi-rooted teeth. According to Schroeder [7], cementum is thicker in areas exposed to tensional forces, whereas Dastmalchi et al. [55] found that cementum thickness is increased more rapidly at the distal than the mesial parts of root surface.
Root Surface Caries and Cementum
The increased number of teeth in older individuals, due to caries preventive strategies, resulted in higher prevalence of root caries during the past two decades. Root caries occurs on exposed tooth surfaces below the cemento-enamel junction after gingival recession. Cariogenic plaque, rather than periodontitis or gingival inflammation, is the essential factor responsible for root caries development. The different forms of root surface caries, ranging from minor undemineralized and discolored areas to extensive yellow-brown soft areas, are rarely associated with cavitation below the affected cementum [56, 57].
The histopathology of root caries is similar to that of enamel carious lesions. The early root caries lesion appears as a zone of demineralization deep to the root surface. The radiolucent area is usually detected below a well-mineralized layer of acellular extrinsic fiber cementum, which varies in thickness and mineral contents. Nyvad et al. [58] described in an experimentally induced root caries model the initial histopathological changes seen after covering of exposed cementum with plaque. Within 1-3 months a continuous subsurface loss of mineral was associated with redeposition of minerals in the surface cementum layer which appeared hypermineralized when compared with the neighboring cementum. Larger apatite crystals were found in the root surface of the caries-affected cementum [59]. Bacteria infiltrating the lesion split the collagen fibers of the cementum and were frequently detected at the dentino-cementum junction [60, 61]. Bacteria were also identified within the subjacent dentinal tubules during early root caries development [62]. As in enamel, caries active and arrested or slowly progressive lesions may be seen in cementum. Active root surface lesion presents a well-defined area of softening and yellowish discoloration. Inactive root surface lesion appears hard on probing with dark discoloration. Transitory stages between active and arrested root surface caries cannot be excluded.
Changes of Cementum in Periodontally Involved Teeth
In periodontally involved teeth, the exposure of acellular extrinsic fiber cementum to the environment of oral cavity or periodontal pocket creates constantly changes in the surface structure and composition of cemental matrix and eventually diseases of the root calcified tissues, such as development of cementum caries and cervical root resorption.
Structural modifications of the cementum surface are characterized mainly by loss of collagen cross-banding, breakdown of dentogingival fibers, and dissolution of mineral components [63, 64]. Bosshardt and Selvig [9] state that these surfacelimited modifications transitionally extent to the subsurface tissue area and can be described only by electron microscopy. It has been reported that surface fractures and cracks are also developed at the exposed cementum.
It has been repeatedly demonstrated by chemical [45, 66], microradiographic [58, 64, 67, 68], and SEM-microprobe [43] investigations that the exposed cementum surface is progressively hypermineralized, although Barton and Van Swol [47] and Cohen et al. [69] did not detect such a hypermineralized zone. Furseth [70] found that this zone can be experimentally created 21 days after a gingivectomy procedure, whereas it can be re-established 4-8 weeks after removal of the hypermineralized cementum surface by root planning [64]. The cementum hypermineralization is related to the loss of cross-banding after chronic exposure of root surface to the oral environment [43, 71]. Atypical development and orientation of large apatite crystals characterize the surface structure of hypermineralized cementum in an SEM. The atypical distribution of mineral components and the increased fluoride concentration in this zone may explain the resistance of hypermineralized cementum to acid demineralization [68].
In periodontally involved teeth, especially in cases of hyperplastic gingivitis, large resorptive defects in the cervical region of vital teeth are usually found. This phenomenon is called cervical root resorption or idiopathic root resorption, and its nature has not been adequately elucidated [37, 72
Stay updated, free dental videos. Join our Telegram channel

VIDEdental - Online dental courses


