Figure 12.1. Anatomy of the normal palate. Reprinted with permission from Elsevier in Ref. 20, p. 273. Copyright © 2009 Elsevier.
The middle 50% of the velum also contains the insertion of two additional muscles, which extend posteriorly to become the anterior and posterior tonsillar pillars. The anterior tonsillar pillar is the palatoglossus muscle, which originates from the dorsolateral transverse fibers of the tongue and inserts as the most superficial muscle of the base of the soft palate. The posterior tonsillar pillar, the palatopharyngeus muscle, has two origins: an inferior head on the oral surface of the intravelar levator muscle and a superior head on the nasal aspect of the intravelar levator muscle. The palatopharyngeus extends inferiorly toward the larynx, bounded by the posterior pharyngeal walls. It plays a role in elevating the pharynx over the bolus of food when swallowing.
The posterior 25% of the velum contains the remainder of the palatoglossus and palatopharyngeus, as well as the muscularis uvulae. The latter originates from the tensor aponeurosis anteriorly and proceeds as a paired midline structure to the base of the uvula posteriorly. This muscle controls the shape of the uvula. The anterior 25% of the velum contains the aponeurosis of the tensor veli palatini muscle. The tensor veli palatini muscle takes its origin from the scaphoid fossa of the greater wing of the sphenoid and the length of the superolateral aspect of the eustachian tube. The muscle descends almost vertically to hook around the anterior aspect of the hamulus and continues medially as an aponeurosis, spreading across the midline to join its contralateral mate and forming a fibrous band posterior to the hard palate but anterior to the levator veli palatini sling.
The superior constrictor originates from the posterior pharyngeal raphe and courses inferiorly and anteriorly to insert into the pterygoid hamulus, lateral pterygoid plate, pterygomaxillary ligament, mandible, and floor of the mouth. It may play some role in constricting the posterior pharynx, thus contributing to velopharyngeal function.
The palate is vascularized by two sets of neurovascular bundles, anteriorly the incisive and posteriorly the greater palatine. These vessels do not cross the midline extensively, leaving each half of the palate reliant on its own vasculature. The palate soft tissues extend posteriorly to the soft palate where they cover the functional muscles of the velum.
A cleft of the palate predictably alters the normal functional sling of the paired levator veli palatini (Figure 12.2). The clefted levator veli palatini inserts longitudinally onto the posterior edge of the clefted hard palate, never transversely joining the contralateral muscle. Therefore, a functional levator sling does not exist. Three abnormal attachments or associations of each levator veli palatini muscle have been described and can easily be seen during a cleft palate repair: (1) the posterior medial edge of the hard palate, (2) the aponeurosis of the similarly interrupted tensor veli palatini, and (3) the superior pharyngeal constrictor (Figure 12.3).13,14 The functional reconstruction of the levator sling therefore requires the complete release of the levator muscles from the three abnormal attachments in order to reposition them anatomically into a transverse position within the middle 50% of the velum.15
The tensor and levator veli palatini muscles are both associated with the eustachian tube, and are equally affected in the cleft palate anomaly. Classically, the levator elevates the palate, while the tensor tenses, or contracts, to dilate the eustachian tube; however, it is likely that these muscles work synergistically to dilate, or at least modulate, the eustachian tube. The tensor may draw on the lateral aspect to dilate the tubal lumen, and the levator may rotate the lip of the tubal cartilage to maximize the vector of the tensor’s contraction.16 As explained by Huang et al.,11 the paratubal muscles have abnormal insertions in the cleft palate pathology, and therefore their actions as tubal dilators are likely compromised. Patients with cleft palate have eustachian tube dysfunction resulting in chronic effusions and frequent ear infections. Eustachian tube dysfunction secondary to cleft palate can lead to hearing loss, and is regularly treated with ear tube placement.
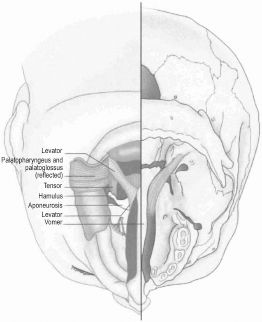
Figure 12.2. Anatomy of the cleft palate. Reprinted with permission from Elsevier in Ref. 20, p. 274. Copyright © 2009 Elsevier.
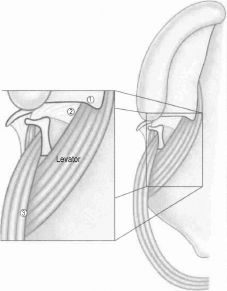
Figure 12.3. The clefted levator veli palatini muscle has three abnormal associations: (1) the hard palate, (2) the tensor aponeurosis of the tensor veli palatini, and (3) the superior pharayngeal constrictor. Reprinted with permission from Elsevier in Ref. 20, p. 275. Copyright © 2009 Elsevier.
12.3 Classification
There are many classifications published for CL ± P. The most useful capture the pathologic anatomy concisely to help summarize the diagnosis and the treatment. Two of the simplest and most commonly used are the Veau17 and the LAHSHAL18 classifications.
The Veau system specifically classifies cleft palate and is intuitively based on anatomy (Figure 12.4). It classifies the four subtypes of cleft palate as follows: I is a cleft of the soft palate only, II is a cleft of the soft and part of the hard palate, III is a complete unilateral cleft of the palate and alveolus, and IV is a complete bilateral cleft palate and alveolus. Frequently, the Veau III and IV clefts include clefts of the lip as well.
The LAHSHAL system is an acronym, using four initials to describe where the clefts occur, as follows: “L” is lip, “A” is alveolus, “H” is hard palate, and “S” is soft palate.18 The initials are repeated to denote the right and left sides. A capital letter denotes a complete cleft, a lowercase letter denotes an incomplete cleft, and an “X” denotes a normal structure. Therefore, XXXSHAL would represent a complete left-sided lip and palate, and lXXsXXX would denote an incomplete right-sided cleft lip with a Veau I cleft of the velum.
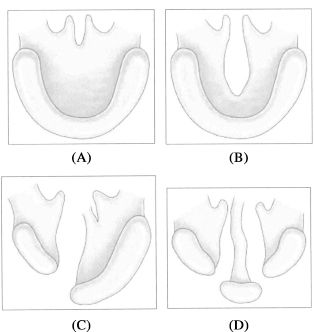
Figure 12.4. Veau classification of palates. (A) Veau I, cleft of the soft palate. (B) Veau II, cleft of the soft and part of the hard palate. (C) Veau III, complete unilateral cleft of the soft and hard palates and alveolus. (D) Veau IV, complete bilateral cleft of the soft and hard palates and alveolus. Reprinted with permission from Elsevier in Ref. 20, p. 276. Copyright © 2009 Elsevier.
An SMCP occurs when the velar muscles are clefted and mis-oriented, as in the overt cleft palate. However, the overlying mucosa of the soft palate is intact; therefore, the clefting is submucosal and hidden from view. The degree of levator clefting and disorganization is variable, as it is understood that not all SMCPs result in velopharyngeal dysfunction. When complete levator muscle clefting occurs, three cardinal signs may appear: (1) midline notching of the posterior edge of the hard palate, (2) midline translucent blue zona pellucida where the muscles are missing, and (3) a bifid uvula. The three classic signs are actually not commonly present; the Children’s Hospital of Pittsburgh series demonstrated that only 29% of patients diagnosed with SMCP possessed all three signs, 34% had any two signs, 17% had only bifid uvula, 14% had only zona pellucida, and 6% had only notched hard palate.19 Far more clinically practical is the abnormal function of the velum as it can be seen elevating in a “vaulted” V-shaped pattern, indicative of abnormal sagittally oriented levator muscles. Therefore, SMCP is a diagnosis of clinical suspicion on the basis of a physical examination, speech evaluation, and frequently a history of recurrent ear infections. Pure idiopathic velopharyngeal insufficiency, with no signs of SMCP, can be found to have “occult SMCP” on confirmation of the diagnosis intraoperatively.20
12.4 Treatment Goals and Preoperative Workup
An infant with orofacial clefting should be seen by a cleft-craniofacial team trained in the unique comprehensive care that will be required. Early evaluation by a qualified feeding specialist (speech language pathologist or occupational therapist) and plastic surgeon will address and assure airway safety and successful feeding. Feeding specialists often assist in optimizing nutrition, with customized bottles that allow easier fluid egress and in positioning the infant in a more upright position to allow for gravity-assisted feeding.21 We prefer having the infant able to drink from a sippy cup and completely weaned from a bottle before cleft palate repair, as the nipple could potentially injure the repair postoperatively.20
Timing of cleft repair depends upon several factors including the size and shape of the mandible, the infant’s airway, the nature of the palatal cleft, and the development of speech. There is debate that early cleft palate surgery may impair maxillary growth, whereas late cleft palate surgery may lead to worse speech problems. As normal speech development occurs at about 1 year of age, in general, a cleft palate repair is timed at approximately 12 months of age, although this may be individualized depending on the patient. Children with other medical conditions or syndromes may require additional perioperative evaluation.
An important subset of infants with cleft palate are those with Pierre Robin sequence, characterized by a retrognathic mandible leading to posterior displacement of the tongue (glossoptosis) and airway compromise. These patients often have a wide, U-shaped Veau II cleft. Infants with Pierre Robin sequence may require preparatory airway correction (tongue–lip adhesion or mandibular distraction) before cleft palate repair. Another important subset of patients with cleft palate are those with velocardiofacial syndrome, or 22q11.2 deletion, also known as Shprintzen or DiGeorge syndrome. These patients may have cardiac anomalies, immunoincompetence, hypotonia, or other manifestations that require intervention.22
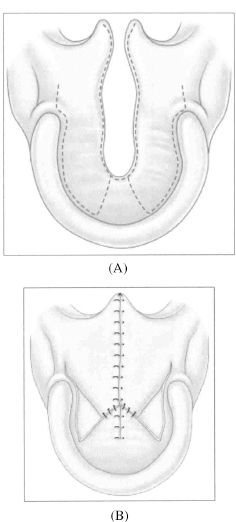
Figure 12.5. In a Veau II cleft of the soft and hard palates, unipedicled flaps can be designed to be elevated (A) and set back in a V-Y pushback manner that closes the cleft as well as potentially lengthening the palate (B). Reprinted with permission from Elsevier in Ref. 20, p. 289. Copyright © 2009 Elsevier.
12.5 Primary Repair of the Soft Palate
The earliest soft palate repairs by Von Graefe and Roux in the early 19th century23 simply reapproximated the separated soft palate, and did not specifically try to reconstruct the muscle. The realization that the length of the palate plays a potential role in velopharyngeal competence did not come until Veau, Wardill, and Kilner designed repairs that “pushed back” the hard palate flaps, potentially adding length to the soft palate23 (Figure 12.5). It was not until 1969 that Kriens specifically repaired the levator sling to reconstruct the abnormal anatomy of the muscular sling,23 calling his repair an intravelar veloplasty (IVVP). The options for velar repair today include straight-line or Z-plasty closure with or without an IVVP.
12.5.1 Intravelar veloplasty
Most modern-day palatoplasties include an attempt at repairing the levator veli palatini muscle sling. However, not all IVVPs are the same, and it is important to distinguish between an “incomplete” IVVP and a “complete” IVVP. An incomplete IVVP occurs when only the most medial ends of the pathologically oriented levator muscles are dissected free and repaired. This unfortunately results in most of the levator muscle remaining in a sagittal orientation and the reapproximated medial ends of the levator muscle positioned anteriorly along the posterior edge of the hard palate. Without completely detaching the levator from its three abnormal attachments (Figure 12.3) and radically repositioning them into an anatomic transverse sling in the middle 50% of the velum, a complete IVVP cannot be performed.15,24 It is important to identify and release the clefted levator from (1) the medial aspect of the posterior hard palate, (2) the aponeurosis of the tensor veli palatini, and (3) the superior constrictor laterally.
12.5.2 Z-plasty velar repair
In the late 1980s, Furlow realized that he could reroute the anomalous sagittal levator veli palatini into its proper transverse orientation.25 Using double-opposing Z-plasties, two flaps of oral lining and two flaps of nasal lining are created. The posteriorly based oral and nasal flaps each contain their respective levator muscle, which is subsequently transferred with the Z-plasty flap into the middle 50% of the velum. As well, when the muscle is transferred on a Z-plasty flap, it eliminates the need to completely dissect both surfaces of the muscle, thereby potentially reducing scarring. The levator muscles are overlapped, being placed on “functional tension,” and the transverse narrowing effect of the Z-plasty produces a secondary pharyngoplasty effect by narrowing the size of the velopharyngeal port. Similarly, the Z-plasty effect sagittally lengthens the velum and prevents longitudinal contracture scarring that can occur with straight-line repairs.
12.5.3 Preparation
An oral Ring–Adair–Elwyn endotracheal tube is used to facilitate the placement of the Dingman retractor. A roll is placed beneath the shoulders and the head in a gel doughnut. Loupes and a headlight are donned, and the patient is prepped and draped. Permissive hypotension can be used to potentially decrease blood loss. Simple silk sutures are placed on both sides of the anterior tongue and are used to control positioning. The Dingman mouth gag is positioned, and dental rolls are placed on either side of the endotracheal tube (at the bend) to prevent tube compression (Figure 12.6). The Dingman is “released” for 5 to 10 min every hour, reducing the threat of postoperative tongue-swelling. A 10 French suction catheter passed through the nostril to rest on the posterior pharyngeal wall provides continuous suctioning of irrigation and blood from the operative field, negating the need for a throat pack; as well, leaving this suction catheter in place after completion of the operation gives the anesthesia team a properly positioned tool to suction the hypopharynx. It is removed just before extubation. An appropriately sized nasopharyngeal airway is placed in the other nostril; alternatively, it can be placed under direct vision just before repair of the nasal layer. This nasopharyngeal airway provides postoperative airway maintenance in case there is undue postoperative tongue-swelling, and is removed on the first postoperative day when it is certain that tongue-swelling and airway compromise are no longer concerns.
Figure 12.6. At the 90° bend of the oral Ring–Adair–Elwyn endotracheal tube in the Dingman tongue retractor, dental rolls can be placed on either side of the tube to prevent kinking. Reprinted with permission from Elsevier in Ref. 20, p. 278. Copyright © 2009 Elsevier.
12.5.4 Markings
The double-opposing Z-plasty refers to two Z-plasties: one on the oral mucosa and one on the nasal mucosa, each containing two flaps. Therefore, there are two oral mucosa flaps (one anteriorly based and one posteriorly based) and two nasal mucosa flaps (one anteriorly based and one posteriorly based). Both posteriorly based flaps include the respective ipsilateral levator muscle.
Furlow’s traditional operation did not include “classical” relaxing incisions; however, the operation described herein is the modified procedure made popular by the Children’s Hospital of Philadelphia. Relaxing incisions are freely used as necessary to repair the velum without tension. Bilateral velar relaxing incisions, in the crease of the palatal–buccal junction and extending posteriorly from the retromolar trigone of the mandible and anteriorly to the maxillary tuberosity, are marked with a marking pen. They extend anteriorly onto the hard palate between the attached gingiva and the hard palate mucosa (Figure 12.7). The hamulus can be palpated just posterior to the maxillary tuberosity and marked bilaterally, taking care to leave enough tissue between the hamulus mark and the relaxing incision. The posterior edge of the hard palate is identified, and the “normal expected” junction of the hard and soft palates, at the margin of the cleft medially, is marked. Alternatively, this location can be estimated to be on a transverse line connecting the maxillary tuberosities. The bases of the uvulae, at the uvula–velum junction, are marked bilaterally. The medial edge of the entire hard and soft cleft, from uvular base to the anterior extent of the cleft is now outlined, taking care to leave enough additional millimeters of nasal lining to facilitate its repair. The uvular halves are marked to be demucosalized (Figure 12.7).
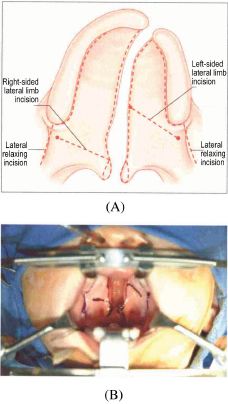
Figure 12.7. (A) Markings for the double-opposing Z-plasty velar repair. (B) Intraoperative markings. Reprinted with permission from Elsevier in Ref. 20, p. 279. Copyright © 2009 Elsevier.
The limbs of the oral Z-plasty flaps are now marked. The lateral limb of the left-sided posteriorly based oral musculomucosal flap is marked by joining the left hamulus to the junction of the hard and soft palates, creating a 60–90° flap. The lateral limb of the right-sided anteriorly based oral mucosal flap is marked by joining the right hamulus with the right uvular base, creating a 60–90° flap.
The velum and relaxing incisions are infiltrated with epinephrine for hemostasis. We prefer a fresh mixture of epinephrine with a concentration of 10 μg/mL. We consider the maximum dose of epinephrine in the otherwise healthy pediatric patient (not receiving halothane anesthetic) to be 10 μg/kg, so 1 mL/kg can be injected. Every 20 to 30 min, further dosing of the epinephrine may be administered to enhance hemostasis if needed.
12.5.5 Incisions and elevation of the oral flaps
Scissors are used to demucosalize the uvular halves, and 4-0 chromic gut suture on a tapered needle is used to mark both uvular tips, making it easier to identify them later (Figure 12.8). A No. 15 blade is used to cut the left lateral limb and cleft-margin incisions (Figure 12.9
Stay updated, free dental videos. Join our Telegram channel

VIDEdental - Online dental courses


