Fig. 3.1
Dental ceramic prosthetics
The use of all-ceramic crowns has been questioned because of their lack of strength. Their counterparts, the metal-ceramic crowns, have been used successfully; the majority of all full-coverage restorations and fixed prostheses are fabricated from metal-ceramic systems that have a failure rate of only 1–3 % over 5 years. Metal-ceramic systems (MCS) have come under scrutiny, however, because of (1) potential alloy corrosion leading to toxicity and allergy concerns; (2) esthetic problems such as lack of translucency, discoloration of some ceramics from silver in the alloy, and excessive value in the cervical third; (3) the amount of tooth reduction necessary, and the associated tendency to overcontour the restoration; and (4) incompatibility between metal and ceramic, and the difficulty in establishing standard tests for bond strength and thermal compatibility.
Fixed prosthodontic treatment, whether involving complete or partial coverage and natural tooth or dental implant abutments, commonly relies on indirect fabrication of definitive prostheses in the dental laboratory. Historically, the necessity for provisional treatment has been primarily derived from this methodological process.
The importance of interim treatment, however, is more far-reaching than is portrayed by this procedural necessity and the requirements for satisfactory provisional restorations differ only slightly from the definitive treatment they precede. In the last few decades, ceramic materials for restorative. Dentistry have evolved significantly, and esthetic restorations, such as all-ceramic crowns and veneers, are routinely used in practice.
Different materials and production systems are available for all-ceramic dental restorations. There are two main options when using ceramics for prosthetic restorations; a single layer ceramic that is attached to the tooth structure or a two-layered structure with a high-strength ceramic core material, which supports the more fragile veneering ceramic.
Some high strength materials are designed for computerized milling techniques (CAD/CAM) where the core structure is milled from the presintered block of an oxide ceramic. A relative recent dental ceramic is yttria-stabilized zirconium oxide polycrystals. These materials are manufactured from fine particles of ZrO2 and 3–5 % Y2O3, which form a partly stabilized tetragonal structure at room temperature after heat treatment. Due to the optical opacity of these materials, they are covered with veneering ceramic, usually feldspathic types, with esthetic characteristics similar to the natural tooth substance.
The veneering process involves a firing procedure at high temperatures (750–900 °C) and subsequent cooling of the restoration. This process is performed at least once, usually two to five times. It is not fully known what effect this thermal history has on the properties of the core ceramic.
Patients have become more and more demanding regarding the esthetic and biocompatibility of their dental restorations, ceramic as material for inlays, onlays, crowns and bridges has become a main goal for scientific interest especially from the material point of view.
Lithium disilicate ceramic restorations became available in 2006, and can be fabricated with a heat-pressed or computer-aided manufacturing (CAD/CAM). The better translucency and shade variety makes possible anatomically contoured monolithic restorations. Lithium disilicate restorations may be fabricated in the dental office and/or dental laboratory. Improved in-office chairside milling technology allows restorations to be designed, milled, and inserted at the same appointment as tooth preparation. With the dental laboratory-based cut-back method nano-fluorapatite ceramic can be layered onto a lithium disilicate substructure at the incisal, occlusal, or facial areas to improve the optical properties and to customize the esthetic and anatomic contours.
Dental ceramics are inherently fragile in tension. While the theoretical tensile strength of porcelain is dependent upon the silicon-oxygen bond, the practical strength is 10–1,000 times less than the nominal strength.
Generally cracks may propagate in different ways in the polycrystalline ceramic material, as seen in Fig. 3.2. The crack is formed from the coalescence of microcavities inside the material, giving an appearance of acute brittle fracture (Fig. 3.2a). The crack propagates with sharp point, without any visible permanent deformation, running through the grains (transgranular fracture—Fig. 3.2b), or between grains (intergranular fracture—Fig. 3.2c).
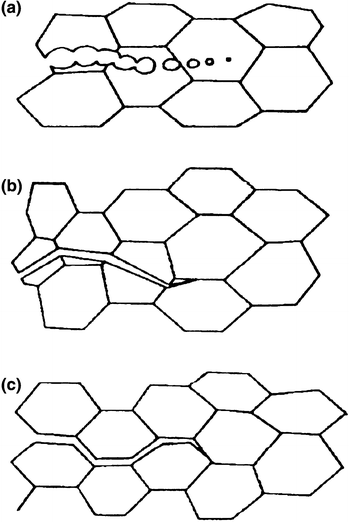

Fig. 3.2
Brittle fracture and crack propagation in the polycrystalline ceramic material: a coalescence of microcavities; b transgranular crack propagation; c intergranular crack propagation (Adapted of Askeland 2009)
Susceptibility to fracture is the result of several factors. A volume and surface distribution of stress-raising microcracks is present in manufactured ceramics. Microcracks are caused by the condensation, melting, and sintering process; by the high contact angle of ceramics on metal; by differences in the coefficient of thermal expansion between alloy or core and veneers; by grinding and abrasion; and by tensile stresses during manufacture, function, and trauma.
Strength is most dependent upon the number and severity of these flaws. The flaws of greatest importance are located in surface areas, and are in the range of 100 pm in diameter. The largely covalent: ionic bonded structure of ceramics confers resistance to chemical degradation in the oral environment; however, it also imparts brittleness. Ductile materials, such as metallic alloys, can dissipate stress by slip and plastic deformation. Brittle materials, such as dental ceramics, have a limited capacity for distributing localized stress at nominal temperatures.
The critical strain of dental ceramics is low; the material can withstand a deformation of approximately 0.1 % before fracture. Repetitive loading, resulting in fluctuating stresses and strains, may be the most common mechanism of failure of dental ceramics.
Stay updated, free dental videos. Join our Telegram channel

VIDEdental - Online dental courses


