For the general dentist, the use of botulinum toxin type A (BTA) confers the ability to exert control over the soft tissues surrounding the mouth to better create a harmonious smile. The injection of BTA into the facial musculature requires a level of finesse to achieve the desired outcomes. A sound understanding of the toxin’s mechanism of action and the ability to manage potential complications are also necessary, as the dentist administering BTA must be competent to the same level as other providers who have traditionally been the gatekeepers of such agents.
Key points
- •
For the general dentist, the use of BTA confers the ability to exert control over the soft tissues surrounding the mouth to better create a harmonious smile.
- •
Although not technically challenging, the injection of BTA into the facial musculature requires a level of finesse to achieve the desired outcomes.
- •
A sound understanding of the toxin’s mechanism of action and the ability to manage potential complications are also necessary, as the dentist administering BTA must be competent to the same level as other providers who have traditionally been the gatekeepers of such agents.
Once firmly secured in the armamentarium of plastic surgeons, the use of botulinum toxin type A (BTA), perhaps best-known commercially by the household name Botox, has recently begun to see a diversification in the types of practitioners employing its use. Few could argue the impact that this and other neurotoxins have had on the practice of improving facial aesthetics. Although cosmetic purposes remain the most common application, this was actually not the original indication, and still several other indications continue to emerge.
For the general dentist, BTA can be an excellent practice builder when properly utilized. Because the perioral region contributes greatly to dental aesthetics, the ability to exert control over the soft tissues surrounding the mouth equips the dentist with additional tools to create a harmonious smile.
Although not technically challenging, the injection of BTA into the facial musculature requires a level of finesse to achieve the desired outcomes. A sound understanding of the toxin’s mechanism of action and the ability to manage potential complications are also a requisite, as the dentist administering BTA must be competent to the same level as other providers who have traditionally been the gatekeepers of such agents.
Mechanism of action
Prior to the advent of Botox and other subsequent neurotoxins used for therapeutic purposes, BTA was exclusively known as the causative agent in botulism poisoning. BTA is derived from the obligate anaerobe Clostridium botulinum . The earliest studied accounts of poisoning from this microbe date back to 1793, when an outbreak in Wildbad, Germany killed 6 people and affected 7 other people. The source was determined to be a contaminated batch of Blutwurst , or blood sausage—hence the name “botulism,” after the Latin word for sausage, botulus . A larger outbreak in Belgium nearly a century later allowed Emile Van Ermengem to identify toxins produced by C botulism as the cause of botulism poisoning. This most commonly occurs when consuming food contaminated and stored under anaerobic conditions (eg, improperly canned). Per unit mass, botulinum toxin remains the most potent and lethal toxin known, with an LD50 of 1 to 3 ng/kg in people.
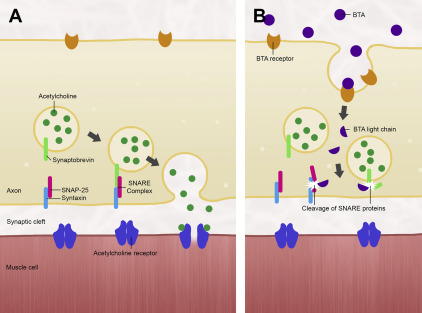
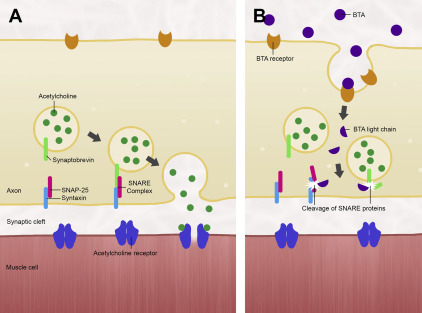
Type A is the most potent of 8 serotypes of botulinum toxin that have been identified thus far (designated A–H, the latter being the most recently discovered in 2013). Most variants cause paralysis at the neuromuscular junction by inhibiting the release of acetylcholine. Ordinarily, acetylcholine produced by the neuron remains contained in vesicles that upon depolarization of the neuron, fuse with the neuronal membrane to deposit the acetylcholine into the synaptic cleft. This process is facilitated by a complex of SNARE proteins: VAMP-2 and synaptobrevin on the vesicular surface, and syntaxin 1A and SNAP-25 on the neuronal membrane. When botulinum toxin is present, it binds to a separate class of surface proteins and becomes internalized by the neuron, subsequently cleaving the SNARE proteins that allow acetylcholine’s exit into the synapse ( Fig. 1 ). Owing to the storage vesicles of acetylcholine already within the motor endplate, the effect of paralysis is not manifested until 24 to 48 hours later, when these reserves are depleted. Paralysis would be permanent were it not for new axonal sprouts that are generated in 2 to 6 months, reestablishing the functional neuromuscular junction.
Mechanism of action
Prior to the advent of Botox and other subsequent neurotoxins used for therapeutic purposes, BTA was exclusively known as the causative agent in botulism poisoning. BTA is derived from the obligate anaerobe Clostridium botulinum . The earliest studied accounts of poisoning from this microbe date back to 1793, when an outbreak in Wildbad, Germany killed 6 people and affected 7 other people. The source was determined to be a contaminated batch of Blutwurst , or blood sausage—hence the name “botulism,” after the Latin word for sausage, botulus . A larger outbreak in Belgium nearly a century later allowed Emile Van Ermengem to identify toxins produced by C botulism as the cause of botulism poisoning. This most commonly occurs when consuming food contaminated and stored under anaerobic conditions (eg, improperly canned). Per unit mass, botulinum toxin remains the most potent and lethal toxin known, with an LD50 of 1 to 3 ng/kg in people.
Type A is the most potent of 8 serotypes of botulinum toxin that have been identified thus far (designated A–H, the latter being the most recently discovered in 2013). Most variants cause paralysis at the neuromuscular junction by inhibiting the release of acetylcholine. Ordinarily, acetylcholine produced by the neuron remains contained in vesicles that upon depolarization of the neuron, fuse with the neuronal membrane to deposit the acetylcholine into the synaptic cleft. This process is facilitated by a complex of SNARE proteins: VAMP-2 and synaptobrevin on the vesicular surface, and syntaxin 1A and SNAP-25 on the neuronal membrane. When botulinum toxin is present, it binds to a separate class of surface proteins and becomes internalized by the neuron, subsequently cleaving the SNARE proteins that allow acetylcholine’s exit into the synapse ( Fig. 1 ). Owing to the storage vesicles of acetylcholine already within the motor endplate, the effect of paralysis is not manifested until 24 to 48 hours later, when these reserves are depleted. Paralysis would be permanent were it not for new axonal sprouts that are generated in 2 to 6 months, reestablishing the functional neuromuscular junction.
Preparation and general considerations
The first US Food and Drug Administration (FDA)-approved pharmaceutical preparations of BTA in the late 1980s were indicated for the treatment of strabismus and blepharospasm. The side effect of eliminating wrinkles in the lateral canthal region of the eye (“crow’s feet”) was quickly realized, and following extensive study, FDA approval for additional indications soon followed, including cervical dystonia (2000), glabellar rhytids (2002), and axillary hyperhidrosis (2004).
In the United States, BTA is available by the trade names Botox Cosmetic, Dysport, and Xeomin. All medications have similar FDA-approved indications, but Botox’s ona botulinumtoxinA differs from Dysport’s abo botulinumtoxinA and Xeomin’s inco botulinumtoxinA primarily in regards to unit potency and nonprotein components that arise from different manufacturing processes. Commercially available vials of any of the medications contain a given number of biologically active units. It is important for the clinician to realize that these units are essentially arbitrary quantities used for convenience in dosing. In general, most literature agrees on a potency equivalence of 2.5 to 3 units of Dysport to 1 unit of Botox. Despite containing very similar toxins, this difference in potency can be attributed to the bacterial strain from which the toxin is sourced, the purification method, or differences in methods of testing potency. For purposes of consistency, all doses in this article will refer to Botox units, with the understanding that an equivalent dose of Dysport or Xeomin would be anticipated to be equally effective.
All pharmaceutical preparations of BTA in the United States used from November 1979 to December 1997 came from a single 150 mg batch (dubbed 79–11) isolated by Schantz and colleagues. By the time Botox Cosmetic was approved by the FDA in 2002, a newer batch (BCB 2024) had been isolated; it is from this batch that all modern Botox preparations are derived. A standard 100-unit vial of Botox contains just 4.8 ng of BTA. Because of this minuscule dose, Botox is shipped on dry ice as an empty glass vial containing a thin film of precipitate on the inside. After reconstitution with 2.5 cc preservative-free 0.9% sodium chloride for injection, it is stored under refrigeration and should be used within 24 hours. This dilution ratio allows for aspiration into 5 1 cc syringes, each containing a 0.5 cc solution of 20 units of Botox.
Patients receiving BTA injections should be informed regarding the limitations of its efficacy. Because of the axonal resprouting that occurs, motor function after neurotoxin treatment begins to return after approximately 3 months. This, however, is highly variable; some patients may need to receive touch-up injections in as little as several weeks, while others can last well beyond the often-quoted 90 days before needing to repeat injections. In patients who consistently follow-up for repeat treatment, the atrophy that results from prolonged inactivity creates muscles that are less capable of forming deep rhytids, and treatment can usually be performed less frequently. Thick muscles of the face, such as the masseter, generally require less frequent injections.
Although rare, resistance to BTA has been reported. High-dose treatment with BTA over a short time span in some has been demonstrated to cause resistance to further treatment in patients being treated for cervical dystonia. Some have speculated that a subclinical botulinum poisoning from contaminated food could result in antibodies against BTA in some patients. In these cases, higher doses of BTA may be needed. Off-label treatment with rimabotulinumtoxinB, available commercially as Myobloc may also prove beneficial, although not in every instance. Investigation into other serotypes of botulinum toxin for treatment of these patients is underway.
Like most every intervention performed for a patient, a thorough discussion on risks, benefits, and alternatives is necessary before administering BTA. Contraindications to its use include coexisting conditions that affect neuromuscular transmission, including systemic neuromuscular disease or concomitant use of aminoglycoside or spectinomycin antibiotics, which can potentiate BTA’s effect. Pregnancy, lactation, or a known adverse reaction to any of the components in commercially available BTA are also contraindications to its use.
Perioral region
Excessive Gingival Display
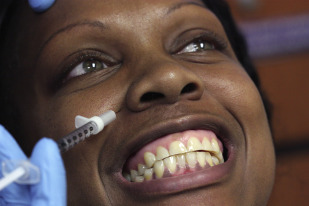
For the dental practitioner, the most obvious application of BTA is in the treatment of gummy smiles, or excessive display of maxillary gingiva. Although Botox does have a role in treating a high smile line resulting from a hyperfunctional upper lip, it is important to distinguish this from other causes of gummy smiles. Short clinical crowns, for instance, would be better treated with gingivoplasty to achieve aesthetic crown lengthening. Likewise, the definitive treatment of vertical maxillary excess would necessitate maxillary impaction via a Le Fort I osteotomy. Attempts to camouflage these conditions with BTA injection would result in an unnatural appearance owing to the excessive loss of function needed to prevent gingival display.
In properly selected patients, however, BTA can have a profound impact in the treatment of high smile lines. Unfortunately, achieving a satisfactory outcome in this region is extremely technique-sensitive, as overtreatment can result in transverse elongation and dysfunctional animation of the upper lip. Novice injectors especially may wish to gradually ease into the final result by treating this area in multiple low-dose treatments over several weeks rather than in a single appointment.
In true cases of hyperfunctional upper lips, the primary contributor to excessive display of maxillary gingiva is the levator labii superioris alaeque nasi (LLSAN) muscle, which participates in the last few millimeters of upper lip elevation. This muscle is also involved in the creation of deep nasolabial folds that develop in some patients. The LLSAN originates from the superior portion of the frontal process of the maxilla near the nasal bridge, traverses inferiorly along the lateral aspect of the nose, and inserts at the nasal cartilage and the lateral upper lip. As such, its function involves dilation of the nares, and—along with the levator anguli oris, leavator labii superioris, and zygomaticus muscles—elevation of the upper lip ( Fig. 2 ).
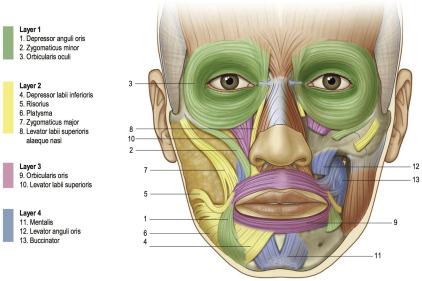
For treatment of either gummy smiles or deep nasolabial folds, anywhere from 1 to 5 units of Botox may be needed for each side. The injection is placed into the LLSAN at the depth of the fold, just lateral to the piriform aperture, which can be palpated when pressing the alar–facial junction medially ( Fig. 3 ). Again, it should be remembered that it is easy to overtreat this muscle, and the resultant loss of upper lip animation may be more objectionable to the patient than the original presenting complaint.