Introduction
The muscles of mastication are important in positioning the mandible and can therefore affect the patency of the upper airway. The aim of this study was to determine whether resting masticatory muscle activity influences the response to mandibular advancement splint treatment in patients with obstructive sleep apnea.
Methods
Thirty-eight adult patients with obstructive sleep apnea were recruited for the study. Baseline electromyographic activities of the right anterior and posterior temporalis, masseter, and submandibular muscles were recorded with surface electrodes while the patients were awake, in the upright and supine positions, with the jaw in the postural position, and with and without a mandibular advancement splint. Muscle activity of the patients with obstructive sleep apnea was compared between responders (apnea-hypopnea index change ≥50%, and <10 events per hour) and nonresponders (apnea-hypopnea index change <50%) to mandibular advancement splint treatment.
Results
There were 18 responders and 20 nonresponders to mandibular advancement splint treatment. The responders had a trend for increased muscle activity in all muscle groups and scenarios. The resting muscle activity of the submandibular and masseter muscles while lying at rest and of the submandibular and posterior temporalis muscles while lying with the mandibular advancement splint in place were significantly greater ( P <0.05) in the responders than in the nonresponders.
Conclusions
Inherent baseline differences in muscle activity between responders and nonresponders to mandibular advancement splint treatment in adults with obstructive sleep apnea were observed. This preliminary study suggests that there might be a correlation between responsiveness with mandibular advancement splint treatment and baseline muscle activity.
Obstructive sleep apnea (OSA) affects 24% of men, 9% of women, and 1% to 4% of children and is the most common sleep-related breathing disorder. In 1981, Sullivan et al introduced treatment with continuous positive airway pressure (CPAP), which works by pneumatically splinting the airway. This device is the most common and successful treatment modality for OSA. However, because of the cumbersomeness of the CPAP device, dental appliances also have an increasing role in patients with mild to moderate OSA or in those who refuse or cannot tolerate CPAP. The most widely used dental appliance, the mandibular advancement splint, primarily acts by advancing and lowering the mandible and might improve upper airway patency during sleep by enlarging it or by decreasing upper airway collapsibility.
To date, studies have shown that success with the mandibular advancement splint is not achievable in all patients. Reported efficacy varies between studies and also depends on the defintion of the treatment outcome. For example, Ishida et al showed that 68% of patients had a 50% reduction in the apnea-hypopnea index (AHI) with a mandibular advancement splint. In 2 randomized controlled trials, 75% of the patients had an AHI score of less than 15 events per hour after using the mandibular advancement splint, or, with a more strict definition, 36% had less than 5 events per hour after using the mandibular advancement splint. Because of the uncertainty surrounding the treatment outcome with the mandibular advancement splint, it would be desirable to be able to predict treatment outcome so as to appropriately select patients for this form of treatment.
The pathogenesis of OSA is complex, but it is most likely due to a combination of an anatomically small pharyngeal airway and a sleep-related decline in the upper airway dilator muscle activity. Kurtulmus et al recorded the right submental and masseter muscle activities during 2 polysomnography sessions in 1 night in 10 mild and 10 moderate OSA patients. After insertion of the mandibular advancement splint, the mean AHI scores in both groups of patients decreased significantly ( P <0.05), and this correlated with an increase in resting electromyographic activity. The authors concluded that the mandibular advancement splint increased the muscle activity of the masseter and submental muscles, and the increase in electromyographic activity might be responsible for preventing the upper airway from collapsing.
Because improvements in OSA with mandibular advancement splint therapy might be associated with changes in masticatory muscle activity, we hypothesized that the level of resting muscle activity might influence treatment outcome and could have a role as a predictor. Therefore, the aim of this study was to determine, in a group of awake OSA subjects, whether resting masticatory muscle activity differs between responders and nonresponders to mandibular advancement splint treatment. Moreover, the baseline muscle activity was assessed to improve our understanding of the characteristics of responders and nonresponders to the mandibular advancement splint and to determine whether it could be used as a predictor of mandibular advancement splint treatment response.
Material and methods
For this prospective clinical study, we recruited subjects from 4 tertiary referral sleep clinics in Sydney, Australia. Inclusion criteria were at least 2 symptoms of OSA (snoring, witnessed apneas, fragmented sleep, daytime sleepiness) plus evidence of OSA on polysomnography (AHI ≥10 per hour). Subjects were excluded if they had periodontal disease, insufficient numbers of teeth (less than 10 teeth in either dental arch), evidence of temporomandibular disorders including pain and limited opening less than 40 mm, or received treatment for OSA with a mandibular advancement splint within the last 3 months. The study was approved by the institutional ethics committees of the Sydney Local Health District and the Sydney Dental Hospital of New South Wales, Australia. Written informed consent was obtained from all subjects. Fifty-six patients were screened for the study, and 16 were excluded because of periodontal disease (n = 4), insufficient teeth (n = 2), temporomandibular disorder (n = 1), restorative dentistry requirements (n = 6), preference to try CPAP (n = 1), or refusal to participate (n = 2). Consequently, 40 OSA adults participated.
Because some previous OSA research has suggested that cephalometric variables alone or combined with other anthropomorphic and polysomnographic variables can improve the ability to predict responsiveness to the mandibular advancement splint, cephalometry was included in this study. Cone-beam computed tomography (CBCT) scans were taken of all subjects before treatment with the mandibular advancement splint. These images were acquired with a NewTom 3G (QR, Verona, Italy) with a 12-in field of view at 110 kV with an exposure time of 5.4 seconds in a standardized fashion. The DICOM data obtained from the CBCT images were processed to produce right lateral skull cephalometric radiographs. These radiographs were extracted and analyzed using Dolphin Imaging premium software (version 11.0; Dolphin Imaging & Management Solutions, Chatsworth, Calif). The cephalometric measurements are defined in Table I . All cephalometric measurements were made by 1 assessor (S.Y.L.M.) who was blinded to the subject’s apneic status and was assessed for reliability with intraclass correlation by repeated measurements after 3 months under the same conditions.
| SNA | Angle between sella, nasion, and A-point |
| SNB | Angle between sella, nasion, and B-point |
| ANB | Angle between A-point, nasion, and B-point |
| Jarabak’s ratio | Ratio of the distance from sella to gonion compared with the distance from nasion to menton |
| SN | Plane formed by sella to nasion |
| MP (Go’-Me) | Mandibular plane between gonion and menton |
| SN-MP | Mandibular plane angle |
| Co-Gn | Distance between condylion and gnathion |
| Co-A | Distance between condylion and A-point |
Because emotional states have been demonstrated to affect muscle activity, 2 psychological questionnaires were included: the depression anxiety stress scales and the state-trait anxiety inventory.
Each subject attended 3 appointments of approximately 1 hour each: an intial consultation followed approximately 2 weeks later by a second appointment in which all electromyographic records were obtained and the mandibular advancement splint was issued, followed by a posttitration mandibular advancement splint consultation.
At the initial appointment, all subjects completed a medical history form from the Sydney Dental Hospital before a comprehensive orthodontic examination. Pretreatment records were collected, including CBCT scans, study models, and photographs. If the patient fullfilled all inclusion criteria, impressions and bite registrations for the construction of the mandibular advancement splint were also collected.
On the day of the recording (second appointment), the subjects were advised not to consume coffee, alcohol, tea, or other stimulants to reduce confounding muscle activity. The men were requested to shave in the locations of the surface electrodes. Before we collected the electromyographic recordings, all subjects completed the depression anxiety stress scales, the Epworth sleepiness scale, and the state-trait anxiety inventory questionnaire. Body mass index and neck circumference measurements were also recorded.
After completion of the titration or acclimatization, at approximately 4 to 8 weeks, all patients returned for a third appointment in which body mass index and neck circumference measurements were made, the 2 questionnaires were completed, and the electromygraphic recordings sessions were repeated as above. The posttitration electromyographic changes will be the subject of another article.
The baseline electromyographic recordings were acquired and stored on a portable polysomnograph machine, Siesta 802 (Compumedics, Victoria, Australia). Bipolar surface and hook electrodes were used to record electromyographic activity, and signals were processed with the Siesta unit. The signals were passed through an amplifier with a low-pass filter of 200 Hz and a high-pass filter of 10 Hz. The input impedence was 10 MΩ, and the common mode rejection ratio was greater than 100 dB. The notch filter was set at 50 Hz, and the sampling rate was 1024 samples per second.
Three bipolar surface electrodes were placed ( Fig ) over the right submandibular, masseter, and anterior temporalis regions. A pair of subcutaneous needle electrodes was placed over the right posterior temporalis ( Fig ). A reference electrode and a ground electrode were placed on the central forehead and on the left mastoid region, respectively. To minimize electrode impedence, all recording sites were cleansed with 70% isopropyl alcohol and permitted to dry before placement of the electrodes.
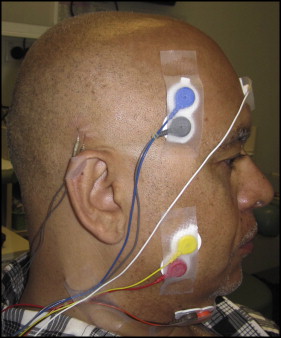
Pregelled, self-adhesive, disposable surface electrode pairs measuring approximately 10 mm in diameter and 10 mm apart (Duo-Trode; MyoTronics, Kent, Wash) were used to measure the electromyographic activity of the muscles. A highly conductive, nonsaline-solution based, electrode gel (Signa Gel; Alpine Biomed ApS, Skovlunde, Denmark) was applied to aid conductivity, and then the electrodes were secured in place with Transpore adhesive tape (3M, St Paul, Minn). All electrodes were placed parallel to the direction of the muscles fibers as described below.
- 1.
Submandibular muscle: the bipolar electrodes were positioned approximately 1 cm from the border of the mandible in the submandibular triangle region.
- 2.
Masseter muscle: after palpation during clenching, the lower electrode of the bipolar surface electrodes was placed approximately 1 cm from the lower border of the mandible, in the lower third of the masseter and 1 cm from its anterior border.
- 3.
Anterior temporalis muscle: bipolar surface electrodes were placed vertically along the anterior margin of the muscle (corresponding to the frontoparietal suture), as close to the hairline as possible.
Two stainless-steel bipolar hook wire electrodes (diameter, 0.12 mm) insulated with synthetic fluorine-containing resin with bare tips (1 mm) were placed approximately 10 mm apart in the posterior temporalis region. EMLA (APP Pharmaceuticals, LLC, Schaumberg, Ill), a topical anasthetic, was placed on the skin before inserting the hook electrodes. After palpation on clenching, the hooks were placed over the posterior temporalis, superior to the highest point on the ear. The electrodes in each region were placed parallel to each other and to the orientation of temporalis fibers according to previous descriptions. The hook electrode leads were secured with the adhesive tape to the most superior part of the ear.
The recordings were standardized with the subject awake, eyes open, and not speaking or swallowing, and with the jaw in a relaxed position without the teeth occluding but the lips together. Each recording trial was repeated 3 times, each about 30 seconds duration, with approximately 15 seconds rest between trials. The scenarios included upright positioning in the dental chair at approximately 75° to the horizontal, with the subject’s head supported by the chair’s head rest; and supine positioning with the chair at 175° to the horizontal, with the subject’s head supported by the chair’s head rest. The randomized order was repeated with the mandibular advancement splint in situ; however, the subjects kept their lips together or apart to maintain an unstrained position. Thus, each subject participated in 4 scenarios: seated upright with and without the mandibular advancement splint, and supine with and without the mandibular advancement splint. To minimize an order effect, the subjects were numbered sequentially according to the order they were recruited to the study. Every odd-numbered subject had the upright recordings at rest done first, followed by the supine at rest recordings, then upright with the mandibular advancement splint, and supine with the mandibular advancement splint. Every even-numbered subject had the supine recordings at rest done first, then the upright at rest recordings, supine with the mandibular advancement splint, and upright with the mandibular advancement splint.
All electromyography signals were obtained from the portable sleep study machine, Siesta 802, which converted the data to digital format. It was then processed using the Profusion PSG 2 software (Compumedics). Full-wave rectification was carried out using the root mean square formula. The root mean square was obtained for a stable 5-second epoch period within 10 to 15 seconds from the beginning of the recording for each trial. Each trial’s electromyographic data were assessed for swallowing or movement as determined by changing electromyographic activity in the trial. If this was noted, those data were excluded from the analysis, and another 5-second stable period of electromyographic activity was selected in the trial by visually inspecting the electromyographic activity on a line graph.
A custom-made 2-piece mandibular advancement splint (SomnoDent; SomnoMed, Crows Nest, New South Wales, Australia) was used. The appliance was initially fabricated at 75% of the subjects’ maximaum advancement using a George Gauge (Great Lakes Orthodontics, Tonawanda, NY). The vertical opening when measured from the maxillary and mandibular incisal edges ranged from 2 to 8 mm. This variation depended on the malocclusion (overbite, curve of Spee, overerupted teeth) and allowed for sufficient thickness of the acrylic. The appliance was incrementally titrated. All subjects were given instructions to titrate the appliance bilaterally 0.2 mm daily until the maximum comfortable limit of the mandibular advancement was reached. All mandibular advancement splint records were taken by the same operator (S.Y.L.M.). The subjects were given a diary to complete daily during the titration period.
All subjects, except one who could not wear the mandibular advancement splint, underwent polysomnography to determine the treatment outcome with the mandibular advancement splint and were scored according to standard criteria, which have been previously described. Apneas were defined by a cessation of airflow for at least 10 seconds in association with oxygen desaturation of at least 3%, or an arousal. Hypopneas were defined by a reduction in the amplitude of airflow, as measured using nasal pressure or thoraco-abdominal wall movement by more than 50% of the baseline measurement for more than 10 seconds, in association with oxygen desaturation of at least 3%, or an arousal. “Responders” were subjects with 50% or greater improvement in AHI and posttreatment AHI scores of less than10 per hour. “Nonresponders” had less than 50% improvement in AHI. Nonresponders were managed appropriately by the referring sleep physician according to standard clinical pathways. Subjective treatment outcome was assessed using the Epworth sleepiness scale.
Statistical analysis
Statistical analyses were performed using the statistical computing package R (version 2.15.0; R Foundation for Statistical Computing, Vienna, Austria). All descriptive statistics are presented as means and standard deviations. Two-sample t tests or Mann-Whitney tests were used, depending on the distribution of the data to compare cephalometric, body mass index, neck circumference, the Epworth sleepiness scale, the 2 questionnaires, and the baseline and postmandibular advancement splint sleep study variables between responders and nonresponders. Paired t tests were used to assess the efficacy of the mandibular advancement splint treatment by assessing the polysomnography variables and the Epworth sleepiness scale. Kruskal-Wallis tests were used to assess the differences between the electromyographic values at each muscle and position separately between the mild, moderate, and severe OSA groups. Intrarater reliability was assessed using the intraclass correlation coefficient (ICC). Statistical significance was accepted at P <0.05. Univariate logistic regression models were constructed to examine the relationship between the electromyographic values between responders and nonresponders for each muscle and each body position.
The method error ICC values ranged from 0.93 to 0.99 for linear and angular lateral cephalometric measurements, with P <0.0001 for all measurements.
Results
From the sample of 40 subjects in the study, 38 completed the protocol appropriately. One subject withdrew for inability to acclimatize to the mandibular advancement splint because of worsening of preexisting migraines; this subject was classified as a compliance failure. Another subject had interceptive medical treatment, a rhinoplasty, before the follow-up sleep study and was therefore excluded from the study.
The mandibular advancement splint treatment resulted in 18 (47%) responders (postmandibular advancement splint AHI range, 1.1-9.9 per hour) and 20 (53%) nonresponders (postmandibular advancement splint AHI range, 5.7-30.1 per hour). The polysomnographic outcomes are summarized in Table II . There were statistically significant improvements with the mandibular advancement splint treatment, in comparison with baseline across a number of sleep study variables, including duration of rapid eye movement sleep, arousal index, AHI, non-rapid eye movement AHI, rapid eye movement AHI, and minimum oxygen saturation (SaO 2 ) ( Table III ). One patient had an increase in AHI, from 15.4 to 20.2 per hour. The Epworth sleepiness scale between baseline and postmandibular advancement splint titration for all the subjects was statistically significantly different (before, 8.95 ± 4.46, vs after, 6.82 ± 4.33; P <0.01).
| Sleep study variable | All subjects | Responders | Nonresponders | P value | |||
|---|---|---|---|---|---|---|---|
| Mean | SD | Mean | SD | Mean | SD | ||
| Initial total sleep time (TST) (min) | 350.06 | 78.38 | 347.9 | 58.6 | 352.05 | 94.27 | 0.8688 |
| TST with MAS (min) | 351.41 | 64.89 | 360.7 | 62 | 343.02 | 67.88 | 0.4061 |
| Initial sleep efficiency (%) | 78.07 | 13.85 | 77.76 | 12.1 | 78.35 | 15.55 | 0.5927 |
| Sleep efficiency with MAS (%) | 80.41 | 12.44 | 81.82 | 12.2 | 79.14 | 12.84 | 0.5927 |
| Initial REM (% TST) | 15.86 | 5.96 | 15.19 | 6.34 | 16.45 | 5.69 | 0.357 |
| REM (% TST) with MAS | 18.8 | 7.13 | 18.61 | 5.31 | 18.98 | 8.58 | 0.8748 |
| Initial arousal index (n/h) | 34.1 | 18.32 | 39.58 | 14.2 | 29.18 | 20.5 | 0.0226 |
| Arousal index (n/h) with MAS | 23.16 | 9.72 | 22.23 | 9.68 | 23.99 | 9.93 | 0.5845 |
| Initial baseline AHI (n/h) | 26.73 | 17.04 | 28.79 | 22.1 | 28.79 | 22.09 | 0.4241 |
| AHI with MAS (n/h) | 9.32 | 6.96 | 4.42 | 2.51 | 13.72 | 6.74 | <0.0001 |
| Initial NREM RDI (n/h) | 24.68 | 16.5 | 23.42 | 7.81 | 25.82 | 21.79 | 0.4471 |
| NREM RDI with MAS (n/h) | 7.32 | 7.06 | 3.41 | 2.43 | 10.84 | 8.01 | 0.0005 |
| Initial REM RDI (n/h) | 38.16 | 21.2 | 32.81 | 17.4 | 42.97 | 23.48 | 0.1357 |
| REM RDI with MAS (n/h) | 24.32 | 18.5 | 11.68 | 11.2 | 35.7 | 16.4 | <0.0001 |
| Initial minimum SaO 2 (%) | 83.66 | 7.92 | 85.76 | 6.78 | 81.85 | 8.58 | 0.089 |
| Minimum SaO 2 (%) with MAS | 87.92 | 4.73 | 90.44 | 3.88 | 85.65 | 4.32 | 0.001 |
| Sleep study variable | Difference | 95% CI | P value |
|---|---|---|---|
| Total sleep time (TST) (min) | −1.35 | −29.62, 26.92 | 0.9234 |
| Sleep efficiency (%) | −2.34 | −6.53, 1.85 | 0.2656 |
| REM (% TST) | −2.95 | −5.48, −0.41 | 0.0238 |
| Arousal index (n/h) | 10.94 | 5.41, 16.48 | 0.001 |
| AHI (n/h) | 17.41 | 12.74, 22,07 | <0,0001 |
| NREM AHI (n/h) | 17.36 | 12.46, 22.26 | <0.0001 |
| REM AHI (n/h) | 13.84 | 6.29, 21.38 | 0.001 |
| Minimum SaO 2 (%) | −4.26 | −6.93, −1.60 | 0.0025 |
Anthropomorphic characteristics at baseline for the entire group and for the subgroups are shown in Table IV . Subjects (n = 38) who completed the titration of the mandibular advancement splint (26 men, 12 women) had a mean age of 52.55 ± 10.67 years (range, 29-74 years), body mass index of 27.73 ± 4.36 kg/m 2 (range, 20.82-38.42 kg/m 2 ), neck circumference of 39.63 ± 3.98 cm (range, 34-48 cm), baseline AHI of 26.73 ± 17.04 per hour (range, 10.1-77.4 per hour), and Epworth sleepiness scale scores of 8.17 ± 4.55 (range, 2-18). The mean acclimatization period was 9 ± 4 weeks (range, 4-31 weeks). The mean mandibular advancement with the mandibular advancement splint was 9.99 ± 2.74 mm at the end of the acclimatization period. No statistically significant differences were found between the responder and nonresponder groups with respect to any anthropomorphic measurements ( Table IV ), cephalometric variables ( Table V ), psychological assessments ( Table IV ), or total mandibular advancement ( P = 0.49).
| Subject variable | All subjects | Responders | Nonresponders | P value | |||
|---|---|---|---|---|---|---|---|
| Mean | SD | Mean | SD | Mean | SD | ||
| Subjects (n) | 38 | 18 | 20 | ||||
| Male sex (%) | 68 | 61 | 75 | 0.5685 | |||
| Skeletal class (I/II/III) (%) | 61/16/24 | 78/6/17 | 45/25/30 | 0.0972 | |||
| Age (y) | 52.55 | 10.7 | 52 | 12 | 53.05 | 9.65 | 0.7692 |
| Height (cm) | 172.9 | 8.91 | 172.14 | 7.91 | 173.62 | 9.9 | 0.6103 |
| Weight (kg) | 83.27 | 21.6 | 80.29 | 15 | 85.96 | 17.52 | 0.2889 |
| BMI (kg/m 2 ) | 27.73 | 4.36 | 26.98 | 3.88 | 28.4 | 4.75 | 0.3154 |
| Neck circumference (cm) | 39.63 | 3.98 | 39.17 | 4.2 | 40.05 | 3.83 | 0.3719 |
| ESS | 8.95 | 4.46 | 9.65 | 4.37 | 8.17 | 4.55 | 0.3112 |
| DASS-stress | 3.92 | 2.68 | 3.88 | 2.71 | 3.95 | 2.72 | 0.9617 |
| DASS-depression | 2.17 | 3.88 | 2.76 | 4.59 | 1.63 | 3.15 | 0.7028 |
| DASS-anxiety | 2.11 | 2.11 | 2 | 1.77 | 2.21 | 2.42 | 0.9342 |
| STAI | 31.39 | 7.99 | 31.28 | 7.23 | 31.5 | 8.8 | 0.8033 |
| Cephalometry | All subjects | Responders | Nonresponders | P value | |||
|---|---|---|---|---|---|---|---|
| Mean | SD | Mean | SD | Mean | SD | ||
| SNA (°) | 82.46 | 4.26 | 82.22 | 4.24 | 82.68 | 4.38 | 0.7464 |
| SNB (°) | 79.62 | 4.04 | 79.5 | 4.11 | 79.74 | 4.08 | 0.8614 |
| ANB (°) | 2.89 | 3.43 | 3 | 1.91 | 2.89 | 3.43 | 0.5292 |
| Jarabak’s ratio | 66.76 | 5.79 | 66.28 | 5.59 | 67.21 | 6.08 | 0.6299 |
| SN-MP (°) | 32.95 | 7.62 | 33.22 | 6.79 | 32.68 | 8.51 | 0.8324 |
| Co-Gn (mm) | 113.44 | 7.91 | 110.91 | 6.85 | 115.72 | 8.27 | 0.05793 |
| Co-A (mm) | 85.25 | 6.83 | 83.58 | 5.2 | 86.76 | 7.84 | 0.1476 |
The mean rectified electromyography values showed a general trend toward an increase in muscle activity for all muscle groups in all scenarios in the responder group ( Table VI ) compared with the nonresponder group.
| Position/muscle | Responders | Nonresponders | ||
|---|---|---|---|---|
| Mean | SD | Mean | SD | |
| Lying at rest | ||||
| Submandibular | 4.47 | 3.16 | 2.82 | 1.2 |
| Masseter | 3.5 | 2.13 | 2.19 | 0.76 |
| Anterior temporalis | 4.7 | 2.3 | 3.28 | 1.68 |
| Posterior temporalis | 7.17 | 5.87 | 4.87 | 2.65 |
| Upright at rest | ||||
| Submandibular | 4.39 | 2.58 | 3.24 | 1.79 |
| Masseter | 4.79 | 3.82 | 2.78 | 1.84 |
| Anterior temporalis | 6.2 | 3.72 | 4.28 | 1.86 |
| Posterior temporalis | 7.26 | 5.11 | 6.51 | 4.09 |
| Lying at 75% protrusion | ||||
| Submandibular | 4.76 | 2.5 | 3.23 | 1.5 |
| Masseter | 4.27 | 2.74 | 3.2 | 1.87 |
| Anterior temporalis | 4.29 | 2.33 | 2.89 | 0.95 |
| Posterior temporalis | 5.96 | 5.03 | 5.14 | 3.45 |
| Upright at 75% protrusion | ||||
| Submandibular | 4.87 | 2.54 | 3.69 | 1.99 |
| Masseter | 4.41 | 3.08 | 3.43 | 2.18 |
| Anterior temporalis | 5.73 | 4.57 | 4.45 | 2.23 |
| Posterior temporalis | 6.86 | 3.86 | 6.02 | 3.65 |
Stay updated, free dental videos. Join our Telegram channel

VIDEdental - Online dental courses


