Dental caries
Cracked tooth
Fractured restoration
Post-restorative sensitivity
Medication sensitivity
Post-nonsurgical/postsurgical periodontal sensitivity
Palatogingival groove
Non-odontogenic origin
In the definition of DH, the word ‘disease’ replaced the previously used word ‘pathology’ following the Canadian consensus. However, disease relates more to the clinical outcome of the condition, rather than its aetiology or pathology which will be discussed below.
Aetiology
DH can be associated with a number of aetiologies including non-surgical and post surgical treatment of periodontal disease, erosion and gingival recession. These are discussed in turn below together with some other rarer aetiology.
Postsurgical Periodontal Disease (Recession Induced by Management)
DH may occur iatrogenically and it has been reported to affect up to 57 % of the general population in one subject-reported study following scaling or root planning (Irwin and McCusker 1997). Another study of periodontal patients reported a prevalence of 85–95 % (Chabanski et al. 1996). In an early in vitro study, it was reported that after root surface debridement; the dentine surface tubules were exposed following the removal of the surface covering or smear layer (in part). In health, the smear layer reforms and prevents 86 % of fluid movement across the dentine (Pashley et al. 1978). The smear layer may be defined as a thin ‘loose’ layer consisting of organic collagen and glycosaminoglycans that form an adherent matrix over mineralised tissue arising from saliva and dentine particles that might occlude the dentine tubules (Brannstrom 1966; Pashley 1984). It has also been suggested that in periodontal disease and any treatment that may expose dentine, bacteria may colonise and break down the smear layer and penetrate into the root dentine (Adriaens 1989) which in turn may initiate inflammation in the pulp. Several Investigators have suggested that this may be an alternative explanation for the mechanism associated with DH (Lundy and Stanley 1969; Brannstrom 1982; Dababneh et al. 1999). Although in the light of the changes in the terminology of DH/RS it may be more relevant as a mechanism for RS. It is the bacterial products, however, rather than the bacteria, which are likely to diffuse towards the pulp. Initially, plasma proteins/immunoglobulins from the blood diffuse into the dentine tubules, decreasing permeability (Pashley 1984). Following chronic periodontal disease and ongoing treatment, increases in pulpal pressures, nerve spouting reduction in pain thresholds, scar tissue and reduced plasma proteins may worsen DH (Pashley 1984).
Tooth Whitening Procedures (Unknown or Not Understood Interrelationship)
Tooth whitening procedures often involve carbamide peroxide, which breaks down into hydrogen peroxide and urea and bleach the tooth. This causes dehydration within the tooth and symptoms of DH. However, the symptoms are often temporary and for the duration of treatment (Ferrari et al. 2007). Bleaching may however, involve a different mechanism to that of DH (Gillam et al. 2013) and therefore is not strictly an aetiology factor for DH and may therefore, require alternative management strategies (Markowitz 2010).
Developmental Lesions (Rare Conditions Where Surface Morphology of Teeth Changes)
These can often affect the primary and later permanent dentition and can cause symptoms of DH. They include, for example, amelogenesis and dentinogenesis imperfecta as well as other conditions of the enamel and/or dentine that cause hypomineralisation and hypoplasia. As with bleaching, these symptoms may involve a mechanism different to that of DH.
Dietary Factors (Erosive Diets) and Gingival Recession
DH is most commonly associated with erosive dietary factors and gingival recession, and these will be discussed in more detail in the following sections of this chapter. The largest European clinical study to investigate the presence or otherwise absence of DH in association with its various aetiologies involved 3,187 adults enrolled from general dental practices in France, Spain, Italy, the United Kingdom, Finland, Latvia and Estonia (West et al. 2013b). The proportion of subjects who were examined clinically to have DH on at least one tooth surface was 42 % following an evaporative stimulus in a clinical setting. The proportion of subjects who reported having DH in the previous 12 months was less (27 %), and this might reflect the transient nature of the pain in addition to good coping mechanisms. According to (West et al. 2013b) when the various aetiological factors were investigated, there was a strong progressive relationship between clinically elicited DH and erosive tooth wear caused by dietary factors and gingival recession. 29 % of these subjects had signs of tooth wear recorded using the Basic Erosive Wear Examination (Bartlett et al. 2013). The prevalence figure of DH was relatively high compared to similar studies conducted in general dental practice, 2.8 % (Rees and Addy 2004), 9.1 % (Amarasena et al. 2011) and 5.3 % (Bahsi et al. 2012), but DH has also been reported in other studies and associated with erosive tooth wear (Lussi and Schaffner 2000; Fares et al. 2009) and gingival recession (Bamise et al. 2008) (see Chap. 3). In a population in the United Kingdom, the prevalence of DH examined clinically was observed to be over 40 % and was associated with gingival recession (Olley et al. 2013). An investigation of the same group of patients also indicated that this population had 43 % of tooth surfaces with tooth wear (Olley et al. 2014b). In a prospective study of randomly selected subjects in Switzerland, 34.8 % of subjects reported DH; however, the prevalence of DH in those subjects with tooth wear was 84.6 % (Lussi and Schaffner 2000). In a recent study (Olley et al. 2014b ahead of print), subjects who recently consumed dietary acids were more likely to have DH when examined at their dental appointment. It is therefore not surprising that DH has been described as a tooth wear phenomenon (Addy 2002). It often occurs with intact periodontal tissues when cervical enamel is removed and coronal dentine exposed and in combination with non-carious cervical lesions (NCCLs) or following gingival recession. Therefore, it is important to recognise the clinical features of tooth wear, in particular erosion. These will be discussed in greater detail together with gingival recession and oral hygiene practices below.
Tooth Wear and Gingival Recession
Tooth wear is the irreversible, non-traumatic loss of dental hard tissues due to aetiological processes classified as erosion, attrition, abrasion (Bartlett and Smith 2000; Ganss et al. 2006) and abfraction (Lee and Eakle 1984). Tooth wear may be considered a normal part of ageing or a physiological process, from the anthropological perspective (Whittacker 2000). Historically, it has been suggested that the human dentition is designed to wear and that this process is important to optimise the functional capabilities of the dentition (Berry and Poole 1974). Within dentistry, Smith and Knight first distinguished physiological and active or pathological tooth wear (Smith and Knight 1984). Tooth wear may be defined as pathological as opposed to physiological if it appears in relatively younger patients and the rate of progression of tooth wear is fast. Crucially, physiological tooth wear allows time for the pulp to lay down reparative or secondary dentine, which could prevent fluid flow within the dentine tubules and may also reduce DH (Krauser 1986). In contrast, pathological tooth wear may cause DH as well as other aesthetic and functional concerns (Smith and Knight 1984; Al-Omiri et al. 2006). During pathological tooth wear, the symptoms of DH are reported more frequently (Smith and Knight 1984; Absi et al. 1987; Addy and Pearce 1994; Dababneh et al. 1999; Addy 2000, 2002). As pathological tooth wear lesions progress into dentine, the radius of dentine tubules become larger and the distance to the pulp decreases. As a result, the hydraulic conductance of fluid within dentine would be expected to increase together with an increase in symptoms associated with DH (Pashley 1990). However, exposure of dentine (as a result of tooth wear or gingival recession) will not necessarily lead to the presence of DH per se (Absi et al. 1987; Yoshiyama et al. 1996; Addy 2002). Therefore, tools to measure tooth wear in terms of surface loss alone may not necessarily reflect the presence of DH.
For the purposes of understanding the aetiological processes involved in DH, the names ‘lesion localisation’ and ‘lesion initiation’ were proposed (Dababneh et al. 1999; Addy 2002). Lesion localisation involves dentine exposure, which may occur as a result of enamel or dentine wear or gingival recession. Lesion initiation may arise following lesion localisation and involves the exposure of patent or un-occluded dentine tubules from the surface of dentine to the pulp. This often occurs following loss of the smear layer. It should be noted that in addition to the smear layer or other surface occlusions, the degree of sclerosis by peritubular dentine and the extent of occlusion by reparative dentine on the pulpal surface might also affect the capacity for fluid movement within the dentine tubules (Yoshiyama et al. 1996). Dentine exposure will not necessarily lead to DH if the dentine tubule system is not patent. Indeed, clinical observation studies show that DH can be uncommon even in cases where the pulp is visible through a thin bridge of dentine (Bartlett and Ide 1999), which is likely to consist of sclerotic or transparent dentine.
Erosion and DH
Erosion is currently considered to be the most common and important aetiological factor for tooth wear in Europe (Seligman et al. 1988; Deery et al. 2000; Addy and Hunter 2003; Grippo et al. 2004; Lussi 2006). This process is due to the superficial demineralisation of hard tissue and the chemical dissolution of the apatite crystals in enamel by an acid (Bartlett 2005) that is not produced by the oral flora but from intrinsic or extrinsic sources (ten Carte et al. 2008), both of which are strongly associated with DH (West et al. 2013a). Typical sources of intrinsic acid are regurgitated stomach acid containing hydrochloric acid (HCL) due to vomiting or gastro-oesophageal reflux (Scheutzel 1996). Typical extrinsic sources are provided below (Zero 1996; Lussi 2006) (Fig. 4.1):
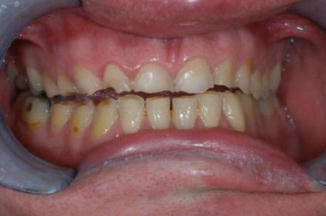

Fig. 4.1
An example of a patient with severe erosion, which has initiated a loss of crown height and an anterior open bite due to gastric erosion
-
Diet (e.g. acidic citrus and other fruits, carbonated beverages and sports drinks, beers and herbal teas, vinegars and pickles, candies)
-
Medicaments (e.g. non-encapsulated HCL replacement, chewing ascorbic acid tablets (vitamin C) and acetylsalicylic acid tablets (aspirin), iron tablets, salivary stimulants)
-
Occupation (e.g. jobs involving wine tasting or working near acidic industrial vapours)
-
Sports (e.g. improperly chlorinated swimming pools)
There is an increasing body of literature indicating that acid erosion caused by relatively small acidic challenges will lead to loss of enamel and dentine and expose the dentine tubules and initiate DH. This literature included laboratory research (Addy et al. 1987b; Absi et al. 1992; West et al. 1999; Vanuspong et al. 2002; Gregg et al. 2004; Ganss et al. 2009), review papers (Addy and Hunter 2003; Addy 2005; Zero and Lussi 2005; Lussi 2006), clinical research (Absi et al. 1992; Hughes et al. 1999; Hunter et al. 2000; Olley et al. 2012, 2014a) and prevalence studies (Lussi and Schaffner 2000; Smith et al. 2008; West et al. 2013b).
Erosive acid challenges are important in removing the smear layer and pellicle (on the tooth surfaces exposed to saliva) and initiating DH. In two clinical studies, cavities were prepared in dentine and hydrostatic pressures were applied to the exposed dentine. Patients reported sensations of short sharp pain in those lesions in which an acid challenge was used to remove the smear layer from the surface of the prepared cavity but not in lesions in which the smear layer was present (Brannstrom 1965; Ahlquist et al. 1994). This can be easily demonstrated in the laboratory. For example, Figure 4.2 shows a high-powered scanning electron microscopy image of the surface of root dentine taken from the buccal cervical region of a premolar tooth. Following treatment of the surface of the dentine with a 6 % solution of citric acid for one minute under agitation, the smear layer was removed and the dentine tubules become visible. This work is supported elsewhere (Pashley et al. 1981; Addy et al. 1987a). Most of the dentine tubules are greater than 1 μm in diameter post-acid challenge. This is greater than 0.83 μm, the minimum diameter reported as being required to elicit DH at the cervical area of the tooth near the dentino-enamel junction (DEJ) (Absi et al. 1987). The effects of acids are to remove the smear layer and expose patent or un-occluded dentine tubules. It was concluded in these studies that the presence of these patent dentine tubules was related clinically to DH.


Fig. 4.2
Scanning electron micrograph (SEM) images (×2,000) of untreated root surface (left) and root surface following a 1 min 6 % citric acid challenge with gentle agitation (right). Scale bar 2 μm
There are a number of food products that contain acids which are popular consumables in the United Kingdom. These are summarised, together with pH and pKa values (explained below), in Table 4.2.
Table 4.2
Erosive products (with associated acids) available in the United Kingdom with pKa and pH values
|
Beverage
|
Acid
|
pKa (titratable acidity)
|
pH
|
|---|---|---|---|
|
Citric fruits including oranges, lemons, grapefruit
|
Citric acid
|
3.14
|
2.2
|
|
Apples, plums and peaches
|
Malic acid
|
3.4
|
2.2
|
|
Grapes and wines
|
Tartaric acid
|
2.89
|
2.2
|
|
Fermented products and yoghurt
|
Lactic acid
|
3.86
|
2.4
|
|
Preservative
|
Acetic acid
|
4.76
|
2.9
|
|
Rhubarb
|
Oxalic acid
|
4.14
|
1.3
|
|
Cola drink
|
Phosphoric acid
|
2.15
|
1.5
|
Despite the acidity of these products, not all popular erosive beverages will initiate the exposure of the dentine tubules. This was demonstrated in an early study, which investigated the effect of various dietary beverages on dentine and reported that many of these popular erosive beverages of acidic or ‘low’ pH would not lead to exposure of the patent dentine tubules (Addy et al. 1987a). These beverages included a low-pH carbonated drink, Coca-Cola and Ribena (a fruit-based soft drink). Instead, consumables, for example, red and white wine, citrus fruit juices, apple juice and yoghurt, produced visible dentine tubules as observed by SEM (Addy et al. 1987a). It should be pointed out that Addy, Absi et al 1987 examined smear layer removal and not tooth structure loss. Citric acid was described at the time as most detrimental to human enamel (Meurman et al. 1987). Grenby et al. then demonstrated that titratability was likely to be more important than pH in determining the erosive potential of carbonated drinks, fruit juices etc (Grenby et al. 1989). For example, the Coca-Cola drink and Ribena were observed to have substantially lower titratable acidities in contrast to fruit juices (lemon, orange and pineapple), and they were therefore less acidic. Titratable acidity or neutralisable acidity (pKa) is the volume of alkali required (typically 0.1 mol solution of sodium hydroxide) to raise the pH of a standardised volume of beverage (typically 25 ml) to pH 7 (Chadwick 2006). In dental erosion, titratability provides an indication of the actual concentration of hydrogen ions available to interact with a mineralised surface, which provides an indication of the erosive potential of a particular acid challenge (Zero 1996). In addition to a higher titratable acidity and lower pH, the severity of an erosive challenge is also likely to increase with the duration of the acid challenge, temperature and ion concentration, frequency of the acid challenge and the presence of chelating agents (Moss 1998; West et al. 2000; Wiegand et al. 2007). The latter is related to the calcium-binding property of the acid. Acids, for example, citric, malic and tartaric acid, contain more than one carboxyl group in their chemical composition (di and poly carboxylic acids), and this will result to the binding of more than one soluble calcium ion complexes at high pH (Meurman et al. 1987). Calcium binding from the saliva will result in the loss of the ion effect of calcium in saliva, which will lead to more dissolution tendency. Also, if calcium from the saliva is bound, there may be a tendency for more dissolution of the dental tissue in order to replace the calcium lost in saliva (Meurman and tenCate 1996). This will result in further erosion of the surface layer, due to the gradual release of tooth mineral due to a buffering action (Grenby et al. 1989). As a result, fruit juices, for example, orange or pineapple juice, will result in more demineralisation than other popular erosive beverages such as the Coca-Cola and lemonade drinks, which contain phosphate (Grenby et al. 1989). Despite their low pH, the phosphate in Cola drinks raises the degree of saturation of the solution with respect to tooth mineral; Calcium and phosphate. Furthermore, in contrast to most other organic acids, the calcium salt of oxalic acid is insoluble. Foods containing this acid should be therefore less erosive in nature.
The importance of a mature salivary pellicle in providing a protective role during erosion must not however be underestimated; in particular the phosphate, calcium and fluoride content of an erosive challenge may prevent dental wear (Zero and Lussi 2005), and sleeping medications (which may reduce salivary flow) are associated with more reported DH (West et al. 2013a). Currently there is ongoing research in this area, and clinical experiments have demonstrated that the salivary pellicle forms a protective layer against erosion (Moazzez et al. 2014).
Abrasion and DH
Abrasion is a physical process, which occurs as a result of the mechanical wear of dental tissues by foreign bodies. Toothbrushing and toothpaste formulations are common forms of dental abrasion (Addy and Hunter 2003; Addy and West 2013). Toothbrush abrasion is influenced by brushing habits, force applied and the time spent brushing (Hooper et al. 2003). There are additional habits linked to abrasion, for example, onychophagia, clips and other tools, which may come into contact with teeth. Unlike erosion, there are limited data to support the importance of abrasion in causing DH, but toothbrushing with a toothpaste has been previously implicated in the aetiology of DH (Addy and Hunter 2003; Abrahamsen 2005; Bartlett and Shah 2006; Ganss et al. 2009; Addy and West 2013) (Fig. 4.3).
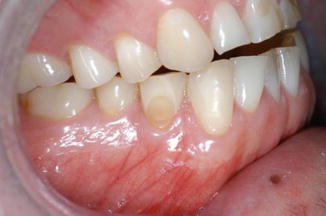

Fig. 4.3
An example of abrasion on the buccal cervical region commonly referred to as a non-carious cervical lesion (NCCL)
According to Addy (2005), the effects of normal toothbrushing on wear of the enamel are negligible and unlikely to lead to exposure of the underlying dentine alone unless erosion is also occurring. Normal toothbrushing, even for extended periods of time (measured in years), will also cause limited wear of dentine, and the wear may be limited to the smear layer, which would presumably have a subsequent effect on DH (Absi et al. 1992). Increasing the force of toothbrushing can, in addition, result in increased tooth surface loss in dentine. Manual, as opposed to electric toothbrushing, has been demonstrated to cause more dentine wear because the force applied with a manual toothbrush was often higher (Knezevic et al. 2010; Van der Weijden et al. 2011). Similarly, prevalence studies have demonstrated that brushing with medium and hard, rather than soft, stiffness toothbrushes will cause more dentine wear (Smith et al. 2008). It has also been reported that when greater forces are applied to healthy dentine using a manual toothbrush, patients are more likely to report pain that resembles DH (Addy 2005).
Toothbrushing with a toothpaste adds an abrasive component. Nonetheless, normal toothbrushing with toothpaste formulations (with the exception of non-hydrated alumina) in the absence of acid is likely to cause little or no wear of enamel and dentine (Hunter et al. 2002; Addy 2005). Abnormal or abusive use (e.g. using excessive toothpaste) can however lead to pathological wear (Hunter et al. 2002; Hooper et al. 2003; Turssi et al. 2010). The filament stiffness of toothbrushes may also be important in dentine wear initiated by a toothpaste. Laboratory studies have demonstrated that smaller filament stiffness (decreasing diameter of filament) initiated more wear in dentine using various abrasives of toothpaste formulations (post-acid erosion) and that the abrasivity of the dentifrice is more important than the filament stiffness (Wiegand et al. 2009). This would suggest that multifilament soft brushes may retain more toothpaste, which in turn might lead to more tooth wear.
Dentifrice (toothpaste) abrasivity is normally measured using the RDA (Relative Dentine Abrasivity), which has a numeric value. The International Organization for Standardization guidelines state that for dentine, the abrasivity of test formulation should not exceed 2.5 times the reference abrasive, e.g. RDA must not exceed 250 (International Organization for Standardization (ISO) 11609, 1995). Interestingly, the allowed pH range for a toothpaste (pH 4–10) might be more a cause for concern as this would suggest that some toothpaste formulations of low pH could intrinsically lead to chemico-physical dental wear. Despite this, ISO standards ensure that all toothpaste products are above a pH that may cause demineralisation (pH 5.5 for enamel and pH 6.5 for dentine) or the contained fluoride balances the low-pH effect (Hunter et al. 2002). Toothpaste formulations saturated with dental mineral and in particular fluoride actively encourages remineralisation due to their buffering capacity and consequently abrasion is less (Betke et al. 2003; Zero and Lussi 2005).
In summary, although toothpaste formulations are unlikely to cause DH lesion localisation, toothpaste formulations of higher abrasivity or overzealous brushing or use of a toothpaste may initiate dentine wear and DH lesion initiation by removal of the smear layer and establishment of patent dentine tubules (Addy and Hunter 2003). Despite this, some toothpaste formulations, especially those containing silica, may have a therapeutic effect in preventing DH by partially occluding the dentine tubules (Addy and Mostafa 1989; West et al. 2002
Stay updated, free dental videos. Join our Telegram channel

VIDEdental - Online dental courses


