Chapter 9 The immune response
Chapter 8 described the development of B and T cell repertoires. At birth, the immature immune system consists of B cells selected for low-affinity antibody production, while the T cell repertoire consists of T cell antigen receptors (TCRs) potentially able to recognize foreign but usually not self peptides presented by major histocompatibility complex (MHC) molecules on antigen-presenting cells (APCs). The latter must also provide co-stimulatory signals for full T cell activation.
Antibodies
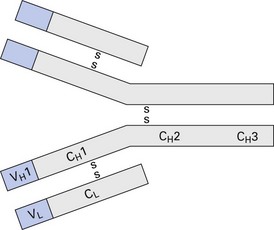
The basic Y-shaped, four-chain structure of the antibody molecule is shown in Figure 9.1. Antigen-binding specificity is provided by the combined variable (V) regions of heavy (H) and light (L) chains. Since the basic Ig unit has two such pairings, the molecule can bind two identical epitopes; i.e. it is bivalent. The Ig heavy-chain constant region, particularly domains 2 and 3, which make up the Fc region, largely determines the biological activity of the molecule.
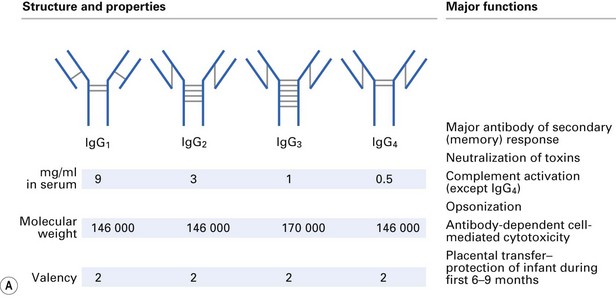
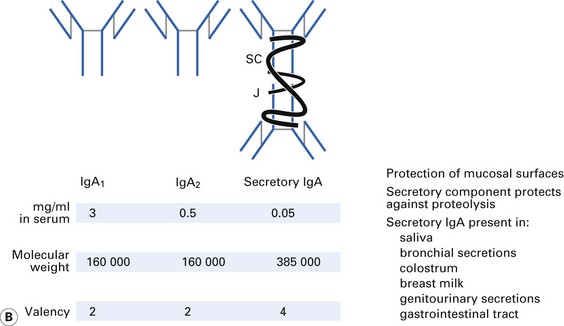
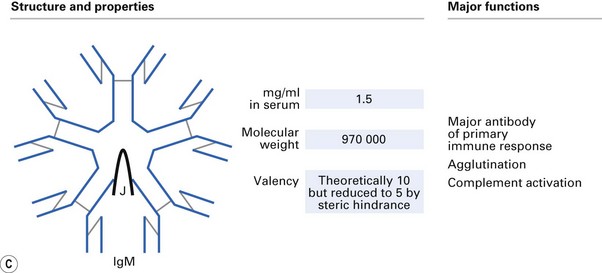
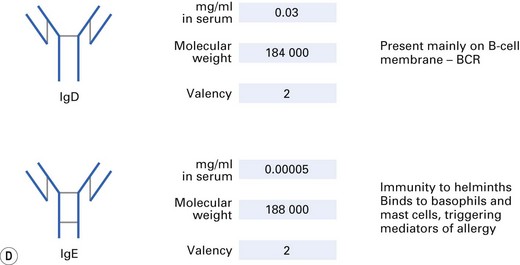
There are five distinct classes of Ig (IgG, IgA, IgM, IgD, IgE), four subclasses of IgG (IgG1, IgG2, IgG3, IgG4) and two subclasses of IgA (IgA1, IgA2). These are derived from usage of different heavy-chain genes, as described in Chapter 8. The different structures and properties of Ig molecules are summarized in Figure 9.2.
Cytokines
Table 9.1 outlines the main sources and activities of cytokines. It is not exhaustive, and new cytokines and activities are undoubtedly awaiting discovery. The exciting field of cytokine research has led to the isolation of genes for cytokines and their receptors and inhibitors and the ability to manufacture these molecules by recombinant DNA technology. There is optimism that therapeutic use of these reagents will, in the near future, benefit patients with infections, autoimmunity, allergy and other immunologically mediated diseases.
| Cytokine | Main producers | Major actions |
|---|---|---|
| IL-1 | Macrophages | Mediator of inflammation; augments immune response |
| IL-2 | T cells | T cell activation and proliferation |
| IL-3 | T cells | Haematopoiesis (early progenitors) |
| IL-4 | T cells | T cell, B cell, mast cell proliferation; IgE production |
| IL-5 | T cells | B cell proliferation; IgA production; eosinophil, basophil differentiation |
| IL-6 | Macrophages, T cells | Mediator of inflammation; B cell differentiation |
| IL-7 | Bone marrow cells, thymic stroma | Haematopoiesis (lymphocytes) |
| IL-8 | Macrophages | Neutrophil chemotaxis |
| IL-9 | T cells | T cell proliferation |
| IL-10 | Macrophages, T cells | Inhibitor of cytokine production |
| IL-11 | Bone marrow stromal cells | Haematopoiesis (early progenitors) |
| IL-12 | Macrophages | T cell differentiation |
| IL-13 | T cells | Similar to IL-4 |
| IL-14 | T cells | Proliferation of activated B cells |
| IL-15 | Stromal cells | Similar to IL-2 |
| IL-16 | T cells | T cell chemotaxis |
| IL-17 | T cells | Mediator of inflammation and haematopoiesis |
| IL-18 | Macrophages | Similar to IL-12 |
| IFN-α | Leukocytes | Activation of macrophages, NK cells; upregulation of MHC expression; protection of cells against virus infection |
| IFN-β | Fibroblasts | |
| IFN-γ | T cells, NK cells | |
| LT | T cells | Mediator of inflammation; killing of tumour cells; inhibition of tumour growth |
| OSM | Macrophages, T cells | |
| TGF-β | Macrophages, lymphocytes, endothelial cells, platelets | Wound healing; IgA production; suppression of cytokine production |
| TNF-α | Macrophages, T cells | Mediator of inflammation; killing of tumour cells |
| gCSF | Macrophages | Haematopoiesis (granulocytes) |
| mCSF | Monocytes | Haematopoiesis (monocyte/macrophages) |
| gmCSF | T cells | Haematopoiesis (granulocytes, monocyte/macrophages) |
IL, interleukin; IFN, interferon; LT, lymphotoxin; OSM, oncostatin M; TGF, transforming growth factor; TNF, tumour necrosis factor; CSF, colony-stimulating factor; g, granulocyte; m, monocyte/macrophage; NK, natural killer; Ig, immunoglobulin; MHC, major histocompatibility complex.
B cell activation
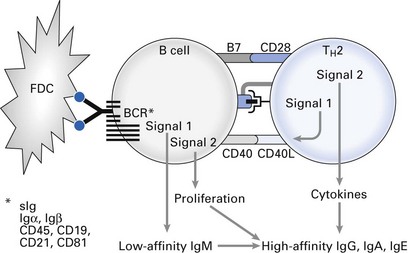
B cells are highly efficient APCs. They receive signal 1 for activation by binding antigen, often concentrated on the surface of follicular dendritic cells within lymph node germinal centres, to the BCR, and then proceed to internalize antigen and process peptides on to MHC II molecules for presentation to T-helper cells (Fig. 9.3). They are then induced to express co-stimulatory B7 and can therefore provide signal 2 for T-helper cell activation through CD28. Activated T-helper cells are induced to express CD40L for binding to B cell CD40. Interaction between these two molecules induces B cell activation, Ig production and isotype switching.
Stay updated, free dental videos. Join our Telegram channel

VIDEdental - Online dental courses