Chapter 9
Neuromuscular Blockers
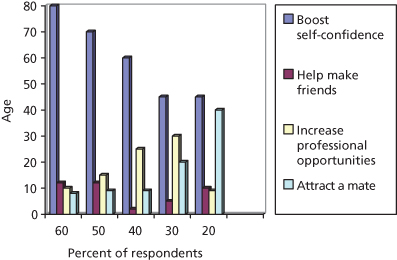
The attainment and maintenance of beauty and youthfulness has long been associated with vitality and wellness. As early as 1500 BC, people have tried a variety of treatments such as ingesting tiger gonads to avoid aging and restore youth. One has only to peruse popular magazines to see numerous advertisements for treatments and interventions that claim to defy or correct the signs of aging. The BeautyforLife website recently conducted a survey of men and women in their 20s to 60s and asked about “the most important reason to maintain your appearance.” Across all age groups, the overwhelming reason was to boost one’s self-confidence, followed by “increasing professional opportunities,” “attracting potential partners,” and “to help make friends”(Figure 9.1). This drive has led to the explosion in development of nonsurgical and minimally invasive aesthetic treatments.
Figure 9.1 BeautyforLife user survey result: “Which of the following best describes the most important reasons to maintain your appearance?”
(From http://beautyforlife.com/beauty-survey-results.aspx.)
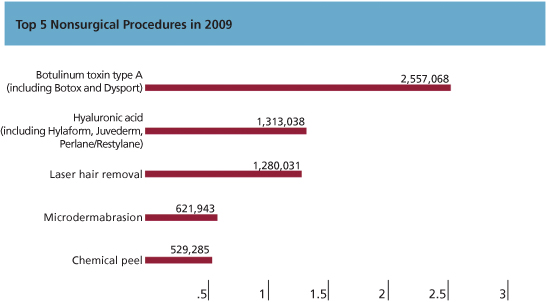
Selective neuromodulation of the muscles of facial expression using botulinum toxin type A (BTX-A) has led the way in the treatment of dynamic facial rhytides. According to the American Society of Aesthetic Plastic Surgeons 2008 statistics, more than 2 million botulinum toxin injections were performed, making it the most frequently performed nonsurgical procedure (Figure 9.2).
Figure 9.2 American Society of Aesthetic Plastic Surgeons 2009 statistics on nonsurgical procedures.
(With permission.)
As aging occurs, genetics, environmental exposures, and dynamic facial expression lead to the formation of wrinkles. To correct this, botulinum toxin may be used as a stand-alone treatment or used in combination with injectable fillers, energy-based therapies, and/or surgery. By balancing facial dynamics and reducing the activity of mimetic muscles of the face, a more youthful appearance is produced. With further knowledge and experience, botulinum toxin will continue its ever-expanding role in aesthetic medicine.
History
Although it seems commonplace today to discuss the use of botulinum toxin openly, at one time the mention of it spread fear of death. In 1895 at a picnic, 34 attendees fell desperately ill after eating raw salted ham. Three of these patients eventually succumbed to the disease known as botulism. It begins with symptoms such as blurred vision, dry mouth, nausea, and dizziness and progresses to flaccid paralysis and eventually death. Botulism derived its name from the Greek word for sausage because early outbreaks were thought to be associated with ingestion of contaminated sausage.
Professor Emile Pierre van Ermengen recognized the pattern of progressive paralysis in the patients from the picnic and worked to isolate the causative agent. He named the bacterium Bacillus botulinus. It is now known as Clostridium botulinum, an anaerobic, spore-forming rodlike bacterium. Further studies on the new isolate reveal a number of hypotheses that we know are key characteristics of botulinum toxin. Foremost, a toxin found in contaminated food creates the symptoms of botulism. The toxin is relatively stable to mild chemical agents but not heat. Not all animal species react similarly to the toxin.
Isolation and purification for its use as a biological weapon dominated the next stage of development. Herman Sommer, MD, at the University of California, San Francisco, first isolated BTX-A as a purified stable acid precipitate in the 1920s. In 1946, Edward J. Schwartz, PhD, purified the toxin into a crystalline form that allowed further study of its molecular structure in detail. By 1949, the mechanism of action of the toxin had been discovered. This led to early medical testing in the 1950s by Dr. Vernon Brooks and the possibility of the type A toxin as a therapeutic agent.
Trials of BTX-A began in the late 1960s by Alan B Scott, MD, at the Smith-Kettlewell Eye Research Foundation in San Francisco. He hypothesized that selective weakening of ocular muscles in monkeys could provide an effective nonsurgical therapy for strabismus. His results published in 1973 confirmed his theory. While collaborating with Dr. Shantz, the first batch of BTX-A for human treatment was produced and trialed in 1977. By 1979, the Food and Drug Administration (FDA) approved “botox” for limited use in humans for the treatment of strabismus. Dr. Scott’s company, Oculinum Inc., continued to study BTX-A treatment in human volunteers and expanded the indications to include blepharospasm in 1985 and hemifacial spasm in 1989. Allergan (Irvine, CA) acquired the rights to distribute BTX-A in 1988 and purchased Oculinum in 1989. BOTOX® was born.
A pivotal study in 1992 by Drs. Jean and Alastair Carruthers evaluated the use of BTX-A for the treatment of glabellar frown lines. They performed further investigation comparing BTX-A and nonanimal stabilized hyaluronic acid (NASHA) for the treatment of severe glabellar rhytides in adult females. These results, published in 2003, helped achieve FDA approval for BOTOX Cosmetic® in 2002 for the temporary improvement in the appearance of moderate to severe glabellar lines in adult women and men ages 18–65 years. Numerous other studies have been published documenting the safe and effective usage of BTX-A for the treatment of dynamic wrinkling in the forehead, periorbital, and neck areas. BTX-A continues to provide an expanding role in our clinical armamentarium.
Physiology
Currently, seven distinct serological subtypes of botulinum toxin exist (A, B, C, D, E, F, and G). Type A is the most effective in humans, although types B and E also show activity. Botulinum neurotoxin acts as a zinc-dependent endoprotease that prevents the release of acetylcholine at the neuromuscular junction (NMJ) of striated muscle, thus creating a flaccid paralysis. BTX-A is a complex structure composed of a 150-kDa BTX-A molecule coupled with hemagglutinin and nonhemagglutinin protective proteins. Various botulinum toxin complexes are purported to weigh in the range of 300–900 kDa depending in the coupled proteins. The proteins are held together by covalent bonds that are stable in an acidic environment but dissociate to release the neurotoxin as the pH in the blood stream rises.
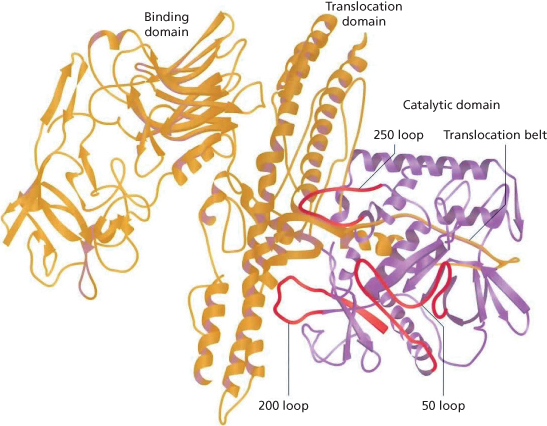
The BTX-A neurotoxin molecule consists of a 100-kDa heavy chain and a 50-kDa light chain held together by disulfide bonds and associated with a zinc molecule (Figure 9.3). The heavy chain is responsible for the irreversible binding to the presynaptic nerve membrane. Following binding, the endoprotease is internalized and the disulfide bond cleaved, releasing the light chain. The light chain cleaves proteins in the synaptic fusion complex inhibiting release of acetylcholine from preformed vesicles in the nerve terminus creating flaccid paralysis.
Figure 9.3 Structure of 150-kDa botulinum toxin A.
(Figure 2.2; Rohrer T, Beer K. Background to botulinum toxin. In: Carruthers A, Carruthers J (eds.). Botulinum Toxin. Philadelphia: Saunders/Elsevier, 2008.)
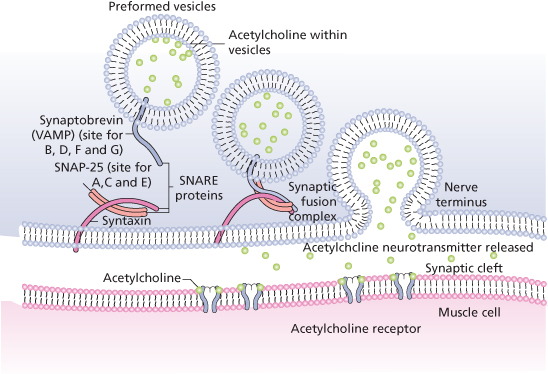
Normal neuromuscular activity occurs when an action potential travels down a nerve and, upon reaching the nerve ending, releases acetylcholine from numerous small vesicles at the NMJ. The synaptic fusion complex responsible for this release is a set of proteins that allow the vesicles to dock and fuse to the cell membrane. These proteins are called soluble N-ethylmaleimide-sensitive factor attachment protein receptors (SNARE proteins).
Two sets of SNARE proteins are required for the docking and acetylcholine release, one on the vesicle (VAMP-2/synaptobrevin) and one on the membrane wall (syntaxin 1A and SNAP-25). The membrane-associated SNARE proteins form the bridge that leads to docking and release. It is at these SNARE proteins that the various botulinum toxin types interact (Figure 9.4). If any of the SNARE proteins are damaged, neurotransmitter release is inhibited and the associated muscle cell is rendered flaccid. Botulinum toxin types A, C, and E act to cleave the SNAP-25 protein, whereas type B works on synaptobrevin. Although striated muscle is most sensitive to BTX-A, nociceptors and autonomic cholinergic nerves are also affected. Return of function starts with peripheral sprouting at the terminal nerve ending. This begins about 28 days after treatment. As new SNAP-25 proteins regenerate, the peripheral sprouts retract and disappear. Normal function is evident 2–5 months after treatment and is dose dependent. Evidence suggests that with repeated treatment at regular intervals, some level of atrophy occurs.
Figure 9.4 Illustration of the SNARE proteins in the synaptic fusion complex at the neuromuscular junction.
(Figure 2.5; Rohrer T, Beer K. Background to botulinum toxin. In: Carruthers A, Carruthers J (eds.). Botulinum Toxin. Philadelphia: Saunders/Elsevier, 2008.)
Packaging and Reconstitution
One vial of BOTOX Cosmetic (onabotulinumtoxinA) contains 100 units of lyophilized BTX-A, sodium chloride, and human albumin. Dysport™ (abobotulinumtoxinA, Scottsdale, AZ), another commercially available neurotoxin, contains 300 units of lyophilized BTX-A plus lactose and albumin. Vials are packaged and shipped in a Styrofoam container with dry ice anticipating delivery within 24 hours of shipping. Upon arrival at the office, the vials should be immediately placed into an appropriate storage container that maintains a temperature within a range of 2–8°C. Vials may be stored at this temperature for up to 36 months for a 100-unit vial or up to 24 months of a 50-unit vial of BOTOX Cosmetic.
Reconstitution is accomplished by adding either 0.9% sterile preserved or preservative-free saline to the vial. Dilution technique is performed using a 21-gauge needle and an appropriate-size syringe to draw up the appropriate volume of saline. The needle is inserted into the stopper and the vacuum in the BOTOX Cosmetic vial should pull the diluent into the vial.
Upon reconstitution, the manufacturer of BOTOX Cosmetic recommends administration of the product within 4 hours. During this time, the product should be stored in a refrigerator between 2 and 8°C. The dilution ratio is based on anticipated clinical results varying widely among physicians. Table 9.1 shows standard dilutions and corresponding concentrations of active neurotoxin per 0.1 cc of injectate. It is recommended that each practitioner evaluate the benefits of various dilution ratios and develop a comfort level with a single standard dilution.
Table 9.1 Reconstitution ratios for BOTOX Cosmetic®
| 1 cc | 10.0 units |
| 2 cc | 5.0 units |
| 2.5 cc | 4.0 units |
| 4 cc | 2.5 units |
In the author’s practice, BOTOX Cosmetic is diluted with 1 cc of preservative-free 0.9% normal saline for facial aesthetics and 2 cc for hyperhydrosis. The benefit of a higher dilution ratio is the potential for diffusion.
Care of the product, once reconstituted, has frequently been debated. A recent study evaluated gentle handling versus shaking of the product with no statistical difference in efficacy between the two. Another investigation reviewed duration of action between bottles that were reconstituted 2 weeks, 4 weeks, and even 6 weeks prior to the time of injection. In 88 patients studied, no statistically significant difference in efficacy or duration was seen at any time frame after reconstitution.
Dysport™ reconstitution is recommended at 1.0 cc of 0.9% sterilized preservative-free normal saline per 300 or 500 units of toxin. It is important to realize that this produces a solution that has three to five times as many units per cubic centimeter as does BOTOX. However, its onset and duration of action appear to be approximately similar to BOTOX Cosmetic.
Clinical Anatomy
Clinical use of BTX-A is only effective if appropriate muscular anatomy and its relationship to dynamic evaluation of the face are understood.
The face is composed of a series of muscles that either depress or elevate structures or regions (Table 9.2). Coupling comprehension of anatomy with evaluation of muscle volume and strength leads to an appropriate treatment template, meticulous execution of that plan, and an aesthetically pleasing balance of facial dynamics. A thorough understanding of the involved muscles as well as the underlying vascular and nerve anatomy also helps to minimize the chance of injection-related complications.
Table 9.2 Dynamic balance of the face. Muscles that lift and muscles that depress
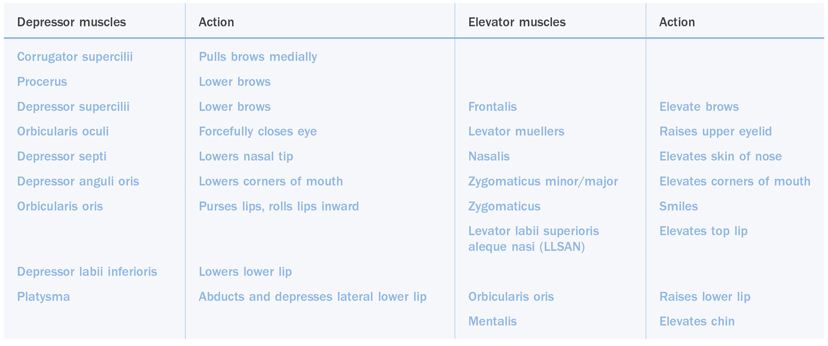
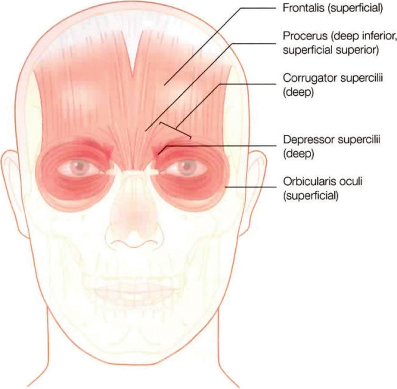
For the purposes of evaluation and treatment, the face is generally divided into thirds. Although each third may be evaluated independent of other thirds, they ultimately impact upon each other. The upper third of the face includes the frontalis, procerus, corrugator supercilii, depressor supercilii, and the orbicularis oculi muscles (Figure 9.5). One assesses this area of the face by observing transverse forehead lines, glabellar creases, the lateral orbital or crow’s feet creases, and brow position. This is the most common region treated with BTX-A and influences the appearance of mood and facial expression more than any other region of the face. Injection of BTX-A in this area may have a profound effect on brow position and facial expression.
Figure 9.5 Upper third facial muscles.
(Figure 3.1; Finn JC, Cox SE. Background to botulinum toxin. In: Carruthers A, Carruthers J (eds.). Botulinum Toxin. Philadelphia: Saunders/Elsevier, 2008.)
Frontalis Muscle
The frontalis muscle is the sole elevator of the brow. It originates on the galea aponeurotica along the coronal suture and inserts into the dermis at the level of the eyebrow in the arcus marginalis at the superior orbital rim. At this level, it interdigitates with fibers from the procerus, corrugator supercilii, and orbicularis oculi muscles. Clinically, two muscle patterns are seen, either one continuous muscle or two distinct muscle bellies separated in the midline. Transverse forehead rhytid patterns correlate to the underlying muscular anatomy. The most common is a single muscle with continuous transverse forehead lines.
Corrugator Supercilii Muscle
Many descriptions of the corrugator supercilii muscle, frequently called the “frowning muscle,” exist. The corrugator supercilii acts to depress and adduct the eyebrow, moving it inferiorly and medially. Repeated contraction of this muscle leads to vertical glabellar creases, also known as the “the elevens.” Impaired corrugator muscles originate from the medial supraorbital ridge of the frontal bone starting deep and then inserting via interdigitations with the frontalis muscle on the skin in the region of the midbrow.
Two common muscle patterns predominate: a short narrow corrugator supercilii with a more medial insertion; and a more common wide-based lateral insertion toward the midbrow region (Figure 9.5). The clinical appearance of muscle contracture for each muscle type is distinct. Understanding of this muscular anatomy directs the optimal injection pattern for the desired clinical results.
Procerus Muscle
The procerus muscle is a thin midline muscle, known as a brow depressor. The origin of the muscle is deep at the level of the periosteum of the nasal bone and the insertion is into the glabellar or midforehead dermis. Activation of this muscle can produce transverse lines over the nasal bridge (Figure 9.5).
Depressor Supercilii Muscle
Existence of the depressor supercilii muscle has been debated; however, multiple cadaver dissections reveal a distinct muscle pattern with origin along the periosteum of the nasal portion of the frontal bone approximately 1 cm above the medial canthal ligament and insertion into the dermis just inferior to the medial head of the eyebrow (Figure 9.5). The primary function of this muscle is to depress the brow medially.
Orbicularis Oculi Muscle
The orbicularis oculi is a broad-based sphincter muscle with three distinct portions. It is divided into the pretarsal, preseptal, and orbital portions. The muscle originates from bony origins near the medial canthus and has multiple dermal insertions. It is intermittently associated with very thin eyelid skin. The medial portion acts as a brow depressor medially. The upper orbicularis acts as a brow adductor and laterally it acts as a lateral brow depressor. It is this lateral segment of the muscle that is responsible for producing the lateral orbital wrinkles known as crow’s feet. There is extensive variability in activity and crow’s feet patterns among patients. However, this area responds quite well to treatment with BTX-A.
The middle third of the face is one of the most challenging areas to understand anatomically and therefore to treat clinically. Muscular structures are complex (Figure 9.6). The most common aesthetic issues treated in this complicated region include abnormalities of the nasolabial folds and the lips (asymmetries, smile-related problems, and rhytides).
Stay updated, free dental videos. Join our Telegram channel

VIDEdental - Online dental courses


