Chapter 8
Aesthetic Facial Implants
Introduction
The last four decades have seen an increase in the use of facial implants in aesthetic surgery. Alloplastic materials offer a long-term solution to augment skeletal deficiencies, restore facial contour irregularities, and rejuvenate the face. Implants are used throughout the face with commonly augmented areas including as follows: the cheeks to balance the effects of malar hypoplasia; submalar and midfacial implants to augment the hollowness found with aging; nasal implants for dorsal augmentation; mandibular augmentation to create a stronger jaw profile and better nose–chin relationship; mandibular prejowl and angle implants to augment traditional cervicofacial rhytidectomy; and premaxillary implants to augment a retrusive midface. The marked improvement in biomaterials, in addition to the development of computer-assisted custom-designed implants, now provide solutions for more complex facial defects due to trauma, congenital deformities, or lipoatrophy.1,2
The concept of facial contouring includes changing the shape of the face. Modern hallmarks of youth and beauty are distinguished by bold facial contours emphasized by convex malar-midface configurations and a distinct, well-defined jawline. Any of these promontories that are too small or too large influence the aesthetic importance of the others. For example, reducing the nasal prominence results in increased definition of volume and projection of both the malar-midface and the mandibular-jawline. Similarly, enhancement of the mandibular or malar-midface volumes makes the nose appear smaller and less imposing. Substantive contour modifications may be surgically produced by judiciously altering mass and volume in various anatomical regions and redistributing the overlying soft tissue.
A thorough understanding and analysis of the face, including proportions, anatomy, and the aging process, is critical to the success of using facial implants. The appropriate implant will depend on the relationship between the bony promontories and the surrounding soft tissue. The individual arrangement of the malar-midface area, nose, and mandible-jawline determine the fundamental architectural proportions and contour of the face. Balance between these structures and the distribution of the overlying soft tissue determines facial beauty and harmony. Augmentation is typically accomplished through selecting implants with the proper shape and design while controlling their position over the facial skeleton and under the soft tissue. As a result, alloplastic facial contouring can be utilized to augment both bony and/or soft tissue anomalies.
Implants and Biomaterials
All implants induce the creation of fibroconnective tissue encapsulation, creating a barrier between the host and the implant.3,4 Adverse reactions are primarily a consequence of unresolved inflammatory response to implant materials. Characteristics of the site of implantation also influence the resultant reaction, including the thickness of overlying skin, scarring of the tissue bed, and underlying osseous architecture that may create a condition for implant instability. For example, implants that are deeply placed with thick overlying soft tissue rarely become exposed or extrude. Other important factors such as prevention of perioperative hematoma, seroma, and infection can significantly reduce interaction between the host and implant, and thereby improve implant survivability.
The Ideal Implant
The ideal implant material should be nontoxic, nonantigenic, noncarcinogenic, resistant to infection, and cost-effective. The implant itself should be easily shaped, conformable, simple to insert, static, and able to permanently maintain its form. Additionally, tailoring the implant to the needs of the recipient area during the surgical procedure should not be time-consuming, should not compromise the integrity of the implant, and it must be easy to autoclave without degradation.
Favorable surface characteristics are important for implant placement and stabilization, and paradoxically equally important to facilitate easy removal and exchange without causing injury to surrounding tissues. Implant immobilization is dependent on their capability to be permanently fixed in place for the lifetime of the patient. The characteristics of the implant material also heavily determine immobilization. For example, silicone elastomer induces the formation of a surrounding capsule that maintains implant position, while expanded polytetrafluoroethylene (ePTFE), which encapsulates to a lesser degree, provides fixation with minimal tissue ingrowth. The various material–host interactions provide certain advantages in different clinical settings. Materials that cause significant tissue ingrowth and permanent fixation are often undesirable, particularly if the patient wishes to revise augmentation characteristics in later years. The natural encapsulation process of silicone and the minimal surface ingrowth in ePTFE products insure immobility yet provide easy exchangeability without damage to surrounding soft tissue.
The ideal implant design should have tapered edges that blend on to the adjacent bony surface to create a nonpalpable and smooth transition to the surrounding recipient area. An implant that is malleable and readily conforms to the underlying structures further reduces mobility, while the anterior surface shape should duplicate the natural anatomical configuration. Newer silicone implants are currently being engineered for enhanced adaptability to the underlying bony surface and surrounding soft tissue. For example, Conform™ implants (Implantech Associates Inc., Ventura, CA) have a new type of grid backing that reduces the stiffness of the silicone elastomer and improves flexibility. Increased malleability to irregular bony surfaces reduces the potential for movement and prevents posterior dead space from occurring between the implant and underlying bone (Figure 8.1). Recent advances in research and development in biomaterial engineering have developed a composite implant (using both silicone and ePTFE) that promises to combine the advantages of both biomaterials for future use in facial implants.5
Figure 8.1 The Conform™ type of implant is made from a softer silicone material and has a grid design on the posterior surface of the implant that reduces its memory to more easily adapt to the underlying bone surface. The grid feature also reduces the chances of implant slippage and prevents displacement.

Implant Biomaterials
Polymeric Materials
Silicone Polymers
Various forms of silicone have been used in clinical settings since the 1950s with excellent safety and efficacy profiles. Silicone is polymerized dimethylsiloxane that can take the form of a solid, gel, or liquid, depending on its polymerization and cross-linking. The gel form of silicone can potentially leak some of its internal molecular substances over time. Recent studies, however, examining the safety of silicone gel as breast implants have shown no objective cause and effect for silicone in producing scleroderma, lupus, collagen vascular, or other autoimmune diseases.6,7
Solid silicone products tend to be more stable as implants, as solid silicone elastomer has a high degree of chemical inertness. It is hydrophobic and extremely stable without any evidence of toxicity or allergic reactions.8 Tissue response to solid silicone implants is characterized by a fibrous tissue capsule without ingrowth. When unstable or placed without adequate soft tissue coverage, implants may produce ongoing moderate inflammation and possible seroma formation. Capsular contracture and implant deformity rarely occur unless the implant is placed too superficially or if it migrates to the overlying skin.
Polymethacrylate (Acrylic) Polymers
Polymethacrylate is supplied as a powdered mixture and is catalyzed to produce a very hard material. The rigidity and hardness of acrylic implants cause difficulty in many of the procedures utilizing large implants inserted through small openings. Furthermore, difficulties may exist in adapting the acrylic form to the underlying bony contour when using preformed implants.
Polyethylene
Polyethylene can be produced in a variety of consistencies and is now most commonly used in a porous form. Porous polyethylene, also known as Medpor® (Porex Surgical, Newnan, GA), causes minimal inflammatory cell reaction. The material is hard, and as such, difficult to sculpt. Additionally, the porosity of polyethylene permits extensive fibrous tissue ingrowth that provides an advantage for enhanced implant stability but makes it extremely difficult to remove.
Polytetrafluoroethylene
Polytetrafluoroethylene is composed of a group of materials that has had a defined history of clinical application. The known brand name was Proplast, which is no longer available in the United States because of the related complications of its use in temporomandibular joints. Under excessive mechanical stress, this implant material was subject to breakdown, intense inflammation, thick capsule formation, infection, and ultimately, extrusion or explantation. It is mentioned here because of its historical significance.
ePTFE
ePTFE was originally produced medically for cardiovascular applications.9,10 Animal studies showed the material to elicit limited fibrous tissue ingrowth without capsule formation and minimum inflammatory cell response. The reaction seen over time compared favorably with many of the materials already in use for facial augmentation. The material has shown acceptable results in subcutaneous tissue augmentation and for use as preformed implants. Due to lack of significant tissue ingrowth, ePTFE offers advantages in subcutaneous tissue augmentation since it can be modified secondarily and relatively easily removed in the event of infection.
Mesh Polymers
The mesh polymers, which include Marlex® (crystalline polypropylene), Dacron® (polyethylene terephthalate), and Mersilene® (polyethylene terephthalate), have similar advantages in their ability to be folded, sutured, and shaped with relative ease, but the materials also promote fibrous tissue ingrowth causing difficulty with secondary removal.
Supramid® (Resorba Wundversorgung, Nürnberg, Germany) is a polyamide mesh derivative of nylon that is unstable in vivo. It elicits a mild foreign body reaction with multinucleated giant cells, and over time causes implant degradation and resorption.11
Metals
Metal implants consist essentially of stainless steel, vitallium, gold, and titanium. Except for use of gold in eyelid reanimation and in dentistry, titanium has become the metal of choice for long-term implantation. The advantages of titanium include high biocompatibility, corrosion resistance, strength, and minimal X-ray attenuation during computed tomographic scanning or magnetic resonance imaging. Titanium is primarily used in craniofacial and dental implant reconstruction and does not lend itself for use in facial augmentation.
Calcium Phosphate
Calcium phosphate or hydroxyapatite materials are not osteoconductive, but do provide a substrate into which bone from adjacent areas can be deposited.12 The granular form of hydroxyapatite crystals are regularly used in oral and maxillofacial surgery for augmenting the alveolar ridge. The block form has been used as interpositional grafts during osteotomies.13 These materials, however, have been shown to be of less value as an augmentation or onlay material due to their brittleness, difficulty in contouring, and inability to adapt to bone surface irregularities and mobility.
Autografts and Homografts
Autografts, available as autogenous bone, cartilage, and/or fat, are limited by donor site morbidity and the amount of available donor material. Processed homograft cartilage has been used in nasal reconstruction, but eventually succumbs to resorption and fibrosis.
Tissue-Engineered Biocompatible Implants
Tissue engineering of implant materials has evolved into a multidisciplinary field in the past few years. Characteristics and properties of synthetic compounds are now manipulated to facilitate delivery of an aggregate composed of dissociated cells into a host in order to recreate new functional tissue. This has evolved by combining scientific advances in multiple fields including materials science, tissue culture, and transplantation.
The protocol consists of seeding cells into a suspension that provides a three-dimensional structure to promote matrix formation. This structure anchors the cells and permits nutrition and gas exchange with the ultimate formation of new tissue in the shape of a gelatinous material.14 A number of tissue-engineered cartilage implants have previously been generated based on these new principles, including joint articular cartilage, tracheal rings, and auricular constructs.
Tissue engineering offers the potential to grow cartilage in a precisely predetermined shape, and presently is in the developmental stage of generating various types of contoured facial implants consisting of immunocompatible cells and matrix.15 Once employed on a commercial basis, these techniques would require minimal donor site morbidity and, like alloplastic implants, reduce operative time.
Surgical Considerations for Alloplastic Implants
General
Patients with prominent, well-balanced, and strong skeletal features will better withstand the negative effects of aging.16 An analysis of the adolescent face reveals an abundance of soft tissue that provides the harmonious composite of youthful facial form. Full cheeks with smooth, symmetrical contours free of sharp, irregular projections, indentations, rhytides, or dyschromias commonly embody these youthful qualities.17 Facial aging is influenced by genetic factors, sun exposure, smoking, underlying diseases, gravity, and the effects of muscular action, which produce hyperfunctional lines of aging.18
Depending on the underlying skeletal structure, involutional soft tissue changes associated with the aging process result in transformations of the face that appear progressively more pronounced, and thus obvious, with time.
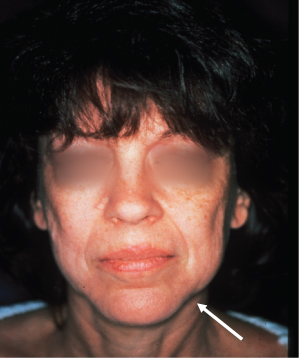
Recognizing structural and soft tissue defects and altered anatomy is an integral part of assessing whether a patient is a candidate for facial contouring procedures. Involutional facial changes include flattening of the midface, thinning and/or inversion of the vermillion border of the lips, development of deep cavitary depressions in the cheek, and formation of deep skin folds and rhytides.19 Specific to soft tissue, alterations due to aging also include increased prominence of the nasolabial folds, flattening of the soft tissue button of the chin, and formation of the prejowl sulcus20,21 (Figure 8.2).
Figure 8.2 Resorption of bone within the anterior mandibular groove, coupled with relaxation of the soft tissue causing progressive encroachment of the jowl, creates the prejowl sulcus (arrow) and contributes to the development of marionette lines. In these conditions, a prejowl implant is used to augment and help correct the deficiency and assist the rhytidectomy to achieve the desired straight mandibular line and prevent recurrence of the jowl.
(From Binder WJ. A comprehensive approach for aesthetic contouring of the midface in rhytidectomy. Facial Plast Surg Clin N Am 1993;1:231–55.)
One of the most elusive aspects of facial rejuvenation is the replacement of soft tissue volume in sufficient quantity that will remain for the life of the patient. The recent popularity of facial fat grafting has reemphasized tissue replacement as a key component of the rejuvenation process. Alloplastic augmentation techniques aim to address reductions in volume by softening sharp angles or depressions, reexpanding the underlying surface to reduce rhytides as well as enhance inadequate skeletal structure.22–24
Augmentation of the Midface
Rhytidectomy has become just one of many components of facial rejuvenation. Midfacial augmentation, face-lifts, and resurfacing techniques must all be considered when customizing a surgical plan for the patient. The pathophysiology of the aging process is a key factor in determining the correct surgical treatment. It is now well recognized that the aging process not only results in the descent of midfacial structures but also produces atrophy of the soft tissue in multiple facial planes. Although midfacial rejuvenation is often achieved through suspension techniques alone, the surgeon must also evaluate whether augmentation of the soft tissue and/or skeletal foundation is needed.
Alloplastic augmentation of the midface is an effective way to rejuvenate the whole face in appropriate candidates. Midfacial implants are readily reversible and can be combined with standard rhytidectomy procedures, although some patients may require only augmentation via implants for facial rejuvenation. The procedure is a straightforward, long-lasting, and relatively low-risk surgical option that consistently and predictably improves facial aesthetics while producing changes on more than one level. It replaces soft tissue volume that was lost, which consequently increases the anterolateral projection of the cheeks and cheekbones, thereby improving laxity and decreasing the depth of the nasolabial folds. The net effect is a softening of the sharp angles and depressions of the aged face, resulting in a more natural, “unoperated” look.
Midface augmentation can also enhance rhytidectomy in several ways. The skin and soft tissue can be draped over a broader, more convex midface region after implant placement. There is also minimal traction on the perioral tissues and lateral labial commissures if the implants are placed prior to the rhytidectomy, which can help to avoid an “overpulled” appearance. Many patients who present for revision rhytidectomy who additionally require volume restoration can also be improved by expanding the midface region while decreasing downward vertical traction forces on the lower eyelid.
Specific criteria are available for determining regions of structural and/or soft tissue deficits and their corresponding alloplastic solutions.25,26 In addition, other regions that contribute to the overall appearance of the midface must be considered during evaluation of the patient. In the periorbital region, the aging process results in the weakening of the orbital septum and herniation of the periorbital fat, causing infraorbital bulges.
The orbicularis oculi muscle becomes ptotic, especially in its most inferior aspect. The use of conventional blepharoplasty will tend to aggravate laxity of the lower canthal ligament, which can contribute to the formation of the “tear trough” deformity and malposition of the lower lid.27,28 These signs of aging are exaggerated in patients who have a negative vector to the infraorbital rim and malar bone or a retrusive maxilla.
The hollowness of the eyes found with advanced age is a result of subcutaneous tissue atrophy that has more damaging affects on the very thin infraorbital skin. Skeletal insufficiency and imbalances are usually caused primarily by the hypoplastic development and inherent bony imbalances of the facial skeleton that are exacerbated by the aging process. Midfacial descent involves ptosis of the infraorbital subcutaneous tissues, malar fat pad, the suborbicularis oculi fat (SOOF) and orbicularis muscle. The SOOF is the transition tissue between the orbital septum and the malar fat pad and is a thin layer of granular fat present under the lower orbicularis fibers. It is not connected with the periorbital fat, which remains separated from the SOOF by the orbital septum and its insertion onto the inferior orbital rim at the arcus marginalis.
As the cheek falls and the thicker tissues of the malar fat pad descend, it leaves the infraorbital region exposed to the covering of thin soft tissue. Thus, the tear trough and nasojugal groove areas become defined, the lower eyes appear hollow, and the infraorbital rim becomes more prominent. The mound of tissue on the superior border of the nasolabial fold becomes more defined. Although subcutaneous tissue loss occurs throughout the body, midfacial tissues that include the buccal fat pad, the malar fat pad, and the SOOF are more visibly affected than other areas of the face. As these tissues continue to lose volume and descend, the patterns of midfacial aging developing in the infraorbital and cheek regions become more apparent.
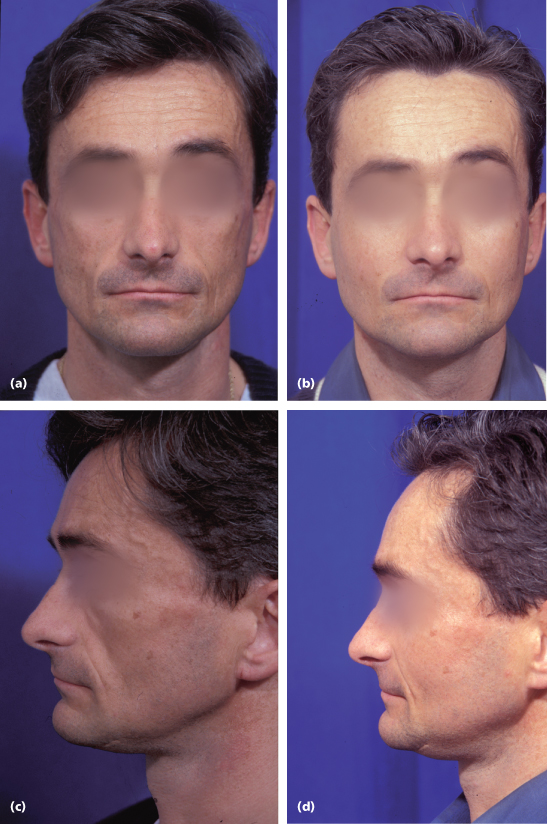
The majority of soft tissue deficiencies in the midface are found within the recess described as the “submalar triangle.”29 This inverted triangular area of midfacial depression is bordered superiorly by the prominence of the zygoma, medially by the nasolabial fold, and laterally by the body of the masseter muscle (Figure 8.3). The combination of significant soft tissue involutional changes associated with deficient underlying bone structure exaggerates the effects of the aging process. Individuals with thin skin lacking subcutaneous or deep supporting fat but have prominent cheekbones may also exhibit depressions in the cheek area. This type of pattern causes a gaunt appearance in an otherwise healthy person, although the severe form of this pattern can be seen in anorexia nervosa, starvation, or HIV-associated lipoatrophy. In combination with the primary disease process, protease inhibitors and other newer generation HIV therapies have a predilection for erosion of the midfacial fat and the buccal fat pad1,2 (Figure 8.4). These conditions of volume loss, in addition to the aging process, often preclude rhytidectomy or fillers alone to completely rejuvenate the face, but can be successfully and permanently treated with the use of computer-assisted custom-designed facial implants.30
Figure 8.3 The inverted submalar triangle is an area of midfacial depression bordered medially by the nasolabial fold, superiorly by the malar eminence, and laterally by the main body of the masseter muscle.
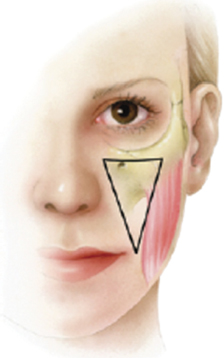
Figure 8.4 (a and c) Preoperative photographs of an HIV patient who has been treated with protease inhibitors for a prolonged period of time. Many patients eventually develop complete erosion of the midfacial fat and the buccal fat pad, leaving a particularly deep cavitary depression in the midface. (b and d) One year postsurgery, the condition was successfully treated with computer-assisted custom-designed midfacial implants.
A three-dimensional approach must be utilized for successful rejuvenation of the midface. The descent and volume los/>
Stay updated, free dental videos. Join our Telegram channel

VIDEdental - Online dental courses


