Chapter 8. Intracanal medication
D. Ørstavik
CHAPTER CONTENTS
Summary111
Introduction112
History112
Resistance of oral microbes to medicaments116
Concept of predictable disinfection in endodontics116
Induction of hard tissue formation119
Pain of endodontic origin119
Exudation and bleeding120
Root resorption120
Tissue toxicity and biological considerations121
Learning outcomes123
References123
SUMMARY
Endodontics may be considered the treatment or prevention of apical periodontitis, which translates into the elimination or control of root canal infection. Over the years, medicaments used for this purpose have changed from strong and toxic chemicals to more selective and effective, yet tissue tolerant agents. Antibiotics have found limited use in root canal treatment, but may be useful in the treatment of immature teeth. The bacterial flora of different types of root canal infections is variable, and so is their susceptibility to conventional medicaments; this has led to alternatives, or combinations of, medicaments that show promise in managing persistent and recurrent infections. A better understanding of factors influencing the local availability and potency of medicaments has helped facilitate improved means of application. Medicaments are also used with the intention of controlling pain and supporting the tissue healing process; however, they are always secondary to their main, antimicrobial function. While irrigation with sodium hypochlorite and dressing with calcium hydroxide remain standard treatment protocol, chlorhexidine and iodine compounds are emerging as alternatives or supplements to these classical medicaments.
INTRODUCTION
Endodontic success or failure is related to the absence or presence of signs and symptoms of apical periodontitis. 1 Root canal treatment can, therefore, be considered the prevention or cure of this disease. 2 Apical periodontitis includes periapical granuloma and radicular cyst as well as acute manifestations of inflammation. The aetiology of apical periodontitis is, primarily, a bacterial infection of the root canal system;3.4.5. and 6. consequently, the technical and pharmacological aspects of prevention and treatment are mainly aimed at controlling infection. Thus, preventive endodontics entails treatment of a tooth without previous signs of apical periodontitis by aseptic pulp extirpation. Curative endodontics is the chemomechanical elimination of infection in the root canal system of a tooth with signs of apical periodontitis. Both procedures are completed by the placement of a bacteria-tight filling to prevent new infection.
The use of intracanal medicaments is an adjunct to the prevention, and essential for, the treatment of apical periodontitis. The primary function is to prevent root canal infection where none is present, and/or to inactivate bacteria already infecting the root canal space. Intracanal medicaments would include any agent with intended pharmacological action introduced into the root canal space. Antibacterial and other active compounds currently used as irrigating solutions during instrumentation rightly belong in this category. Intracanal dressings more concisely describe medicaments left in the root canal space to exert their effects over a longer time period.
HISTORY
The role of microorganisms in pulpless teeth was recognized more than a century ago, 7 and strong, caustic antiseptics were popular as intracanal medicaments at the turn of the twentieth century. Formaldehyde-containing materials, e.g. formocresol, 8 and iodoform pastes9 belong to this category and remained popular for decades. Formulations with sulphonamides10 and later antibiotics were tried as intracanal medicaments; Grossman’s polyantibiotic paste11 and Ledermix®12 (Haupt Pharma GmbH, Wolfratshausen, Germany) are examples of these types of dressings.
The reduction of pain through pharmacological control of the inflammatory process has also been attempted in endodontics by the application of eugenol, 13 and later corticosteroids and other anti-inflammatory drugs, 14 as dressings. The focus on the possible adverse toxic effects of medicaments15,16 led to a more systematic selection from the list of disinfectants available for use. Phenol derivatives and iodine formulations gained popularity as medicaments in endodontics; sodium hypochlorite was confirmed as a suitable irrigant.
RATIONALE AND OVERVIEW OF APPLICATIONS
The primary function of endodontic medicaments is to provide antimicrobial activity. In a few instances, other, secondary functions are desirable (Table 8.1). The rationale for applying intracanal medicaments in various clinical situations has been reviewed. 19
| Primary function: antimicrobial activity |
| Antisepsis |
| Disinfection |
| Secondary functions |
| Hard tissue formation |
| Pain control |
| Exudation control |
| Resorption control |
Asepsis, antisepsis and disinfection
Asepsis is the assurance that no pathogenic microorganisms are present in the field of operation. It entails the use, not only of clean, but also of sterile or disinfected instruments and utensils, liquids, etc. In the course of treating teeth with no signs of root canal infection, maintaining asepsis is the primary means of preserving a bacteria-free canal. Antisepsis is the endeavour to prevent or arrest the growth of microorganisms on living tissue. In vital pulp extirpation, antiseptic measures are necessary to prevent infection in case there is a breach in the chain of asepsis. Irrigating solutions and interappointment dressings need to be antibacterial in action to prevent any microorganisms, which may contaminate the root canal system, from multiplying and establishing themselves.
Disinfection is the elimination of pathogenic microorganisms, usually by chemical or physical means. Disinfection by antiseptic agents is what is attempted in the treatment of infected teeth. Sterilization, on the other hand, implies the use of irradiation or heat to reach a state of complete freedom from live microbes and cannot be applied to root canal treatment. Disinfection entails mechanical removal of tissue and debris containing microbes, irrigation and dressing with antiseptic agents; also, surgical removal of an infected apex contributes to the antiseptic efforts of treatment. The presence of radiologically discernible apical periodontitis is a sign that the root canal system is infected. 6 This state of pre-existing infection also has a negative influence on prognosis. 20 In these cases, bacterial reduction, and effective disinfection, of the root canal system is a prerequisite for successful treatment.
Secondary functions of medicaments
Root canal treatment is sometimes associated with clinical features only indirectly related to infection of the root canal system. Pain during and after treatment may occur, and the associated tissue reactions include exudation, transudation, swelling and resorption. Each of these phenomena, either singularly or in conjunction with infection, has been a target for attempts at medication during, between and after treatment sessions.
Pain control
Pain is mostly associated with infection, and the primary means of pain control in endodontic treatment is infection control. Pharmacological agents that result in pain reduction through a decrease in the tissue responses to inflammation may have a role in further alleviating clinical pain from both infectious and aseptic pulpal-periodontal inflammation.
Control of exudation or bleeding
Persistent exudation in the root canal may occur, despite apparently successful technical treatment. Exudation reflects inflammation, however, and residual infection should be suspected. Therefore, treatment is aimed at dealing with potential infection as well as drying, or coagulating the exudating site.
Control of inflammatory root resorption
Trauma to the teeth may result in various forms of resorptive damage; inflammatory root resorption being the most aggressive and destructive. Inflammatory root resorption is normally associated with infection of the root canal combined with physical damage to the cementum; again, a primary function of treatment is to eliminate infection in the root canal system. Secondarily, medicaments may influence the resorption process itself.
Induction of hard tissue formation
It is often considered desirable to allow hard tissue to form, to continue apical root development, to close a wide foramen, or to create a mechanical barrier at a fracture line. Although the mechanism of action is largely unknown, dressings are available with claims of inducing hard tissue formation (see later).
MICROBIOLOGY OF ENDODONTIC INFECTIONS
Following pulpal necrosis, sooner or later, the entire root canal system will become infected. A long-standing infection will have bacteria not only in the main, but also in accessory canals, and for a distance into the dentinal tubules. 21 If apical periodontitis has progressed to the point where resorption of the cementum occurs, bacteria may be found throughout the length of the dentinal tubules. 22
The source of the infecting bacteria may be dental caries, salivary contamination through fractures, cracks or leaking fillings, or contamination of the pulp space during dental, including endodontic, treatment. The microbial flora will vary with the clinical condition, and endodontic infection may be one of three types: primary, secondary, or persistent infection. Infected canals typically contain 2–10 different cultivable species; the total number of microorganism ranging from 103 to 107. 23 The exact number of microorganisms is not known as they vary from tooth to tooth, and because of a lack of established quantitative methods of determination.
Primary root canal infection
With increasing time and depth, a primary root canal infection, i.e. one in existence prior to any intervention, the microbial flora changes from a predominantly facultative, Gram-positive flora to an almost completely anaerobic and mainly Gram-negative set of microorganisms. 24 Strains belonging to the genera Tannerella, Dialister, Fusobacterium, Prevotella, Porphyromonas, Peptostreptococcus, and Treponema are frequently found in teeth with apical periodontitis. 25 Other microorganisms not found by normal culturing techniques may be present. Facultative streptococci are also common. These same types of microorganisms are found in exacerbations or periapical abscesses. 26 In addition, infections involving other microorganisms, e.g. streptococci, Eubacterium, Veillonella27 and Actinomyces/Arachnia28 frequently occur. 29
In most infected teeth, several species are recovered from the root canals. 25 Many of the dominating species, e.g. Fusobacterium, Prevotella and Porphyromonas, may require the presence of some other synergistic species for their survival and propagation. It may be noteworthy in this context that these same species have not been found in dentinal tubules, which when infected harbour less fastidious microorganisms, such as lactobacilli and streptococci. 30
Secondary root canal infection
The microbial flora in retreatment cases, a secondary root canal infection, occurring during or after treatment, has been shown to differ significantly from microorganism in primary apical periodontitis. Typically, retreatment cases show enterococci, streptococci, and anaerobic cocci in high frequencies; 31,32 enteric rods and Candida are also relatively more frequent than in primary apical periodontitis.33.34.35. and 36.
Persistent root canal infection
A similar flora is found in persistent root canal infection. Here, a pre-existing infection has withstood the antimicrobial effects of treatment, and a selection of species survived, having adapted to the new environment in the root canal system. In these cases, dependent on the healing response after non-surgical root canal treatment, follow-up surgery may be necessary.
Extraradicular infection
Some organisms, notably Actinomyces and Propionibacterium species, but also others, are known to survive and propagate in soft tissues and also at the periapex as extraradicular infection. 37 These infections may be one of the causes of what has been termed ‘treatment-resistant cases’. Intracanal medicaments are of little use in extraradicular infections; surgery is indicated.
ANTIMICROBIAL AGENTS
Antibiotics
The successful use of various antibiotics, both systemically and topically, in other areas of medicine made them likely candidates for antibacterial action in the root canal system. There are three main concerns about the local use of antibiotics in the root canal:
• Sensitization. Topical application of an antibiotic increases the risk of the patient becoming allergic to it. 38 Life-threatening anaphylactic reactions may occur from the administration of antibiotics to sensitized individuals. Induced allergy to an antibiotic may limit the options for treatment of more severe infections, which would, otherwise, be curable with that particular drug.
• Development of bacterial drug resistance. The drug kinetics of antibiotics applied in the root canal is not well known. 39 Conditions may become favourable for the development of antibiotic-resistant microbial strains, causing an infection, which in turn is more difficult to treat. 40 Moreover, beyond the scope of treatment of the individual patient, the widespread use of antibiotics causes a general increase in pathogenic and indigenous microorganisms that are resistant to a variety of antibiotics. 41
• Limited spectrum. No one antibiotic is efficacious against all endodontic microorganisms. 42 Given that most endodontic infections are caused by a combination of species, the chance of one antibiotic achieving effective bacterial inhibition or elimination is small.
Sulpha preparations
Sulphathiazole as part of a dressing was advocated in the 1950s and 1960s. 10 While irrefutably antibacterial, variable results were shown in comparative clinical studies. 43,44 Moreover, although effective against many Gram-negative and Gram-positive microorganisms, sulpha drugs are ineffective against enterococci and Pseudomonas aeruginosa.
Penicillin
Grossman’s polyantibiotic paste contained penicillin as an important ingredient. Beta-lactamase produced by several microbial species found in the root canal makes them resistant to penicillin. This includes P. aeruginosa and several anaerobic Gram-negative rods.
Metronidazole
Metronidazole has good effect against several Gram-negative anaerobic microorganisms. 45 It has been suggested for use in irrigating solutions, 46 as an intracanal dressing47 and for parenteral applications in combination with other antibiotics, particularly penicillin. 37 It has limited activity against enterococci. 48
Tetracycline
Tetracycline shows affinity for hard tissues and may be retained on tooth surfaces. 49 It is used locally in periodontics with good clinical and bacteriological results, 50 and the derivative, doxycycline, forms the antibiotic ingredient in Ledermix. 51 However, its antimicrobial spectrum is quite narrow, and it may be ineffective against several oral and endodontic pathogens. The fact that resistance to tetracycline occurs through the formation of transferable R factors also suggests caution in its application.
MTAD (BioPure MTAD, Dentsply Tulsa Dental, Tulsa, OK, USA) is an irrigant containing a mixture of tetracycline, an acid and a detergent. The tetracycline component, doxycycline is retained in the dentine after application. Apart from the criticism that doxycyline is bacteriostatic and not bacteriocidal, another disadvantage is the potential iatrogenic staining of teeth when sodium hypochlorite reacts with MTAD with alternating usage. 52
Clindamycin
One study has reported on the use of clindamycin as an inter-appointment dressing, but only limited antibacterial efficacy could be demonstrated. 53 An experimental delivery device for clindamycin in the root canal has been reported, 54 and in vitro experiments suggest that clindamycin may penetrate deep into dentinal tubules. 55
Antibiotic combinations
While the traditional polyantibiotic pastes11 have been largely discontinued, a new combination of three or four antibiotic compounds has received interest, 56,57 Successful revascularization, with hard tissue formation in the pulp and complete root formation, has been observed in immature, permanent teeth with pulp necrosis and apical periodontitis after application of this mixture.58.59. and 60.
Disinfectants
While antibiotics work through biological interference with essential biochemical processes, disinfectants (Table 8.2) are a group of chemicals that act by direct toxicity to the microbes. Their action is thus quicker and more general, and they usually have a broader antibacterial spectrum than antibiotics. On the other hand, they may be more toxic to host tissues, and their action is generally more dose dependent.
| Halogens |
| Chlorine |
| Irrigating solution: sodium hypochlorite 0.5% in 1% sodium bicarbonate as Dakin’s solution; or 0.5–5.25% in aqueous solution. |
| Iodine |
|
Irrigating solution and short-term dressing: 2% iodine in 5% potassium iodide aqueous solution; iodophors.
Field disinfection: 5% iodine in tincture of alcohol.
|
| Chlorhexidine |
| Chlorhexidine gluconate |
|
Field disinfection and irrigating solution: 0.12–2.0% aqueous solution.
Irrigation and dressing: 1–5% gel.
|
| Calcium hydroxide |
| Dressing: aqueous, viscous or oily suspension/paste with varying amounts of salts added. Other antibacterial agents (iodine, chlorophenols, chlorhexidine) may be added. |
| Aldehydes |
| Formocresol |
| Dressing: 19% formaldehyde, 35% cresol, 46% water and glycerine. |
Aldehydes
Formaldehyde, paraformaldehyde and glutaraldehyde have been widely used in dentistry including endodontics. They are water-soluble, protein-denaturing agents and are among the most potent disinfectants. Aldehydes have applications in the disinfection of surfaces and medical equipment that cannot be sterilized, but they are quite toxic and allergenic, and some may be carcinogenic.
Formocresol is an aqueous solution containing cresol, formaldehyde, and glycerine, used for pulpotomy procedures in primary teeth, 61 but its toxic and mutagenic properties are of concern. It is no longer favoured and it has been suggested that its use should be discontinued. 62
Paraformaldehyde is the polymeric form of formaldehyde, best known for its inclusion in some root canal filling materials, e.g. N2 and Endométhasone. It slowly decomposes to release its monomer, formaldehyde; its toxic, allergenic, and genotoxic properties are as for formaldehyde.
Halogens
Halogens include chlorine and iodine, which are both used in various formulations in endodontics. They are potent oxidizing agents with rapid bactericidal effects. Chlorine is released from sodium hypochlorite and from chloramine. The latter releases active chlorine at a lower rate, and has been used for short-term dressing of the root canal. Sodium hypochlorite is currently the irrigating solution of choice. It is used clinically in concentrations from 0.5–5.25%. Both in vitro and in vivo bacteriological studies support its application. Necrotic tissue and debris are dissolved by sodium hypochlorite, 63 a property exploited in biomechanical cleansing of root canals. 64,65 Its toxicity is low; however, its bleaching properties can be a nuisance if spilled onto a patient’s clothes, its smell is objectionable to some patients, and it may cause severe symptoms if accidentally injected beyond the apex. 66
Iodine is used mainly as iodine potassium iodide and in iodophors, which are organic iodine-containing compounds that release iodine over time. Iodine is also a very potent antibacterial agent of low toxicity, but may stain clothing if spilled. As iodoform (triiodomethane), it was used in a paste formulation as a permanent root canal filling. 9 Iodine compounds are used as an irrigating solution and short-term dressing as 2% solution of iodine in 4% aqueous potassium iodide. It has also been added to gutta-percha points for root canal filling. 67,68 Some patients may be allergic to iodine compounds, and their use in these patients is contraindicated.
Phenol derivatives
Phenol itself is no longer used in endodontics because of its high toxicity, but the derivative paramonochlorophenol has been a very popular component of interappointment dressings. It has been used both in aqueous solution69 and in combination with camphor (as camphorated monochlorophenol); it was long recognized as the dressing of choice for infected teeth. Thymol, similarly, enjoyed widespread popularity, but is less antibacterial than the chlorophenol compounds.
Chlorhexidine
Chlorhexidine has been widely used in periodontology. 72 Its substantivity (persistence in the area of interest), its relatively broad spectrum of activity, and its low toxicity may make it well suited for irrigation and as dressings in endodontics. Results of in vitro studies pointed to the suitability of chlorhexidine in endodontics73.74.75.76. and 77. and some in vivo bacteriological data are emerging. 36,78 Effective concentrations are in the 0.2–2% range. Innovative attempts to utilize the disinfecting properties of chlorhexidine include its inclusion in gutta-percha points for root canal filling. 68
Calcium hydroxide
Calcium hydroxide has reached a unique position as a dressing in endodontics. 79 After its successful clinical application for a variety of indications, 18,80 multiple biological functions have been ascribed to calcium hydroxide. 81 Its primary function is probably antibacterial in most clinical situations, with the added benefits of cauterizing activity and high pH; also in the consistency of a paste, it physically restricts bacterial colonization of the canal space. Calcium hydroxide is applied as a thick, creamy suspension in sterile water, saline, and a variety of other, viscous or oily vehicles. 82,83
Calcium hydroxide with antimicrobial additives
Complete disinfection by calcium hydroxide cannot be expected in all cases. 84 Moreover, in root filled teeth the flora may contain microorganisms relatively resistant to its action. Numerous attempts have been made to mix calcium hydroxide with other aqueous and non-aqueous disinfectants. 85 Parachlorophenol, camphorated parachlorophenol, 82 metacresol, 86 and iodoform87 have all been added to calcium hydroxide suspensions. Combinations of calcium hydroxide with chlorhexidine have also been tested in vitro.88.89. and 90.
RESISTANCE OF ORAL MICROBES TO MEDICAMENTS
In some cases, bacteria persist and produce symptoms of inflammation despite apparently optimal cleansing and disinfection procedures. 91 They may either be inaccessible to the cleaning instruments or to the medicaments, or they may be resistant to the medicaments used. 57 Special interest has recently focussed on streptococci, enterococci and yeasts in persistent infections; these have been shown to be relatively resistant to calcium hydroxide84.90.92. and 93. and occur in high frequency in retreatment cases.27.33. and 94. Organic and inorganic debris in the root canal system will also affect and limit the antimicrobial activity of irrigating solutions and interappointment dressings. 95
CONCEPT OF PREDICTABLE DISINFECTION IN ENDODONTICS
Given the infectious nature of apical periodontitis, any clinical procedure should be based on the ability of each step to prevent contamination and to eliminate infection. A standard procedure should, furthermore, be based on a worst-case scenario, which would be the infected root canal with associated chronic apical periodontitis. Individual treatment steps have been assessed for their efficacy in eliminating bacteria from infected root canals23.91.96.97.98.99. and 100. (Fig. 8.1).
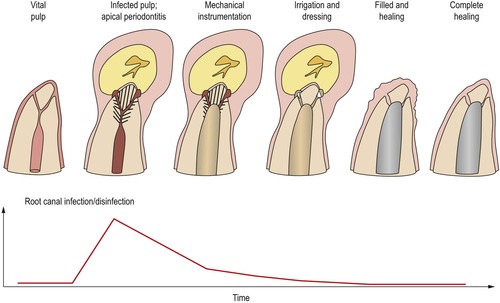 |
| Figure 8.1
Apical periodontitis develops when the root canal system becomes infected. Treatment entails the reduction of bacteria by mechanical instrumentation and antibacterial irrigation. The antibacterial dressing, if effective, eliminates infection. Total disinfection allows for complete healing of the tooth with apical periodontitis following root canal filling.
|
Mechanical instrumentation
Even in the absence of an antibacterial irrigating solution and subsequent dressing, there is still a dramatic decrease in bacterial numbers in a root canal from mechanical cleansing alone.23.101.102. and 103. However, in the majority of cases, bacteria, which are left in the canal, have the potential to multiply between appointments23 and after root canal filling. 94
Antibacterial effect of irrigation
The addition of sodium hypochlorite as an antibacterial irrigating solution increases the number of bacteria-free canals substantially. 96 The use of 5% rather than 0.5% sodium hypochlorite appears slightly more effective, and the reduction in the number of infected teeth has been shown to be even greater, when ethylenediaminetetraacetic acid (EDTA) was alternated with sodium hypochlorite, 96,97 and when ultrasonic instrumentation of the canal was performed. 100 Increasing the temperature of sodium hypochlorite also increases its efficacy. 104 Chlorhexidine (applied as a 2% gel) has been shown to be similarly or even more effective than sodium hypochlorite in reducing intracanal bacteria during instrumentation. 105
Effect of antibacterial dressing
The number of bacteria-free canals may be further increased when a dressing of calcium hydroxide is placed in fully instrumented canals between visits.84.102.106. and 107. Calcium hydroxide has been found to be more effective than camphorated monochlorophenol in comparative experiments, 84 but the added effect on the reduction from instrumentation and irrigation is variable. 79 Attempts at potentiating the antimicrobial effect of calcium hydroxide by the addition of other medicaments in the dressing has met with some, 108 but limited success. 109
Follow-up studies
Teeth treated as described above have been followed for periods of up to 7 years, a successful outcome (definite signs of healing of apical periodontitis) shown in more than 90% of the cases. 91 While success rates from different studies may be difficult to compare, it would appear that these clinical results are better than most, if not all, previous reports. 110
Adequate disinfection in single-visit endodontics
Single-visit (‘one-step’) endodontics implies shaping, cleaning and disinfection of the root canal in the course of one treatment session. This is followed by permanent root canal filling at the same appointment. Immediate root canal filling of teeth that are not infected is not controversial and, in principle, probably preferable to treatment in multiple appointments; if asepsis is maintained, there is no need for a disinfecting dressing between appointments and the risk of leakage. The debatable issue is whether the time duration and the type of medicament that can be applied during one appointment endodontics will provide predictable disinfection and, subsequently, healing of infected teeth with apical periodontitis. It is highly desirable to achieve effective disinfection quickly, and data from clinical studies have shown that treatment in one visit may be as effective, and as predictable, as procedures utilizing an interim dressing. 111,112 However, some clinical follow-up studies suggest better treatment outcome after dressing-based disinfection compared with single-visit procedures. 113,114 Human and animal histological studies have also reported the presence of residual bacteria and inflammation in teeth treated in one visit. 115,116
From controlled to predictable disinfection
Scientific data from clinical studies should be the basis for a rational, evidence-based approach to treatment of infected teeth. 117 Clinical experiments have documented controlled disinfection by advanced bacteriological techniques. When applied to clinical practice, adherence to the principles of mechanical instrumentation, irrigation with sodium hypochlorite and EDTA, and dressing with calcium hydroxide, would be expected to produce predictable disinfection (bacteria-free canals) in a very high percentage of cases (Fig. 8.2), and in turn, a very high rate of clinical and radiological evidence of healing of apical periodontitis. Indeed, large series of follow-up studies using this treatment regimen for infected teeth have borne out the high success rate. 118 A need for routine chairside bacteriological control of procedures is not implied in clinical practice.
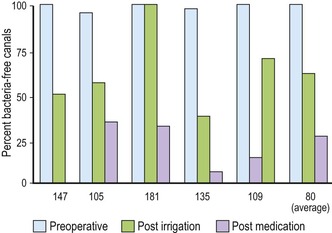 |
| Figure 8.2
Percentage reduction in canals positive for bacterial growth after instrumentation/irrigation and dressing of initially infected root canals. Numbers beneath bars refer to the articles cited.
|
Treatment of non-infected teeth
None of the steps advocated for the treatment of the infected tooth place in jeopardy the success of treatment of a tooth with an initially non-infected pulp. Vital pulp extirpation followed by instrumentation with sodium hypochlorite and a dressing of calcium hydroxide give a clean pulp wound with minimal or no inflammation (Fig. 8.3). Therefore, as a means of securing the absence of microbes in these cases, the same treatment principles should apply. One exception is when permanent root canal filling is possible at the first appointment; then there is no need for a period of canal dressing for the purpose of disinfection. Also, it may be questioned whether EDTA serves any purpose in the treatment of teeth with non-infected pulps.
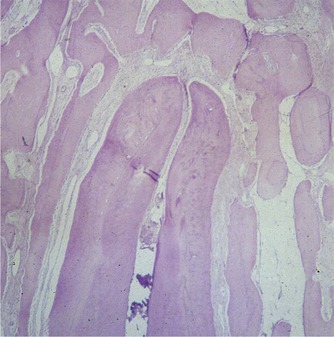 |
| Figure 8.3
Inflammation-free extirpation wound of a mature central incisor (monkey) after two weeks of dressing with calcium hydroxide.
|
Other principles of treatment may have the potential for equal or better efficacy and success rates. However, the extensive literature with clinical and bacteriological controls, and clinical and radiological follow-ups makes the above guidelines a standard of reference. Alternative methods and medicaments should be tested and compared similarly prior to receiving general clinical acceptance.
INDUCTION OF HARD TISSUE FORMATION
The process of creating a hard tissue barrier at an open apex or at a grossly over-instrumented apex is termed root end closure or apexification.119. and 120. When calcium hydroxide is used in long-term treatment of traumatized young permanent incisors and infection is controlled, a barrier of bone/cementum-like tissue at the apex is formed with a high degree of predictability120,121 (Fig. 8.4). This barrier allows mechanical compression of the root filling, and any toxic responses of the tissues to the filling materials are minimized by this intervening barrier. While it may not be essential that calcium hydroxide be used as a dressing for this purpose, its clinical application is the most extensively documented. Similar principles apply in the formation of a hard tissue barrier more coronally, e.g. at the line of a horizontal root fracture or at a pulpotomy or pulp-capping wound surface. 122 Mineral trioxide aggregate (MTA) is now the material of choice for these purposes; it is a filling or sealant material rather than a medicament, and thus falls outside the scope of this chapter123 but is covered elsewhere in this book (see Chs 9& 12).
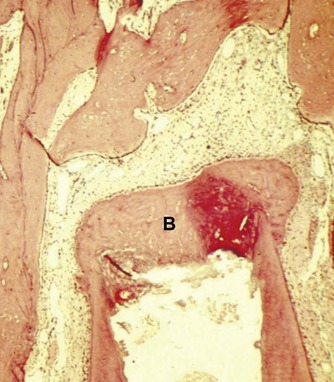 |
| Figure 8.4
Formation of a hard tissue barrier (B) at the apex of an immature central incisor (monkey). The pulp was extirpated and a dressing of calcium hydroxide placed for three weeks.
|
PAIN OF ENDODONTIC ORIGIN
Endodontic pain is mainly associated with both inflammation and infection.124.125.126.127.128. and 129. The inflammatory responses to the trauma of pulp extirpation and instrumentation may elicit pain of lesser magnitude and duration than pain following bacterial activity. 29,130 The rationale behind pain control by interappointment dressings is thus, primarily, to combat infection. This is reflected also in the finding that interappointment pain is significantly more frequent in infected, necrotic teeth than in vital cases. 131,132 There are strong psychological components to the clinical expression of pain of endodontic origin. 133,134 Clinical pain is further confounded by the concomitant presence of microbial and iatrogenic and other factors. The quantification of pain clinically is also very difficult to standardize for comparative purposes.
The incidence of interappointment or post-treatment pain seems to be very much dependent on the criteria defining pain.135.136. and 137. As an operating definition, the incidence of patients requiring an extra, non-scheduled visit following self-reported pain may have some merit. It would not include the discomfort sometimes associated with, the practical necessities of the treatment itself (local anaesthetic injection, rubber dam clamp placement, severance and laceration of the pulp). By such criteria, no significant advantage of one medicament over another has been reported.138.139.140. and 141.
Due to the lack of precise knowledge of the source of pain in individual cases, the introduction of medicaments in dressings to alleviate inter- and post-treatment pain has been by theoretical considerations, and by trial and error, rather than by clinical research. Most interest has focused on the use of corticosteroids in the interappointment dressing; particularly, the use of Ledermix containing triamcinolone, has been popular.12.51.142. and 143. It is doubtful whether initial concerns about the systemic effects of locally administered corticosteroids are justified; there is no indication that harmful side-effects are associated with its use in dentistry, and the doses applied are rather small compared with other medical indications. 144 However, while concerns for side-effects may have been exaggerated, the clinical benefits, if any, over calcium hydroxide medication remain questionable. 143 Non-steroidal anti-inflammatory drugs have also been tested clinically as intracanal dressings, 14 but any clinical advantages again remain obscure.
Stay updated, free dental videos. Join our Telegram channel

VIDEdental - Online dental courses


