Diseases of the Periodontium
The Healthy Periodontium
Gingiva
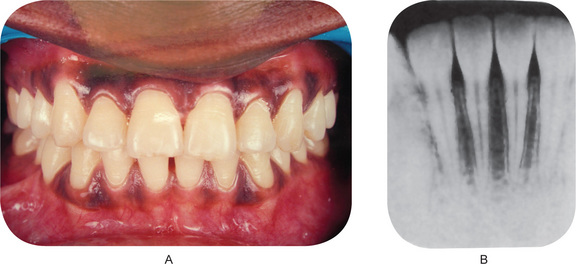
A healthy gingiva is a part of the masticatory mucosa covering the alveolar process and surrounds the cervical portion of the tooth by snugly fitting into each interproximal space between the teeth (Fig. 8-1A).
In the coronal direction, the gingiva terminates in the free gingival margin with a scalloped outline. In the apical direction it is separated from the loose, darker red alveolar mucosa, by a border termed as mucogingival junction or mucogingival line.
The epithelium of the free gingiva is microscopically differentiated as oral epithelium, facing the oral cavity, sulcular epithelium, which faces the tooth, and the junctional epithelium between the gingiva and the tooth.
Lamina Propria
1. Circular fibers, these fiber bundles are contained in the free gingiva and encircle the tooth in a cuff or ring-like fashion.
2. Dentogingival fibers are embedded in the cementum and project in a fan-like configuration into the free gingival tissue.
3. Dentoperiosteal fibers are also embedded in the cementum as dentogingival fibers, but run apically and terminate at the periosteum of the alveolar crest.
4. Alveologingival fibers extend from the crest of the alveolar bone to the lamina propria of the gingiva.
5. Transseptal fibers, these fibers run straight across the interdental septum and are seen embedded in the cementum of the adjacent teeth.
Periodontal Ligament
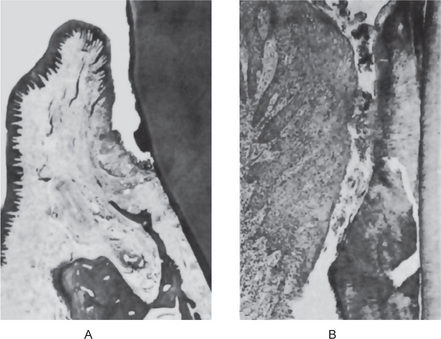
Periodontal ligament is made up of collagen fibers, oxytalan fibers, fibroblasts, amorphous ground substance, and interstitial tissue. Cells of the periodontal ligament include cementoblasts, osteoblasts, osteoclasts and epithelial remnants of Malassez (Figs. 8-2, 8-3). The shape of the periodontal ligament space is of an hourglass-like which is narrowest at the mid-root level. And width of the periodontal ligament ranges from 0.2 to 0.4mm.The mobility of the tooth is largely determined by the width, height and quality of the periodontal ligament.
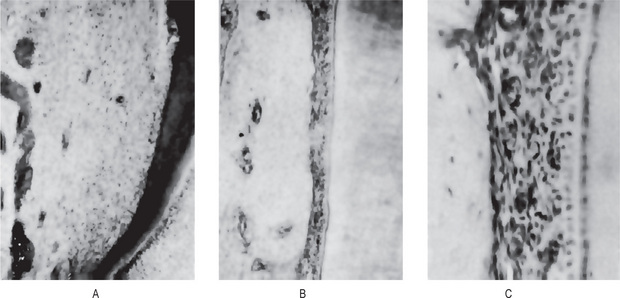
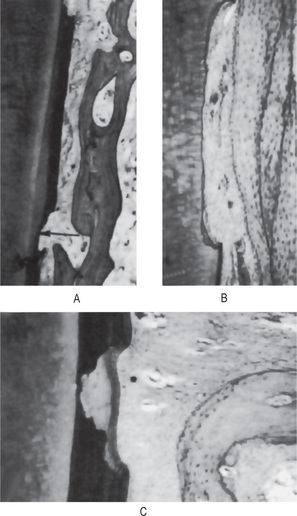
Figure 8-2 Normal immature periodontium.
The unoriented periodontal fibers of a developing tooth are illustrated in (A). The periodontal ligament of an unerupted tooth is shown under low magnification in (B) and high magnification in (C).
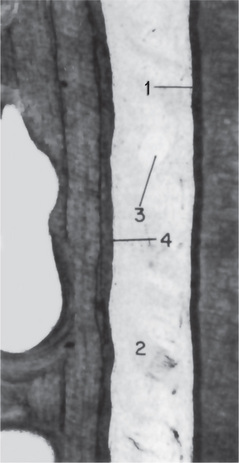
Figure 8-3 Normal mature periodontium.
Note the thin layer of cementum (1), the periodontal fibers (2), the large and empty capillaries (3), and alveolar bone (4).
The principal fibers of the periodontal ligament are collagenous bundles, with a wavy course in longitudinal sections. The principal fibers of the periodontal ligament are broadly grouped into:
1. Alveolar crest group. This group of fibres radiate from the crest of the alveolar process and attach to the cervical part of the cementum
2. Horizontal group. This group of fibers run at right angles to the long axis of the tooth from cementum to the alveolar bone.
3. Oblique group. These are the largest group of fibers extending from the cementum in the coronal direction obliquely to the bone.
4. Apical group. Apical fibers run from the cementum to the bone in an irregular manner at the apical portion of the tooth socket.
5. Interradicular group. This group of fibers which extend from the interradicular cementum of multi-rooted teeth to the crest of the interradicular bone.
Root Cementum
Cementum is a mineralized tissue covering the root surfaces of the tooth. It does not contain blood or lymph vessels or nerves. The different forms of cementum are the cellular, acellular, and afibrillar cementum. The functions of the cementum are to attach the fibers of the periodontal ligament to the root and to repair of the damaged root surface. Cementum of erupted and unerupted teeth undergoes resorption and repair (Fig.8-4). Cementum resorption may be idiopathic or may be caused by local conditions such as occlusal trauma, orthodontic tooth movement, pressure from cysts and tumours, teeth without antagonists, embedded teeth, replanted and transplanted teeth, and tooth with periapical and periodontal diseases. Systemic conditions such as hypothyroidism, calcium deficiency, and Paget’s disease may predispose or induce cemental resorption. Bimstein E and coworkers have examined cemental surface under light microscope and found that teeth from children with leukocyte adhesion deficiency, Down’s syndrome, and aggressive periodontitis have narrower cementum areas and concluded that these cemental anomalies may facilitate the establishment and progress of periodontitis in children. In another study by Bimstein and coworkers on histologic characteristics of root surfaces of primary teeth from children with prepubertal periodontitis revealed bacteria inside dentin tubules or covering cementum, a cuticle, or resorbed dentin; normal, wider than normal, or hypoplastic cementum; resorption lacunae with various depths; aplastic root resorption; alternate resorption and repair; and active repair.
Alveolar Bone
Alveolar process is the part of the maxilla and the mandible containing the sockets, which protects and supports the tooth. The main function is to distribute and resorb forces generated by mastication. Radiographs show that the alveolar bone has a definite cribriform plate with uniform trabeculae and it extends to a definite point between the teeth (Fig. 8-1B)
Deposits on Teeth
Pellicle
The pellicle has been thoroughly investigated by Meckle, by Leach and Saxton, and by Sonju and Rolla, utilizing electron microscopy, electron histochemistry, optical histochemistry, and chemistry. They found that the brownish stained, smooth, structureless deposits, in contradistinction to plaque, did not stain with basic fuchsin. The brown pigment forms due to the presence of tannins in the pellicle. The pellicle frequently penetrates into the enamel, especially on the proximal surfaces of the teeth. The histochemistry and the electron histochemistry indicated that pellicles are of salivary origin. The histochemistry of the pellicle was found to be practically identical with that of dried salivary films on glass. In addition, similar structures were formed in vitro by incubating enamel in saliva. The acquired pellicles were composed of mucoproteins or glycoproteins similar to those found in saliva and contained some lipid material. Primary amino acid groups and 1:2 glycol groups were also demonstrated by both electron histochemistry and chemical analysis. Bacterial enzymatic degradation of salivary glycoproteins did not take place, either because the material was rapidly deposited before bacterial enzyme action could occur or because the stereo-chemical structure of the glycoproteins enabled them to resist enzymatic degradation. A persistent extraneous calcification was always observed in pellicle from the lingual surfaces of lower anterior teeth, but was of such small magnitude that it could not be resolved by light microscopy.
Dental Stains
Pigmented deposits on the tooth surface are called dental stains or extrinsic stains. The oral cavity is subjected to many types of exogenous and endogenous substances that stain teeth. And since the oral flora in many cases contains chromogenic microorganisms, stained deposits are common on the teeth. The stains that are incorporated into tooth structure are known as intrinsic stains and are seen in porphyria, erythroblastosis fetalis and tetracycline therapy (Fig. 8-5A).
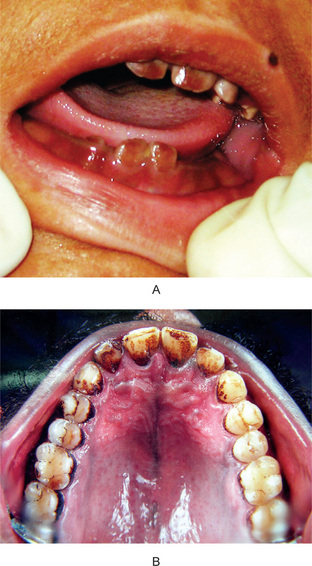
Figure 8-5 (A) Intrinsic stain in congenital erythropoietic porphyria (erythrodontia). (B) Tobacco stain. (Courtesy to Dr. Rashmi Santosh Kumar, Kamineni Institute of Dental Sciences, Narketpalli, Andhra Pradesh).
Stain from Smoking
As a result of the collection of tobacco tars and resins, a yellowish-brown to black deposit forms on the tooth surfaces of persons who smoke often (Fig. 8-5B). This stain varying from a light brown, powdery deposit in the person who smokes an occasional cigarette to a dense black tarry deposit in heavy smokers. The deposit is harmless to the teeth, although it should be removed because of its objectionable appearance and may act as a nidus for calculus or have a mild, irritating effect on the gingiva. If the dentin is exposed, as in older patients by attrition, the staining may be severe.
Calculus
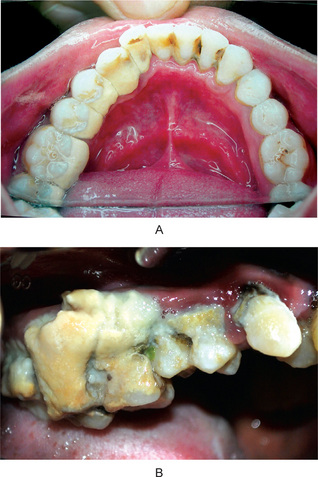
In some children and most adults, varying amounts of a hard, stone-like concretion form on the surfaces of teeth or prosthetic appliances (Figs. 8-6, 8-7, 8-8). These deposits are called ‘calculus’, ‘odontolithiasis’, or ‘tartar.’ Calculus is mineralised dental plaque.
Calculus is deposited as a soft, rather ‘greasy’ material, which gradually hardens by deposition of mineral salts in the organic interstices, until it becomes hard. It varies in color, from yellow to dark brown or black, depending upon the amount of stain present on or within the deposit.
Distribution of Calculus
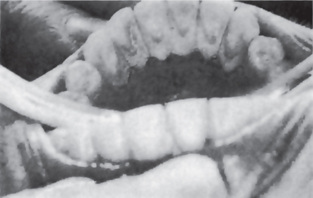
The greatest accumulations of calculus, both supragingival and subgingival, occur on those surfaces of the teeth that are closest to the orifices of the major salivary gland ducts. Thus the lingual surfaces of the mandibular anterior teeth opposite the submandibular and sublingual gland openings and the buccal surfaces of the maxillary molars opposite the parotid duct opening are the common sites of deposition of calculus (Fig. 8-9). It may be localized in its distribution or generalized over many tooth surfaces.
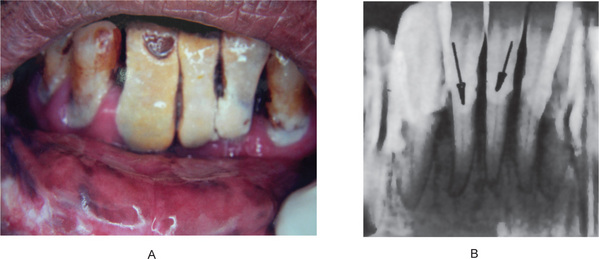
Figure 8-9 Calculus an one side of the lower arch. This patient was not using this side due to the presence of a painful tooth in the upper arch.
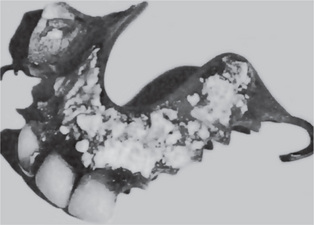
(A) Massive amount of calculus in a patient who had not had a prophylaxis for many years. (B) Radiographic appearance of teeth with a heavy calculus deposit.
Incidence of Calculus Deposition
However, Marshall-Day reported that in a study of persons 13–60 years old, the 19–22-year age group showed an increase in calculus deposition over the younger age group and that there was a further sharp increase in the 31–34-year age group. The incidence of calculus reached a peak of 91% of persons studied in the 56–59-year age group. Of the entire group, 71% of men and 62% of women manifested calculus formation. 26% exhibited supragingival calculus only, with no significant difference between men and women. Persons with subgingival calculus alone were comparatively few, the incidence being 7% for men and 8% for women. The greater proportion of subjects studied exhibited both types of calculus after 30 years of age. Third National Health and Nutrition Examination Survey revealed that 91.8% of the subjects had detectable calculus and 55.1% had subgingival calculus based on their survey involving 9689 adults in the United States.
Bacterial Colonization and Plaque Maturation
Numerous studies indicate that the early plaque is composed of a preponderance of coccal microbial forms. As the plaque ages, fusobacteria and filamentous organisms increase in number and, by the second or third week of plaque formation, about half of the organisms in the plaque become filamentous. Gram-positive cocci and rods make up the remainder of the plaque population. The population dynamics of plaque development indicates a progressive decline in aerobic organisms with a concomitant increase in anaerobic organisms.
Plaque Mineralization
The major mineral present in calculus is a carbonate apatite similar to that formed in bones and teeth. It is not known why some dental plaques mineralize and others do not. The bulk of the calculus mass consists of mineralized bacteria, and the earliest visible mineral deposition is usually associated with them. Certain microorganisms, such as Corynebacterium matruchotii and some strains of Streptococcus mutans, can be isolated from plaque, which forms an apatite intracellularly when cultured in a medium rich in calcium phosphate. In addition, dead microbial cells induce apatite formation when suspended in metastable calcium phosphate solutions. This finding suggested to Ennever and his coworkers that some component of the cell functions as a catalyst for apatite nucleation. They isolated such a catalyst from microbial cells and characterized it as a proteolipid, nonpolar protein-acidic phospholipid complex. Bacterial cells do not calcify if the proteolipid has been completely removed. The dental calculus matrix also contains proteolipid, which is essential for its remineralization in vitro after it has been decalcified. It is chemically similar to microbial proteolipid. Thus it appeared that proteolipid derived from plaque microbial membranes provided the catalyst for calculus formation. Isolated proteolipid and synthetically prepared analogs have been used to determine the mechanisms of calcification. An essential feature of the mechanism is the initial calcium binding by acidic phospholipids of the complex. The binding is followed by dehydration, which forms a microenvironment in which apatite nuclei are stabilized long enough for crystal growth.
Importance of Calculus
Calculus is always covered with the unmineralized plaque. This unmineralized plaque is the chief irritant and the underlying calculus acts as a significant contributing factor. Calculus is uniformly associated with periodontal diseases. Since it is adherent to the tooth surface, it moves with the tooth during its functions. Subsequently, injuries can occur to the adjacent or overlying gingival tissues that do not move in unison with the teeth. Also, when pressure is placed on the gingiva during mastication, the underlying calculus could irritate the gingival tissues. Thus calculus causes an inflammatory reaction in the gingiva with its overlying mat of microorganisms (Fig. 8-10). Occasionally, supragingival calculus collect in prodigious amounts with little more pathosis present than a superficial inflammation; possibly due to high tissue resistance. Removal of calculus results in rapid clinical improvement.
Classification of Periodontal Disease
Many classifications of periodontal disease have been proposed. The classification presented here (Table 8-1) is the most accepted and was presented and discussed at the International Workshop for the classification of the periodontal diseases organized by the American Academy of Periodontology in 1999.
Table 8-1:
Classification of periodontal disease
Gingival diseases
Plaque induced gingival disease
Nonplaque induced gingival lesions
Chronic periodontitis
Localized
Generalized
Aggressive periodontitis
Localized
Generalized
Periodontitis as a manifestation of systemic disease
Necrotizing periodontal disease
Necrotizing ulcerative gingivitis (NUG)
Necrotizing ulcerative periodontitis (NUP)
Abscesses of periodontium
Gingival abscess
Periodontal abscess
Pericoronal abscess
Periodontitis associated with endodontic lesions
Endodotic – periodontal lesion
Periodontal – endodontic lesion
Combined lesion
Developmental or acquired deformities and conditions
Localized tooth related factors that predispose to plaque induced gingival diseases or periodontitis
Mucogingival deformities and conditions around teeth
Mucogingival deformities and conditions on edentulous ridges
Occlusal trauma
Gingival Diseases
Plaque induced gingival disease is the most common form of gingival disease. This may occur on a periodontium with no attachment loss or on a periodontium with a previous attachment loss that is stable and not progressing. Plaque induced gingival disease is the result of the interaction of plaque bacteria and defense cells of the host.
Tooth Malposition
Teeth which have erupted or which have been moved out of physiologic occlusion, where they are repeatedly subjected to abnormal forces during mastication, are apparently very susceptible to the development of periodontal disease. For example, a lower incisor may be ‘bucked’ out of alignment in the second or third decade of life and suddenly, in its new position, receive much of the occlusal stress of one or two upper anterior teeth. Calculus may be deposited on the lingual surface of such a tooth; the bacteria present attack the tissue around this tooth. As a result the gingival tissues may become inflamed and may recede. Teeth in labial positions have less osseous coverage over their radicular surface and hence are more susceptible to trauma from toothbrushing and other local irritations. Abnormally high frenal attachments also contribute to gingival recession.
Chemical or Drug Application
Many drugs are potentially capable of inducing gingivitis, particularly an acute case of gingivitis, owing to a direct local or systemic irritating action. For example, phenol, silver nitrate, volatile oils, or aspirin, if applied to the gingiva, will provoke an inflammatory reaction. Others, such as dilantin sodium, produce gingival changes when administered systemically. These have been discussed specifically in the Chapter 12 on Physical and Chemical Injuries of the Oral Cavity.
Nutritional Disturbances
Nutritional imbalance is frequently manifested in changes in the gingiva and deeper underlying periodontium. The effects of nutritional deficiencies on these structures as well as on the oral cavity as a whole have been considered in detail in Chapter 15 on Oral Aspects of Metabolic Disease. It is sufficient to point out that adequate intake, absorption and utilization of the various vitamins, minerals and other foodstuffs are essential to the maintenance of a normal periodontium.
Pregnancy
Many investigators have reported that the gingiva undergoes certain changes during pregnancy which have been termed ‘pregnancy gingivitis’ (Table 8-2). Among studies of relatively large numbers of pregnant patients, the following may be cited as representative.
Table 8-2:
Incidence of pregnancy gingivitis
| Looby (1946): 475 women | (In percentage) |
| Slight gingivitis | 40 |
| Hypertrophic gingivitis | 10 |
| Pregnancy tumor | 2 |
| Ziskin and Nesse (1946): 416 women | (In percentage) |
| Pregnancy gingivitis | 37.9 |
| Hypertrophic gingivitis | 7.0 |
| Raspberry-red gingiva | 40.0 |
| Combination | 1.8 |
| Maier and Orban (1949): 530 women | (In percentage) |
| No pathosis | 44.6 |
| Mild inflammation | 35.9 |
| Moderate inflammation | 17.5 |
| Severe inflammation | 1.5 |
| Pregnancy tumor | 0.5 |
Other Endocrine Dysfunctions
Gingivitis is reported to occur with some frequency in puberty as the so-called puberty gingivitis. The gingiva appears hyperemic and edematous. The fact that many adolescents are chronic mouth-breathers as a result of lymphoid hyperplasia of the tonsils and adenoids has suggested that the endocrine basis is relatively unimportant, while the local irritant (drying of the mucosa because of the mouth breathing) being the actual cause of the condition. Gingivitis associated with menstruation has been reported by many. In addition, a nonspecific gingivitis with gingival bleeding, vicarious menstruation may occur sometimes. This phenomenon is rare.
Stay updated, free dental videos. Join our Telegram channel

VIDEdental - Online dental courses


 . The Healthy Periodontium
. The Healthy Periodontium . Classification of Periodontal Disease
. Classification of Periodontal Disease . Gingival Diseases
. Gingival Diseases . Enlargement Associated with Systemic Factors
. Enlargement Associated with Systemic Factors . Periodontitis
. Periodontitis . Peri-implantitis
. Peri-implantitis