Techniques for grafting and implant placement for the extraction site
![]() Additional illustrations can be found on the companion website at www.blockdentalimplantsurgery.com
Additional illustrations can be found on the companion website at www.blockdentalimplantsurgery.com
The normal sequence of events in socket healing takes place over approximately 40 days, beginning with clot formation and culminating in a bone-filled socket with a connective and epithelial tissue covering.1,2 Ideally, an extraction site heals with bone formation that completely preserves and also recreates the original dimensions of the bone. Unfortunately, bone resorption is common after tooth extraction; therefore, intervention is necessary with a method that provides ideal bone for implant placement and reconstruction of the patient with an esthetic and functional restoration.
Bone resorption usually is greater in the horizontal plane than in the vertical plane.3,4 Horizontal bone loss may be enhanced by thin facial cortical bone over the roots or bone loss from extension of local infection, such as caries or periodontal disease. Ideal placement of a dental implant centers the implant over the crest in a line connecting the fossae of the adjacent posterior teeth or, for anterior teeth, palatal to the emergence profile of the planned restoration. Unless the horizontal bone dimension is reconstructed or preserved after tooth extraction, implant placement is compromised, and in the esthetic zone, flattening of the ridge will occur, which results in a compromised restoration appearance.
Grafting material
1. Space should be maintained. Bone can repopulate the graft and thus recreate bone volume similar to pre-extraction size.
2. The bone formed should be dense enough to allow stable placement of the implant. The material placed should have osteoconductive features to enhance bone formation.
3. The material should resorb within a selected period of time with replacement by bone that is normal to the site.
4. The resorption rate of the material over time should be taken into consideration to plan the sequencing of therapies such as implant placement, additional contour grafting, and pontic and site development.
5. The material should be relatively inexpensive and readily available and should not transfer pathologic conditions.
Bovine or equine sintered xenograft
Bovine or equine derived bone is a xenograft. It is a carbonate-containing apatite with crystalline architecture and calcium–phosphate ratio similar to that of natural bone mineral in humans.5 With time, the sintered xenograft, mineralized bone graft material becomes integrated with bone. It may be slowly replaced by newly formed bone, but because the sintering process increases the crystallinity of the bone particles, it may not clinically resorb and will often be present years after placement.6–9 When sintered xenograft material is used to graft an extraction site, 6 to 9 months may need to be allowed before placement of the implant, especially if the clinician plans to provisionalize the implant immediately. The relatively inert nature of this material delays revascularization and subsequent bone formation compared with more natural materials such as autogenous bone.
Mineralized bone allograft
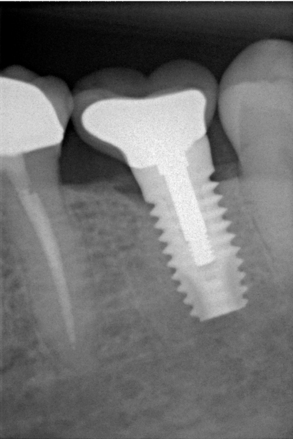
When placed in an extraction site, mineralized bone graft material is still present at 4 months.10 However, the bone forming around the mineralized bone particles usually is sufficiently mineralized to allow immediate provisionalization, with adequate primary stability after placement of the implant in the extraction site grafted with a mineralized allograft.
One goal of grafting of the extraction site is retention and preservation of the original ridge form and maintenance of the crestal bone after the implants have been restored. In one study in which no membrane was used at the time of extraction site grafting, the grafted sites felt “bone hard” at 4 months and appeared to be filled with bone.10 The average mesial crestal bone level was −0.66 ± 0.67 mm (range, 0 to −1.27 mm) at implant placement and 0.51 ± 0.41 mm (range, 0 to −1.91 mm) at final restoration. The average distal crestal bone level was −0.48 ± 0.68 mm (range, 0.64 to −1.91 mm) at implant placement and 0.48 ± 0.53 mm (range, 0 to 1.27 mm) at final restoration. A measurement of 1.27 mm from the top of the shoulder of the implants correlated to the level of the first thread of the implant.10 Bone heights were maintained with mineralized bone graft material.
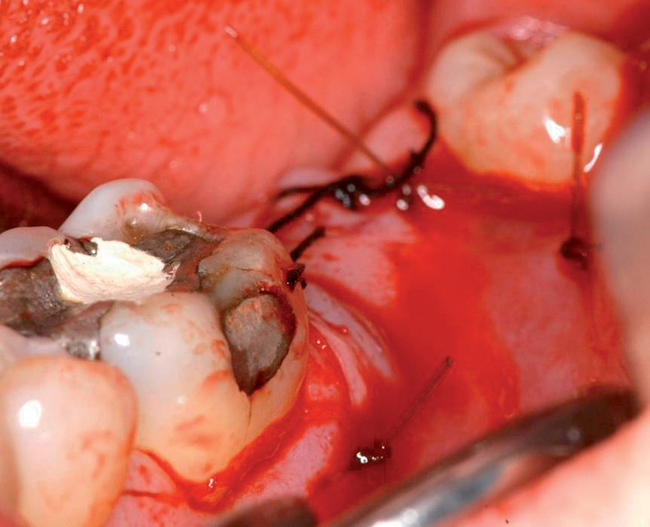
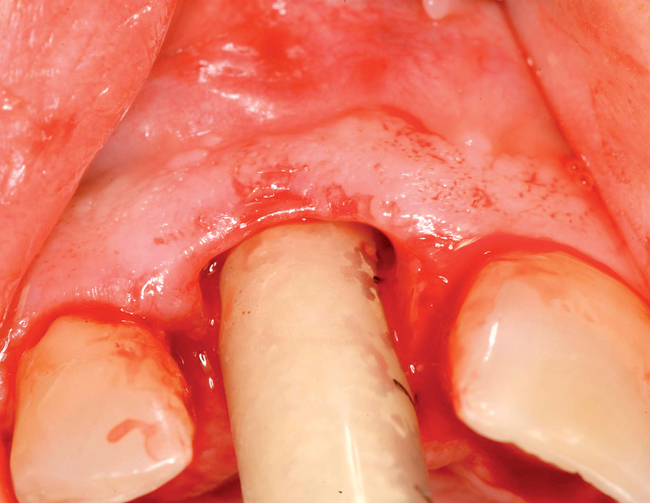
The current technique for premolars, canines, incisors, and the maxillary palatal root sites advocates the additional use of a fast-resorbing material to retain the graft and promote epithelialization over the graft. The graft can be covered with a fast-resorbing hemostatic collagen material that resorbs in less than 7 days.10 In mandible molar sites and for coverage of the buccal root sites for maxillary molars, coverage with advancement of the gingiva is recommended with the periosteum used to retain the graft in the extraction socket.
Autogenous bone
Clinicians believe that the ideal bone replacement graft material has always been autogenous bone.11–14 Very few clinicians use a separate harvest site to obtain autogenous bone to graft extraction sites. The past use of bone harvested from the symphysis, ramus, or maxillary tuberosity is not a common, current procedure. Bone removed during alveoloplasty can be used as a graft. Bone can be scraped from adjacent sites, collected in a sieve after the bone has been shaved with a bur and collected with rongeur forceps from adjacent sites or the alveolar ridge.15
Past animal studies by Block and Kent16 showed that the addition of autogenous bone to HA particles results in more bone formation within the graft compared with HA used alone. This dog study can be extended to sintered xenograft, which because of the sintering process has similar chemistry to synthetic HA. Hallman et al.17 showed in sinus augmentation the advantage of adding autogenous bone to bovine xenograft. Hellem18 showed similar advantages to adding autogenous bone to bovine xenograft for ridge augmentation. Autogenous bone can add bone within a graft of xenograft particles, which is a perceived advantage when implants are planned to be placed within the graft.
The decision to harvest autogenous bone usually is made before the tooth is extracted. The harvesting of autogenous bone is reserved for patients who need autogenous bone for extended grafts or to promote bone formation when using xenografts. Incision designs should consider the need for subperiosteal tunneling or separate incisions to allow harvesting of bone. When multiple teeth are extracted, alveoloplasty can be performed and the particulated bone placed within the extraction sites. An alternative to using alveoloplasty bone is to use a subperiosteal tunnel and one of the available bone-scraping devices to collect bone from the external oblique ridge. Another alternative is to collect bone into a sieve placed in a suction line. Bone particles can be collected from implant preparation drills or by the use of a round bur in the chin or body or ramus region.5
Clinical studies and case reports indicate that comparable end results may be obtained with appropriately used nonautogenous grafting materials.15,19–21
Anterior maxillary teeth
• Level of bone on adjacent tooth
• Presence or absence of root prominence
• Proportions of replacement tooth with regard to adjacent teeth
• Levels of bone around the tooth to be extracted, including apical bone, labial bone concavities, loss of labial or palatal cortical bone, and the presence of apical bone lucencies secondary to previous surgery. It is critical to have an accurate understanding of the presence, thickness, or lack of facial bone before removing the tooth. These preoperative diagnostic items, which include cone-beam scanning, will determine the flow of treatment to achieve an optimal result.
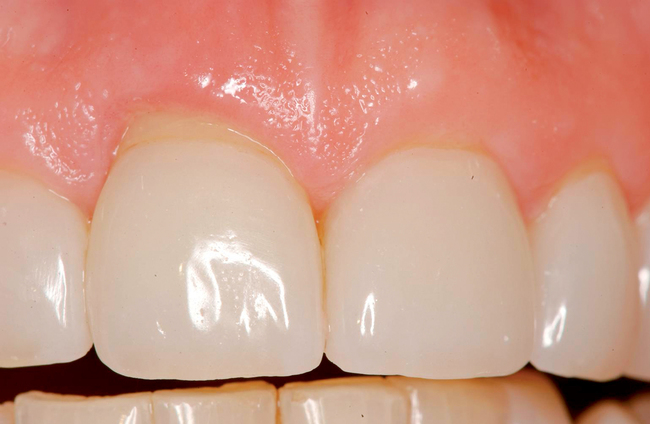
Gingival margin position
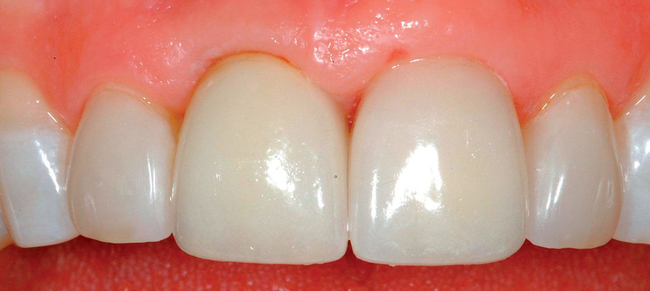
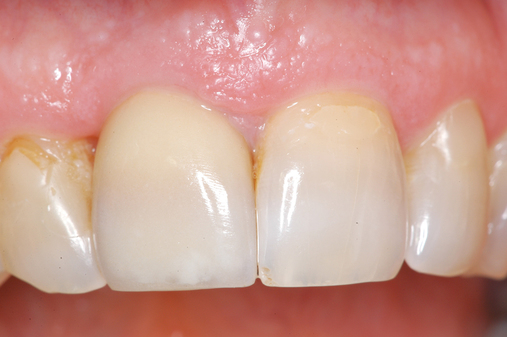
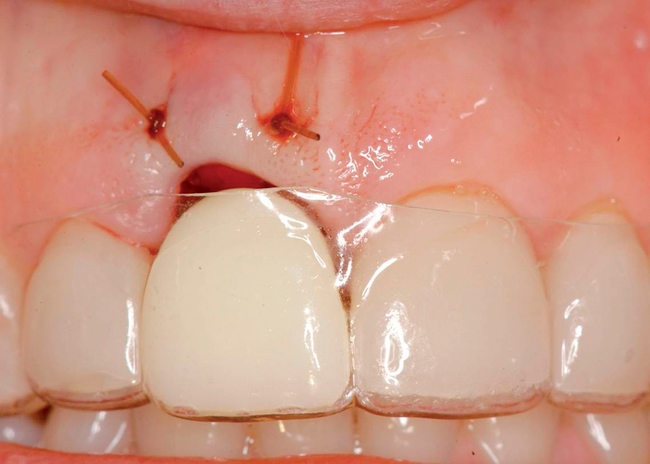
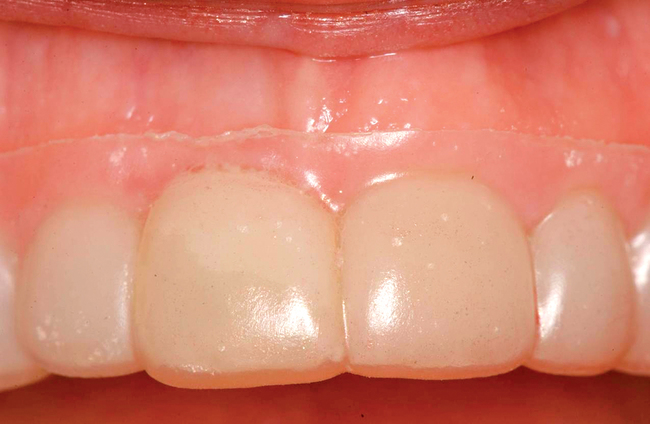
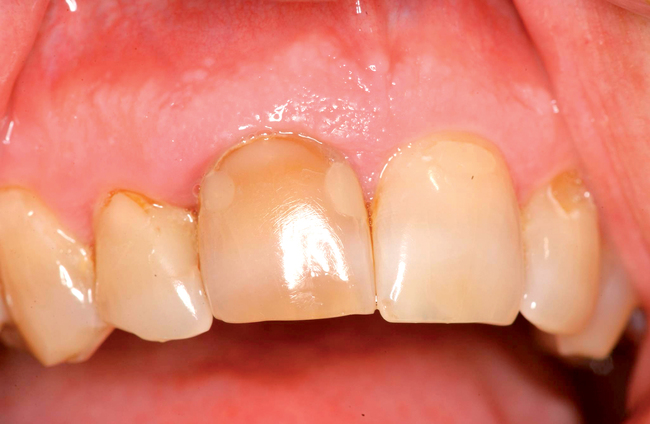
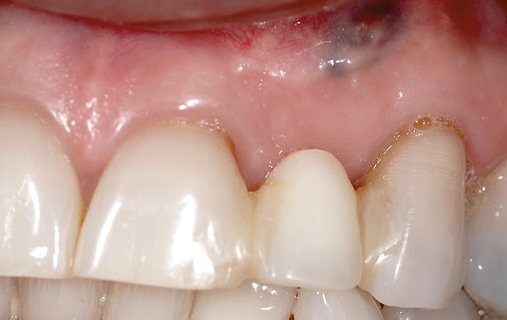
The level of the gingival margin over the implant’s final restoration is influenced by the gingival level before tooth extraction. If the gingival facial margin is ideal, the final implant restoration has an excellent prognosis, especially in a patient with thick gingiva. However, if the facial gingival margin is apically positioned and is not ideal because of crestal bone loss secondary to periodontal disease, attrition, or decay, or restorations invading the zone of biologic width, the final restorative facial gingival margin levels have a guarded prognosis unless interventional treatment is performed to correct or mask the gingival margin levels before tooth extraction (Figure 7-1).
Level of bone on adjacent tooth
Clinical evaluations by Tarnow et al.22 and Ryser et al.23 confirm that the most important factor that predicts the presence of papilla between a tooth and an implant is the distance from the contact point of the final restoration to the level of bone on the adjacent tooth. The distance from the contact point to the level of bone on the implant itself is less discriminating. If the bone level on the adjacent tooth is at the cementoenamel junction (CEJ), the papilla will be adequate as long as the proportions of the final restoration are reasonable.
Local deficiency of bone in the implant site
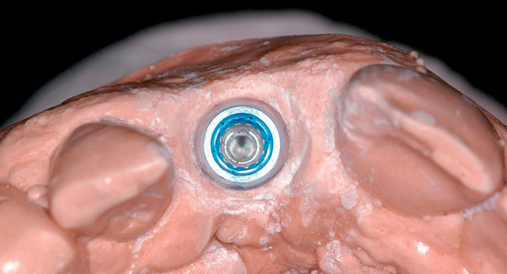
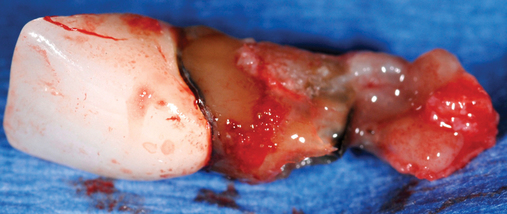
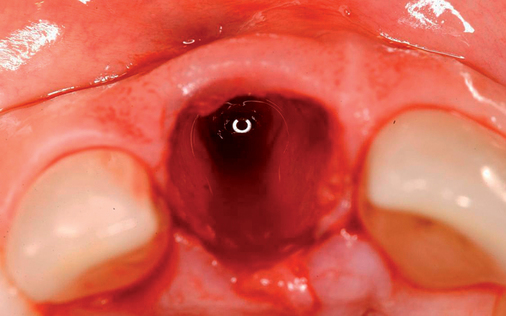
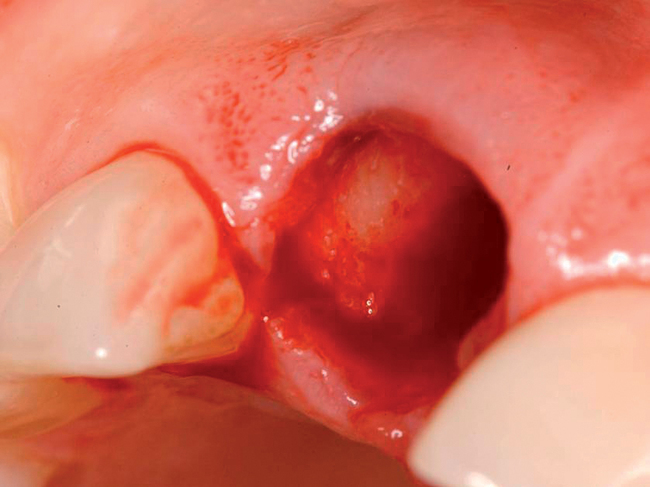
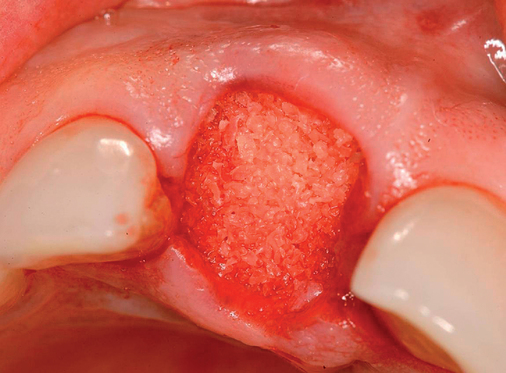
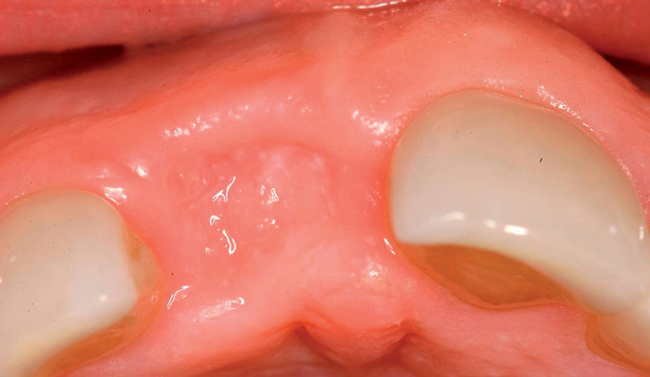
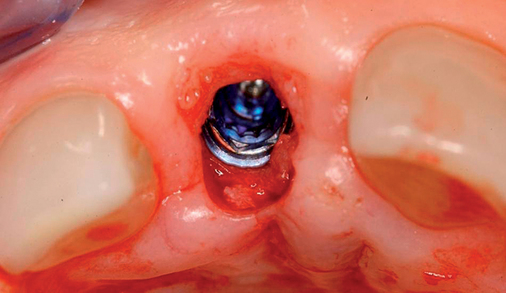
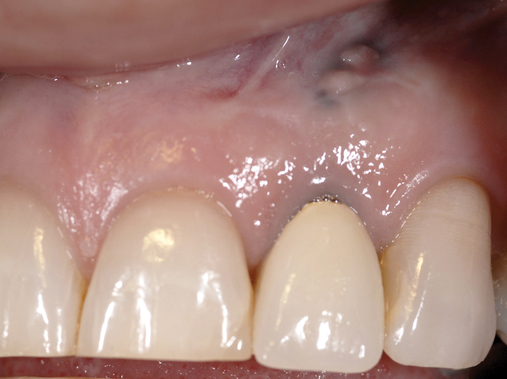
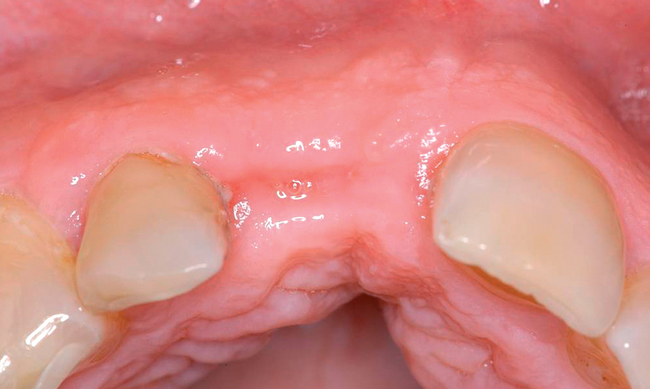
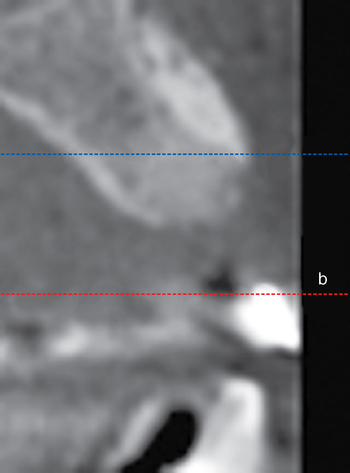
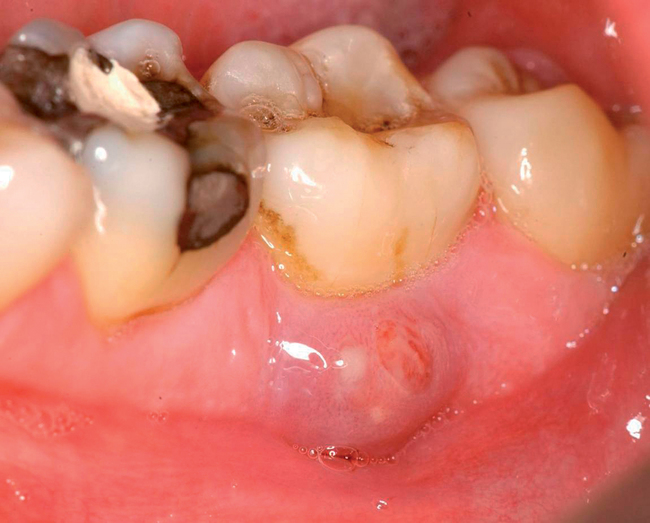
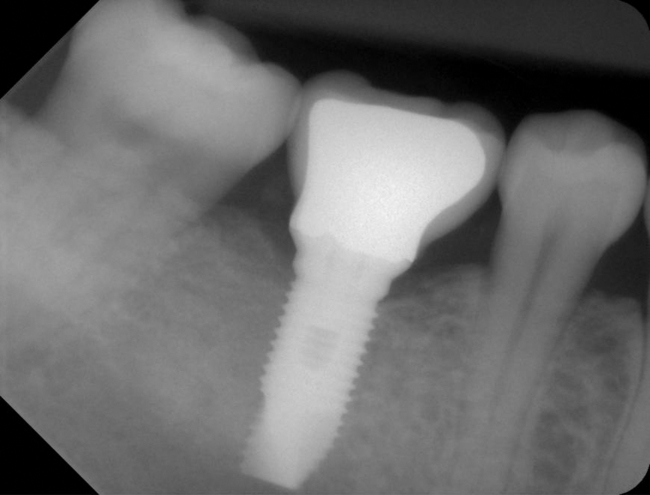
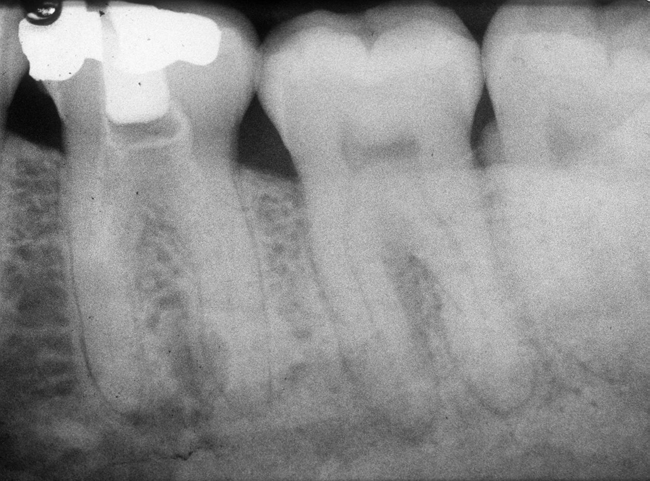
Cone-beam scanning is essential to determine the amount of bone available without a surgical surprise. Apical procedures may result in concavities, which have a direct effect on implant positioning and stability. If an apical bone concavity or labial bone loss is expected, grafting can be performed at the time of the extraction to augment the site before the implant is placed (Figures 7-2 and 7-3). Careful evaluation of these sites is critical so that the necessary surgical procedures can be performed and the desired result achieved. Imaging may be necessary with a tooth in the planned position to assess bone.
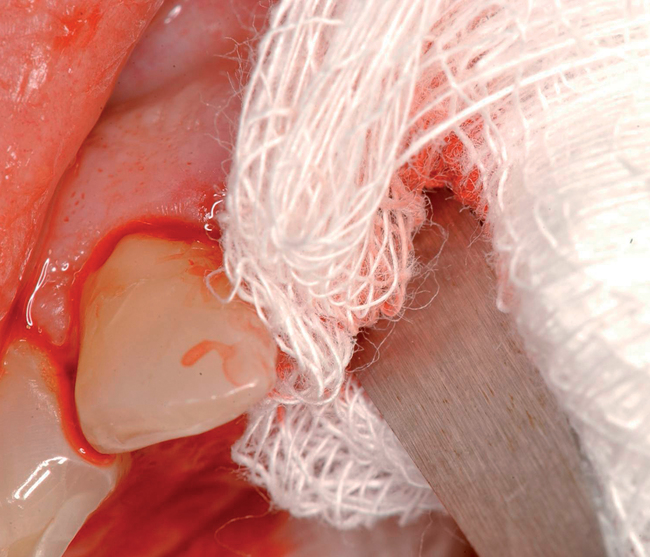
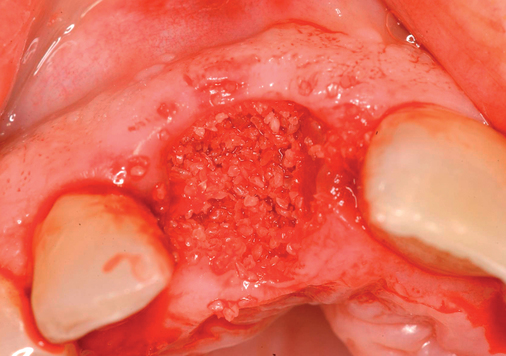
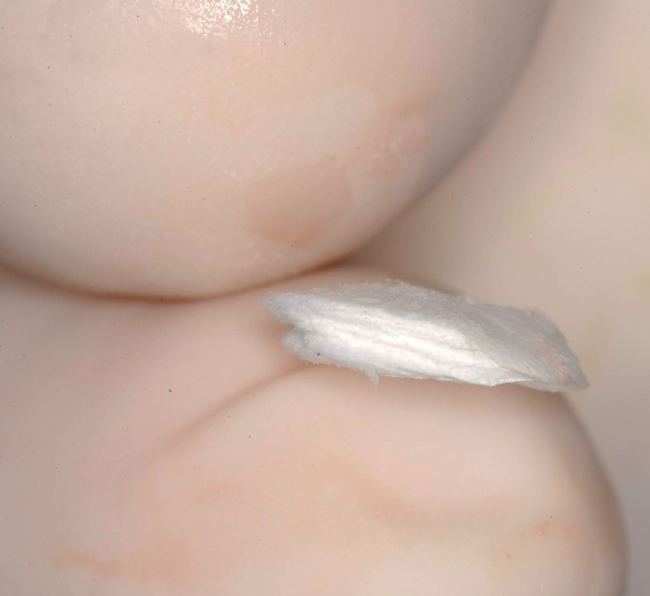
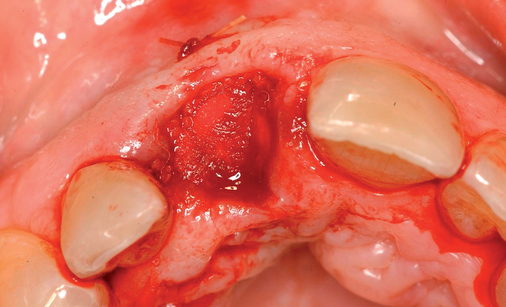
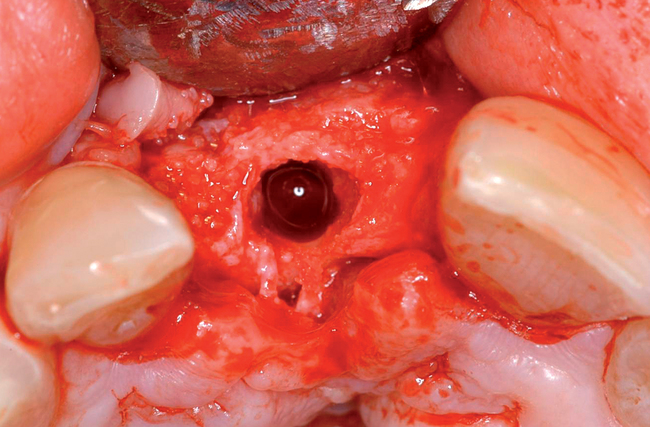
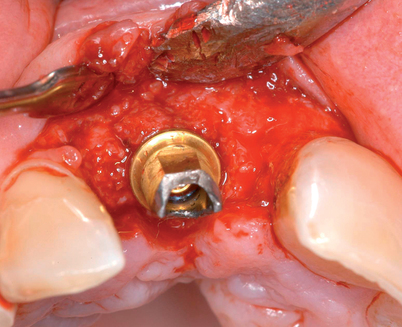
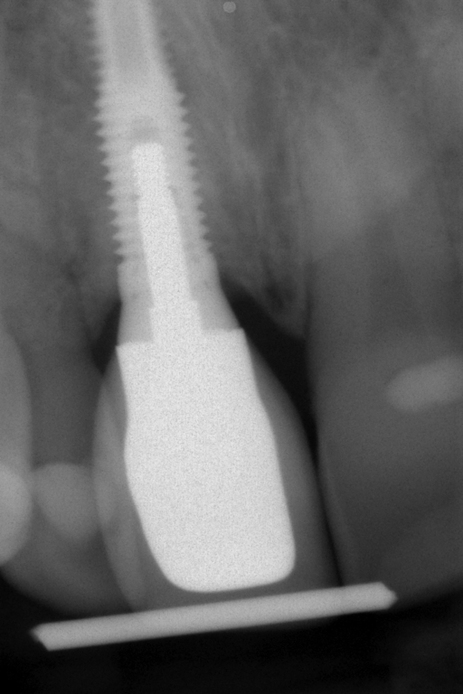
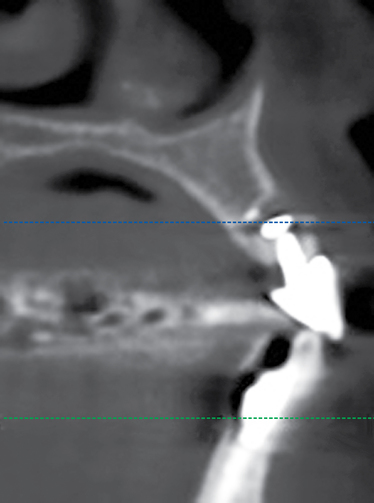
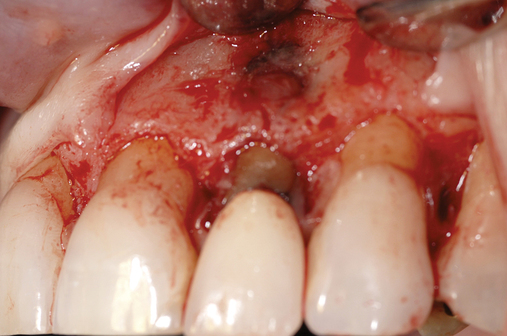
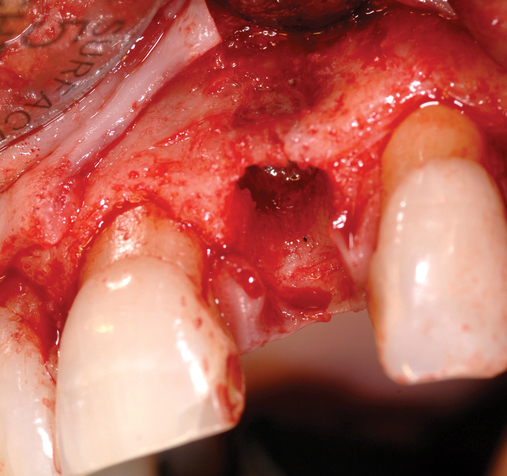
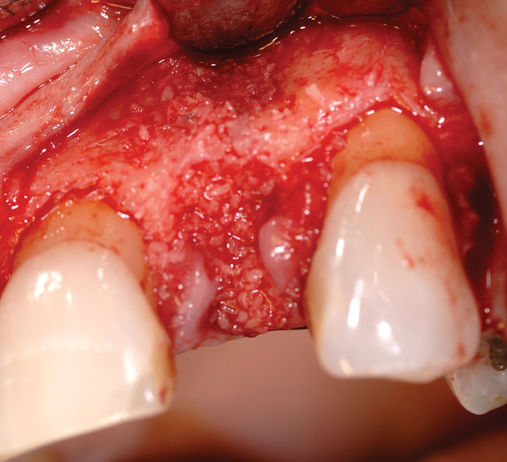
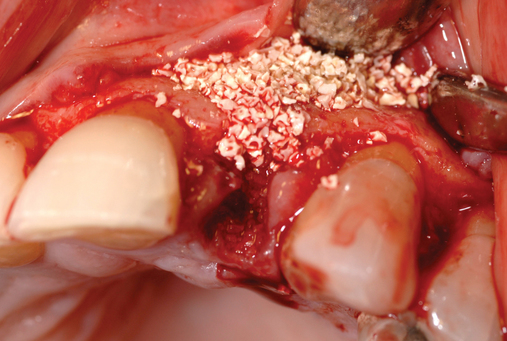
Labial defects
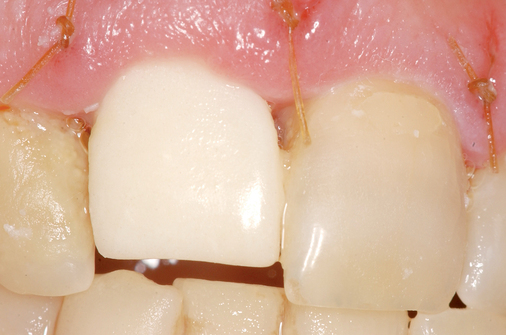
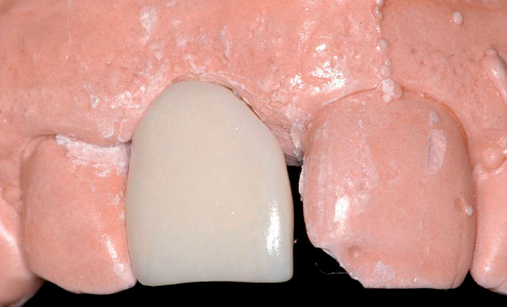
With more than 3 mm but less than 6 mm of labial bone loss, the implant usually can be placed in the appropriate position, but a bone defect may exist, with exposure of the implant threads. The clinician has two choices with labial bone loss after extraction of the tooth: (1) the site can be grafted with mineralized allograft bone with an overlay of sintered xenograft, with the patient returning 16 weeks later for implant placement (see Figure 7-3), or (2) the implant can be placed and the labial defect grafted with or without placement of a membrane to maintain the graft’s position. As the labial bone loss increases, the chance to obtain an ideal, perfect long-lasting gingival margin position diminishes. With moderate labial bone loss (3–6 mm), immediate provisionalization of the implant should be performed only if the radiofrequency index is greater than 60.24 In general, the more bone loss and less healthy gingiva, the less predictable is the resolution of the gingival compromise if an aggressive implant placement and graft scenario is pursued.
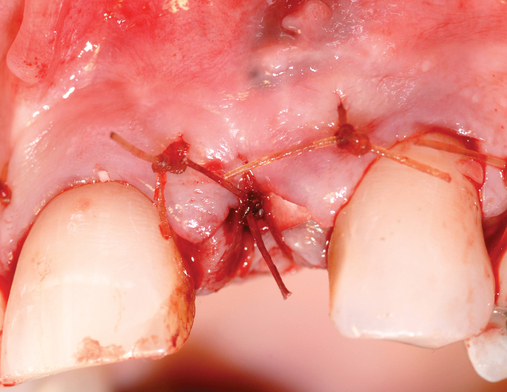
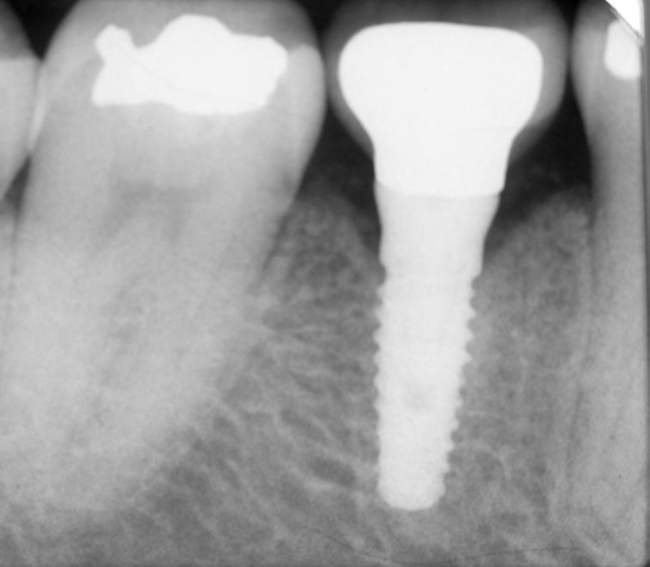
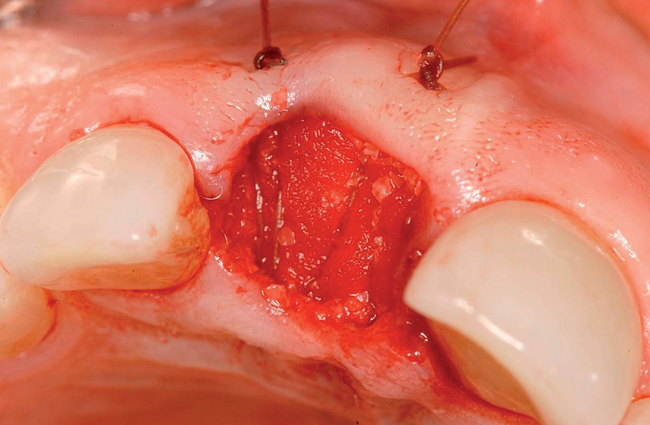
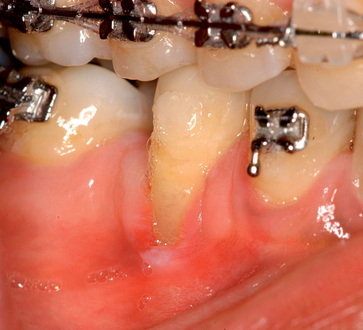
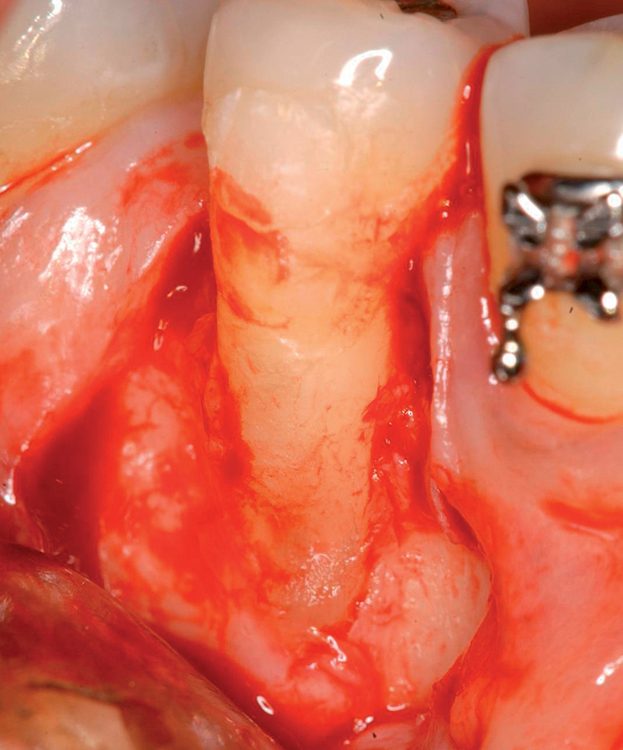
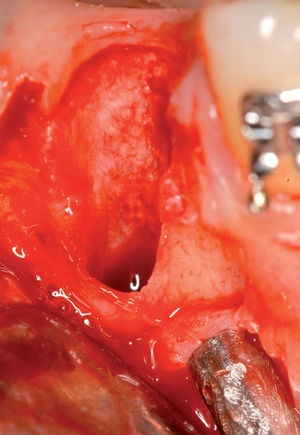
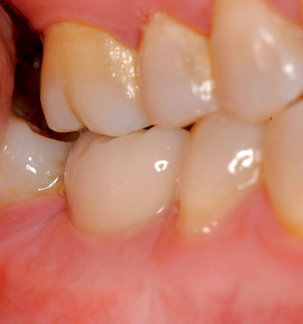
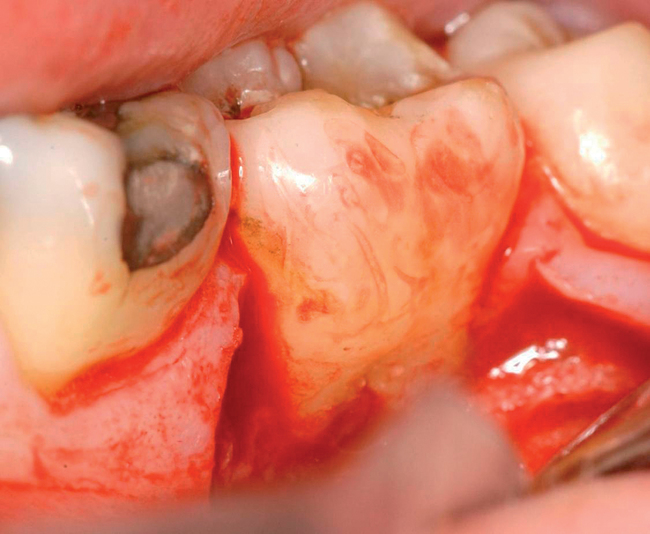
With greater than 6 mm of labial bone loss, implant placement is not as predictable because less labial bone is present, and the chance of micromotion affecting implant integration is higher. In this scenario, the site is grafted, and after the graft has consolidated, an implant is placed (Figures 7-4 to 7-6).
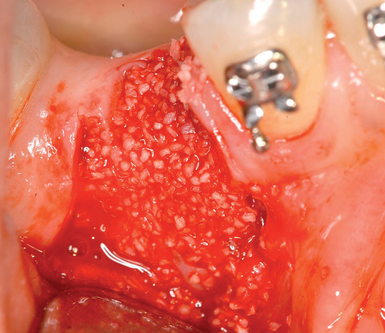
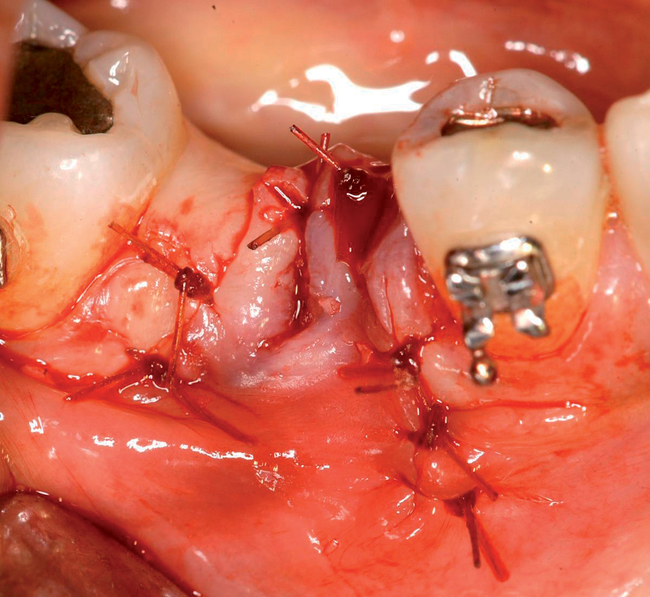
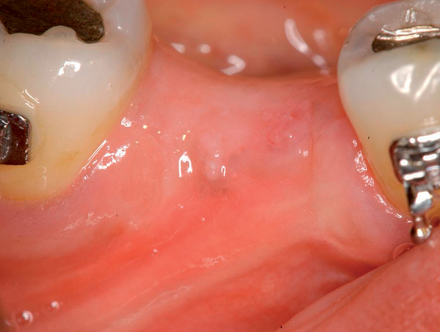
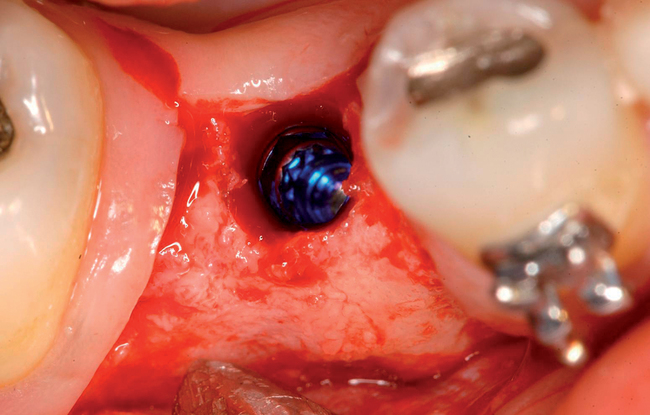
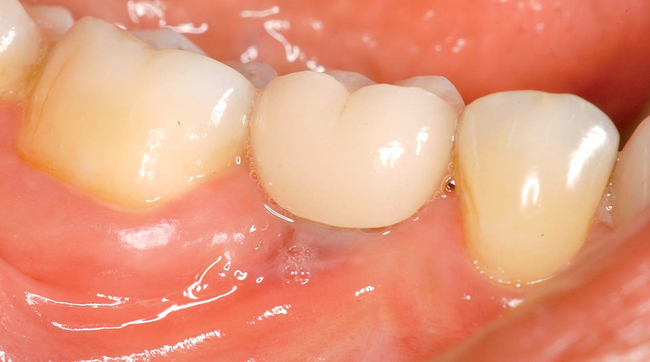
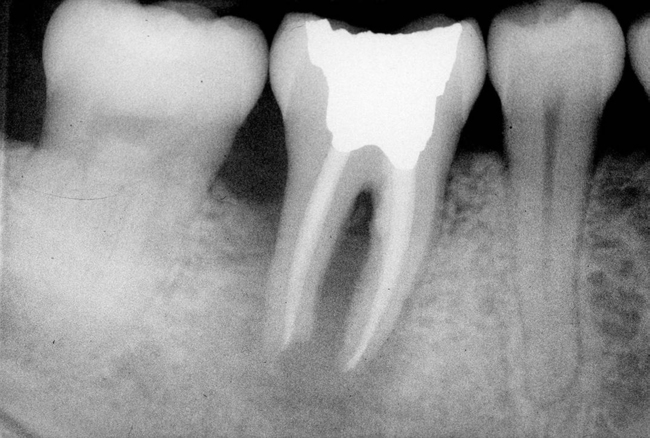
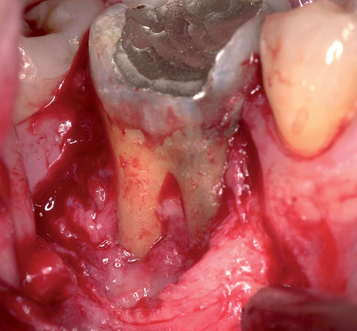
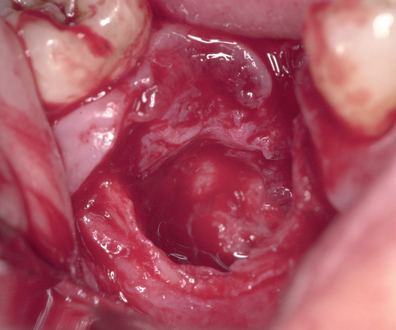
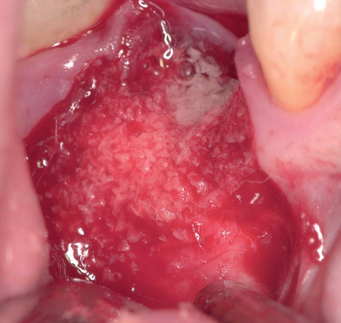
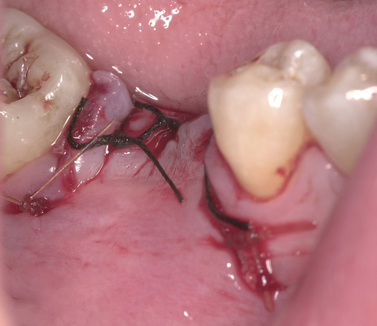
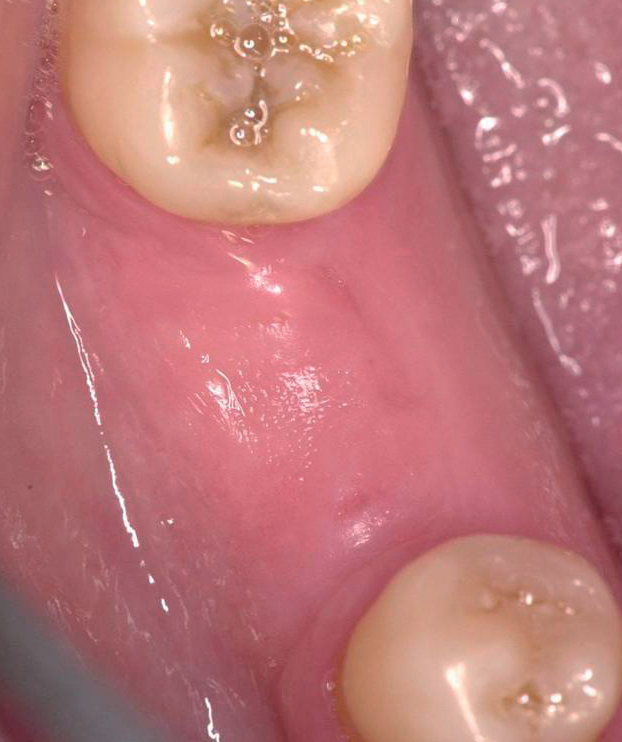
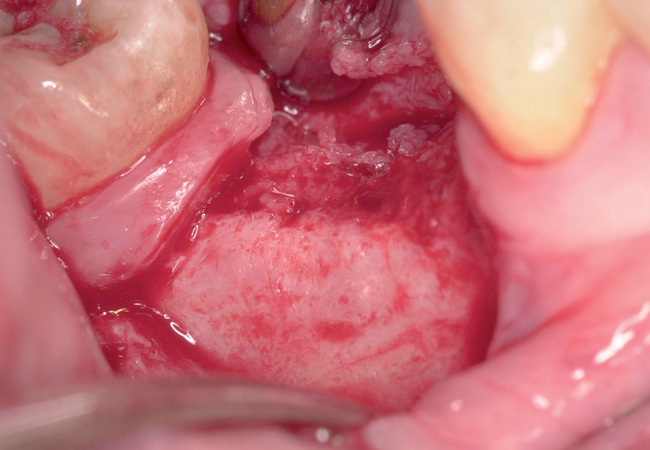
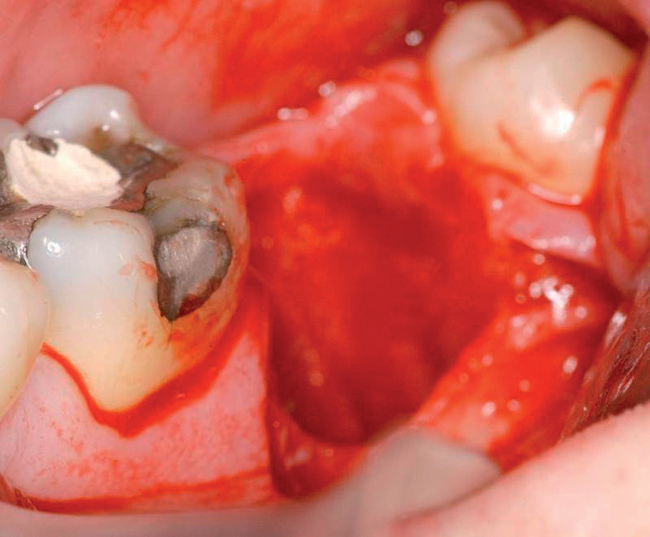
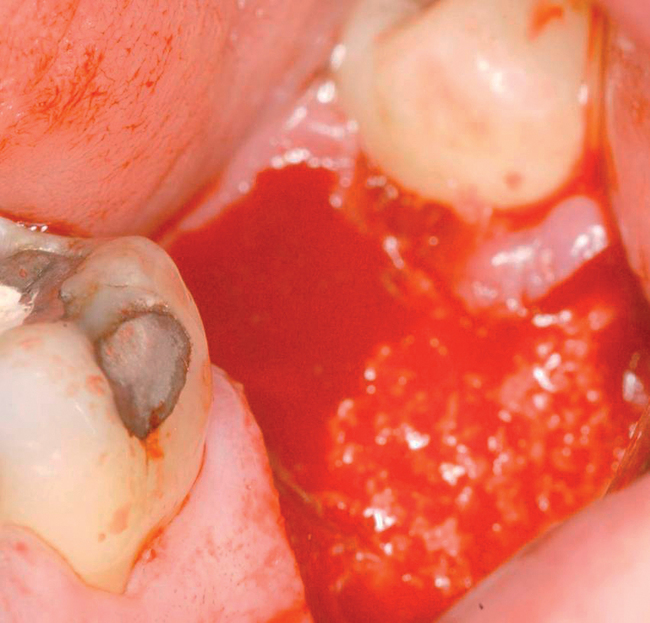
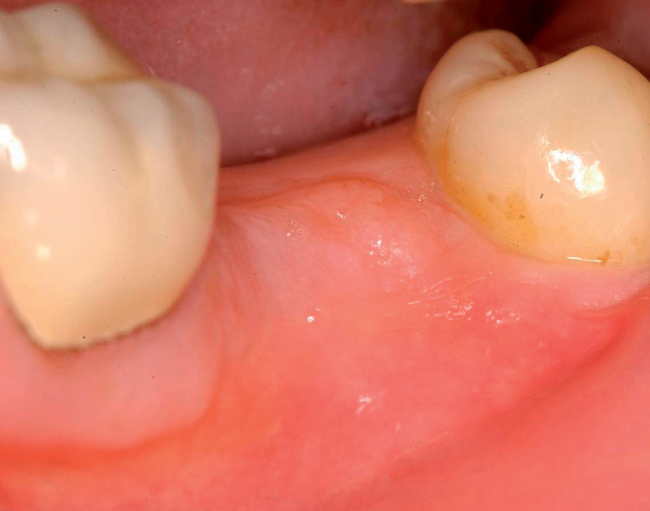
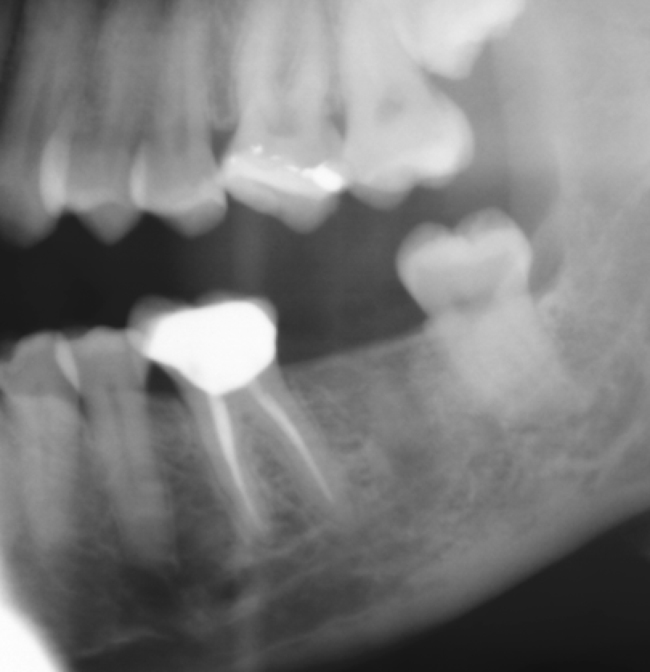
Surgical technique
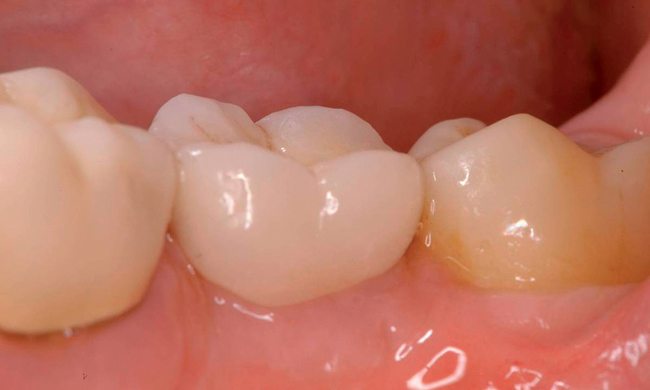
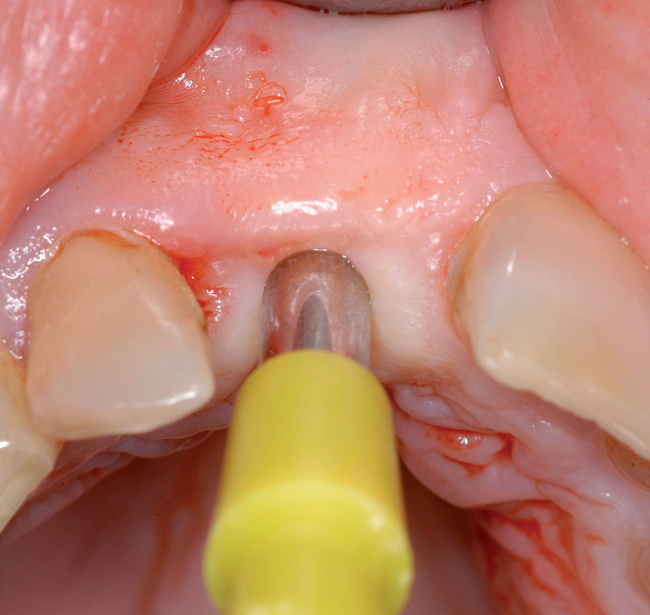
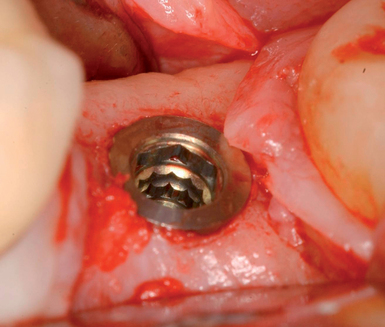
When the treatment plan calls for extraction and grafting without immediate implant placement, an Essix type of a vacuum formed clear thermoformed plastic material temporary prosthesis should be made to provide the patient with immediate temporization with a removable tooth supported device that can be made with no pressure on the surgical site. The crown within the Essix gently approximates to the papilla to provide support without putting pressure on the crestal aspect of the ridge. Tissue-borne temporaries are not recommended. Sixteen weeks after extraction and grafting, the implant can be placed and immediately provisionalized if indicated (Table 7-1).
TABLE 7-1
Grafting the Extraction Site: Anterior Teeth, Including the Premolars
| Procedural Steps | Comments |
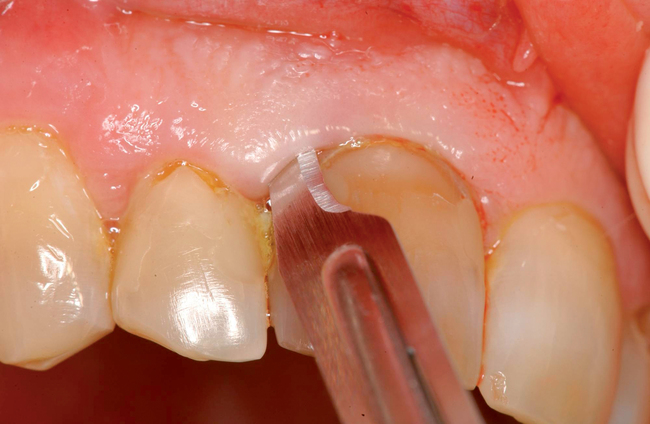
| Make an incision in the sulcus around the tooth. | Use a small scalpel blade; maintain all gingiva. |
| Use a small periosteal elevator (Hirschfeld #20) to identify the junction of tooth and bone. | A small periosteal elevator prevents trauma to the gingiva. Dissect only to identify the bone–tooth junction by feeling, without elevation of the periosteum. |
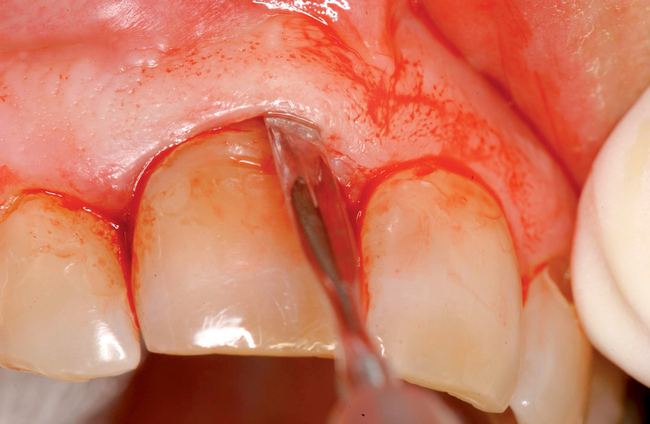
| Use a periotome instrument to separate the bone from the tooth. Use a handheld, piezosurgery, or water-cooled laser. |
Use gentle pressure or gentle mallet to preserve the labial bone. The tooth should be mobile after this step. |
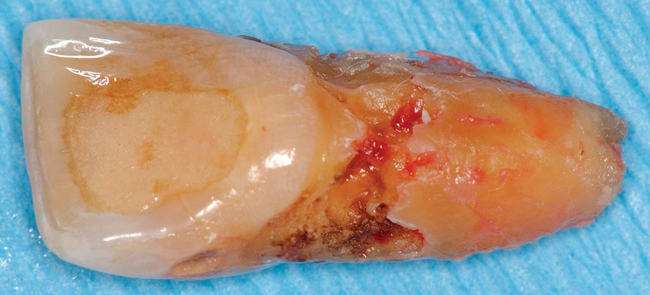
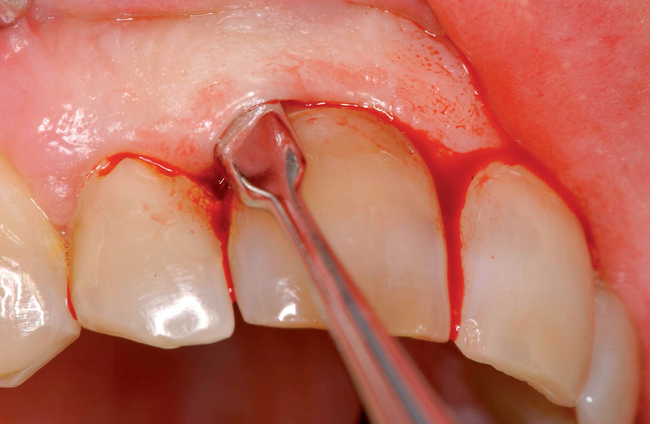
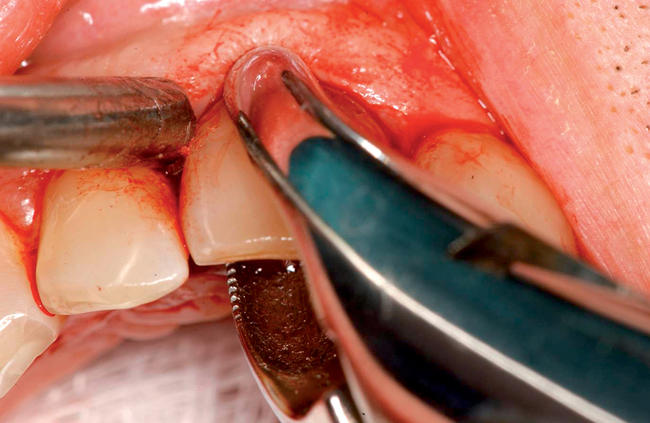
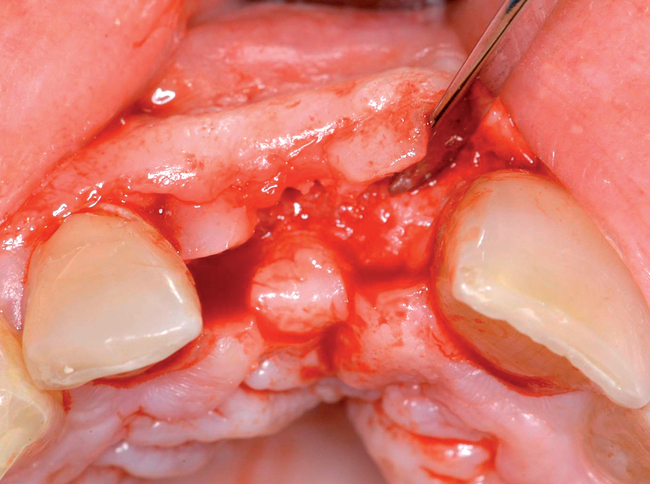
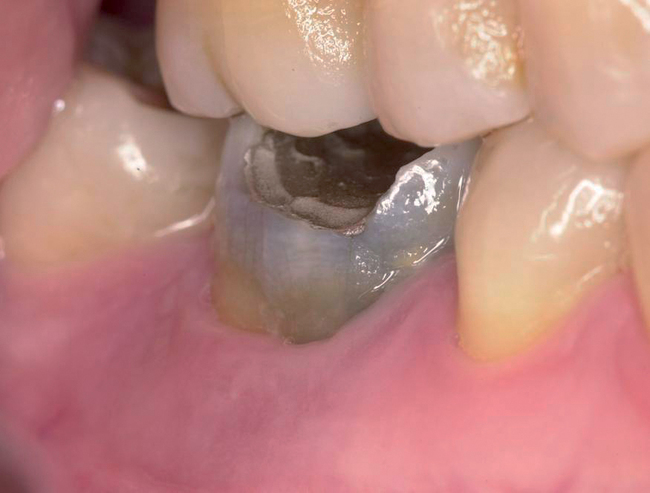
| Extract the tooth. | Remove the tooth without trauma to the labial bone. Use rotary movements and pull the tooth rather than sublux it. |
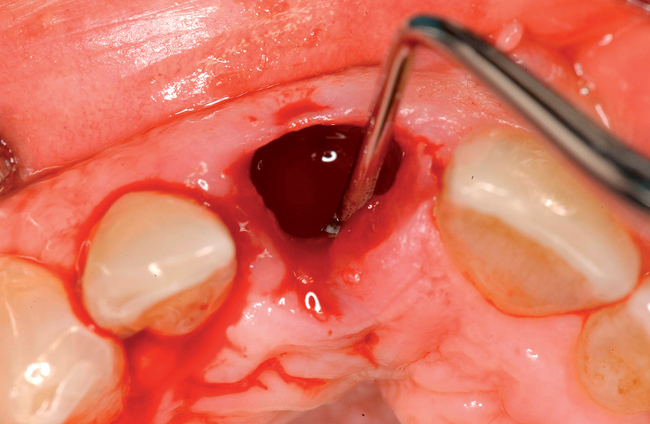
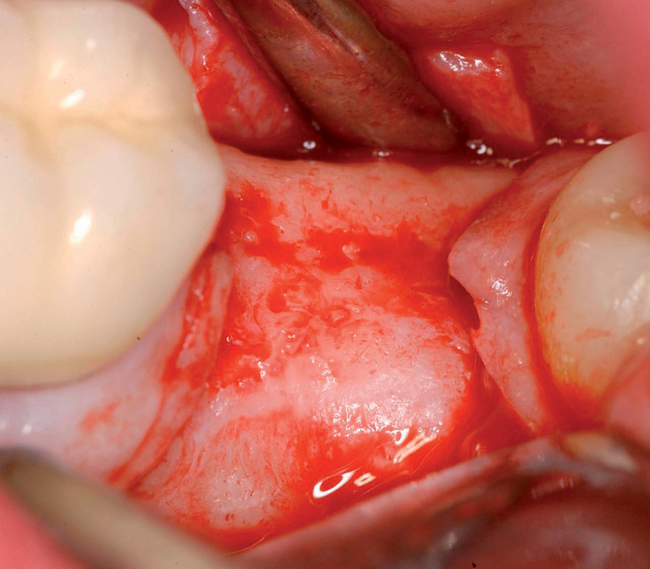
| Gently curette granulation tissue from the socket. | Remove only the granulation tissue. Do not scrape the bone excessively. |
| Evaluate the levels of bone on the mesial, labial, distal, and palatal aspects of the socket. | This step provides insight into the timing of future procedures. |
Stay updated, free dental videos. Join our Telegram channel

VIDEdental - Online dental courses