Chapter 7 Antimicrobial chemotherapy
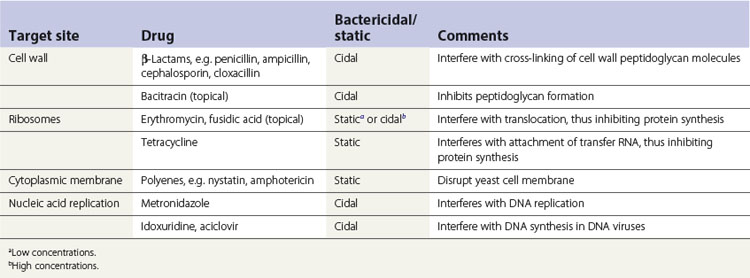
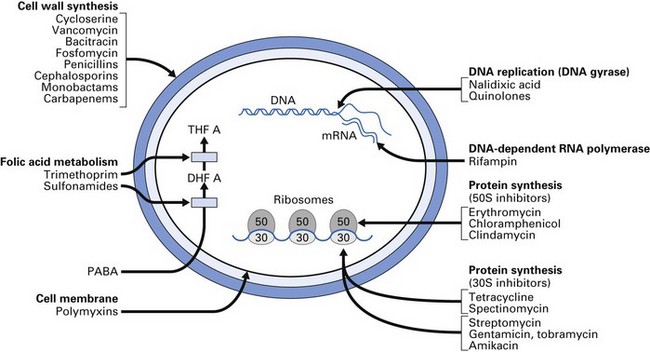
Bacteriostatic and bactericidal antimicrobial agents
Principles of antimicrobial therapy
(Note: this is not an exhaustive list.)
Spectrum of activity of antimicrobial agents
Broad-spectrum antimicrobials (e.g. tetracyclines, ampicillins) are active against many Gram-positive and Gram-negative bacteria, and they are often used for empirical or ‘blind’ treatment of infections when the likely causative pathogen is unknown. This unfortunately leads to ‘abuse’ of broad-spectrum agents, with the consequent emergence of resistance in organisms that were originally sensitive to the drug. The spectrum of activity of some broad-spectrum and narrow-spectrum antimicrobial agents is shown in Table 7.2.
Table 7.2 Spectrum of activity of some commonly used antimicrobial agents
| Drug | Spectrum |
|---|---|
| Phenoxymethylpenicillin (penicillin V) | 1. Aerobic Gram-positives (e.g. streptococci, pneumococci, β-lactamase-negative) |
| 2. Anaerobic Gram-positives (e.g. anaerobic streptococci) | |
| 3. Anaerobic Gram-negatives (e.g. most Bacteroides, fusobacteria, Veillonella) | |
| Penicillinase-resistant penicillins (e.g. flucloxacillin) | All the above, including β-lactamase-producing staphylococci |
| Ampicillin | As for penicillin, also includes Haemophilus spp. |
| Cephalosporins | As for penicillin, also includes some coliforms |
| Erythromycin | Gram-positives mainly but some anaerobes not susceptible at levels obtained by oral administration |
| Tetracycline | Broad-spectrum. Many Gram-positives and -negatives |
| Metronidazole | All strict anaerobes are sensitive, including some protozoa. Of questionable value for facultative anaerobes |
Antimicrobial prophylaxis
Aims
The aims of antimicrobial prophylaxis are:
Pharmacodynamics of antimicrobials
Drug interactions
Drug interactions are becoming increasingly common owing to the extensive use of a variety of drugs. For instance, antibiotics such as penicillin and erythromycin can significantly reduce the efficacy of some oral contraceptives, and antacids can interfere with the action of tetracyclines. All clinicians should therefore be aware of the drug interactions of any antimicrobial they prescribe. The major drug interactions of antimicrobials commonly used in dentistry are given in Table 7.3.
Table 7.3 Some drug interactions of antimicrobials commonly used in dentistry
| Drug affected | Drug interacting | Effect |
|---|---|---|
| Penicillins | Probenecid, neomycin | May potentiate the effect of penicillin. Reduced absorption |
| Erythromycin | Theophylline | Increase theophylline levels, leading to potential toxicity |
| Cephalosporins | Gentamicin | Additive effect leading to nephrotoxicity |
| Furosemide (Lasix) | Possible increase in nephrotoxicity | |
| Tetracycline | Antacids, dairy products, oral iron, zinc sulphate | Reduced absorption |
| Metronidazole | Alcohol | ‘Antabuse’ effect |
| Disulfiram, phenobarbital, phenytoin | Reduced effect |