6
Regenerative Endodontics (Revitalization/Revascularization)
Mahmoud Torabinejad,1 Robert P. Corr,2 and George T.-J. Huang3
1 Department of Endodontics, Loma Linda University School of Dentistry, USA
2 Private Practice, USA
3 Department of Bioscience Research, University of Tennessee Health Science Center, USA
INTRODUCTION
Pulpal necrosis generally occurs as a result of bacterial infection from caries, infractions, or frank exposure of the pulp to contamination of the oral cavity (Kakehashi et al. 1965). Traumatic luxation or avulsion injuries associated with severance of the blood supply result in pulpal ischemia, which also leads to necrosis and often a secondary infection with bacteria (Tsukamoto-Tanaka et al. 2006). Teeth with necrotic and infected pulps are routinely treated with endodontic procedures involving cleaning, shaping, and obturation of the root canal systems with a high rate of long-term success (Torabinejad et al. 2007). The consequence of pulpal necrosis in immature teeth, however, is the cessation of root development, making endodontic treatment with conventional techniques and materials difficult or impossible. Immature teeth have open and often divergent apices that are not suitable for complete cleaning and obturation with traditional materials. In addition, because of their thin walls, these teeth are susceptible to fracture after treatment (Kerekes et al. 1980).
Obtaining an adequate apical seal is widely accepted as a tenet of endodontic therapy (Schilder 1967). Teeth with necrotic pulps and immature apices present special challenges to clinicians during obturation. Apexification procedures that involve the production of an induced or artificial apical stop can allow for condensation of obturation materials. However, apexification procedures do not promote continuation of root development, nor do they increase fracture resistance of the root walls.
The ideal outcome for a necrotic immature tooth would be the regeneration of pulp tissue into a canal which is capable of promoting the continuation of normal root development. The advantages of pulp regeneration lie in the potential for reinforcement of dentinal walls by deposition of hard tissue and the potential for the development of an apical morphology more appropriate for conventional endodontic therapy if future treatment becomes necessary.
The potential for revascularization and continued development of replanted teeth has been well-documented in the dental literature. However, the presence of infection has been shown to interfere with this process (Ham et al. 1972; Kling et al. 1986; Cvek et al. 1990b). It has therefore traditionally been thought that successful revascularization cannot be an expected outcome after a tooth has become infected. However, there is a growing body of evidence to suggest that revitalization of the pulp space, along with continued growth of the root, may in fact be possible after pulpal necrosis and apical pathosis in teeth with immature apices. Nineteen case reports and 14 case series have been published demonstrating apparent continuation of root development of immature teeth with acute apical abscesses, as evidenced by deposition of hard tissue along the canal walls (Rule & Winter 1966; Nevins et al. 1977; Iwaya et al. 2001; Banchs & Trope 2004; Chueh & Huang 2006; Cotti et al. 2008; Jung et al. 2008; Shah et al. 2008; Chueh et al. 2009; Ding et al. 2009; Bose et al. 2009; Reynolds et al. 2009; Shin et al. 2009; Mendoza et al. 2010; Petrino et al. 2010; Thomson & Kahler 2010; Nosrat et al. 2011; Cehreli et al. 2011, 2012; Chen et al. 2011; Jung et al. 2011; Aggarwal et al. 2012; Jadhav et al. 2012; Jeeruphan et al. 2012; Kim et al. 2012; Lenzi & Trope 2012; Miller et al. 2012; Chen et al. 2013; Keswani & Pandey 2013; Soares Ade et al. 2013; Yang et al. 2013). A majority of these cases have predominantly used a blood clot as a scaffold. Platelet-rich plasma (PRP) (Alsousou et al. 2009) has been suggested to be an ideal scaffold for revitalization and regenerative endodontic procedures (Hargreaves et al. 2008; Ding et al. 2009). Platelet-rich plasma has been shown to contain growth factors, stimulate collagen production, recruit other cells to the site of injury, produce anti-inflammatory agents, initiate vascular in-growth, induce cell differentiation, control the local inflammatory response, and improve soft and hard tissue wound healing (Hiremath et al. 2008). Torabinejad and Turman used PRP instead of whole blood as a scaffold for a revitalization procedure in a maxillary premolar with necrotic pulp and open apex (Torabinejad & Turman 2011). After 5.5months, there was radiographic evidence of resolution of periapical lesion, continual root development, and dentinal thickening. Clinically, the tooth responded to vitality tests. The authors suggested that PRP can be an ideal scaffold for regenerative/revitalization procedures.
REVASCULARIZATION AFTER REPLANTATION AND AUTOTRANSPLANTATION
Dental trauma resulting in the severance of blood supply to the pulp has been shown to cause necrosis of the tissue from ischemia (Tsukamoto-Tanaka et al. 2006). The potential for revascularization of the pulp space with functional tissue capable of hard tissue deposition after such dental trauma has been well-documented in the literature. A number of case reports present evidence of revascularization of replanted immature teeth, with reports of continued root thickening and apical closure, sensitivity to thermal stimulus and electrical pulp testing, and normal laser Doppler flowmetry readings (Fuss 1985; Johnson et al. 1985; Mesaros & Trope 1997). Andreasen et al. reported a successful revascularization rate of 34 percent in a prospective study that included 94 replanted immature teeth (Andreasen et al. 1995). Similar results have been found by other investigators (Sheppard & Burich 1980; Kling et al. 1986; Cvek et al. 1990a; Yanpiset & Trope 2000). Although these revascularization rates appear relatively low, in vivo animal studies suggest that pretreatment with topical antibiotics may increase the success of revascularization to 90 percent (Yanpiset & Trope 2000; Ritter et al. 2004).
A number of animal studies in rats, cats, dogs, and monkeys have been published, which provide the histological picture of revascularization after experimental replantation (Kvinnsland & Heyeraas 1989; Yanpiset & Trope 2000; Ritter et al. 2004; Tsukamoto-Tanaka et al. 2006). Replanted teeth in animal models generally show a pattern of slow pulpal degeneration, followed by replacement of the degenerated tissue by in-growth of new tissue through the apical foramen. Soft tissue has been observed to advance coronally to eliminate all the necrotic pulp tissue and fill the pulp chamber in 30 days (Monsour 1971; Skoglund & Tronstad 1981; Kvinnsland & Heyeraas 1989; Tsukamoto-Tanaka et al. 2006). However, few teeth in these studies showed normal pulp tissue with a normal odontoblastic layer and tubular dentin. Instead, the majority of specimens were reported to show an absence of odontoblasts and large areas of the original pulpal cavity filled with hard tissue resembling bone or cementum, which has been described as osteodentin by many investigators (Kvinnsland & Heyeraas 1989; Yanpiset & Trope 2000; Ritter et al. 2004). Soft tissue with morphological characteristics of normal pulp is a rare finding in experimentally replanted immature teeth. Osteodentin formation represents the typical healing pattern reported after revascularization of the pulp space.
Autotransplantation is a clinical situation that is very similar to that of the traumatically avulsed tooth, although performed under controlled surgical and aseptic conditions with minimal extraoral time. Under optimal conditions, success has been reported at 94% (Bauss et al. 2002). Zhao et al. transplanted teeth from green fluorescent protein (GFP) transgenic rats into extraction sockets of wild-type recipient rats (Zhao et al. 2007). The use of the GFP-transgenic rats allowed for later differentiation of donor versus host cells. Using immunohistochemical markers, they confirmed the presence of both bone matrix and dentin matrix in the revascularized pulp tissue. All of the cells associated with the dentin matrix were immunopositive for GFP indicating that the donor pulp tissue formed the matrix. The absence of GFP-negative cells (host cells) implies that the periapical mesenchymal tissues in the socket were not responsible for differentiation into odontoblasts. These findings suggest that some of the original pulp tissue may need to persist in order for odontoblasts to be present after healing.
Despite the large body of literature regarding the revascularization process in transplanted and replanted teeth, the findings cannot be directly applied to nontraumatized teeth with necrotic pulps. The pulp of an avulsed tooth is generally uncontaminated, unless exposed to bacterial insult from exposure during extraoral storage (Love 1996). Furthermore, the replanted tooth contains the amputated pulp tissue graft within the pulp space that likely influences the revascularization process. Although this amputated pulp has been shown to degenerate after extraction (Skoglund 1981), some of the tissue may survive the trauma due to diffusion of nutrients at the apical extent. It has also been suggested that the blood vessels of tissue grafts may re-anastamose with, or provide a nonvital conduit for, the new vasculature during angiogenesis (Barrett & Reade 1981; Goncalves et al. 2007).
REVITALIZATION OF NONVITAL-INFECTED TEETH IN ANIMALS
In 1972, Ham and colleagues experimentally exposed 17 immature teeth in three monkeys to induce development of apical periodontitis (Ham et al. 1972). The canals were then cleaned using broaches and hypochlorite. A paper point saturated in camphorated parachlorophenol was sealed in each canal for 3 days. The walls were subsequently filed with large diameter hedstrom files and beechwood creosote was sealed in the canals for a number of days. Some teeth were filled with calcium hydroxide and Cavit. After producing a blood clot in the remaining teeth, a coronal restoration was placed. The animals were euthanized at varying intervals of up to 165 days. More than half of the controls that remained open to the oral cavity had some degree of vital pulp tissue from one-third to one-half of the canal. Some apical bridging was observed in the calcium hydroxide-treated teeth. However, none of the teeth in the blood clot group showed apical bridging. Bone extended into the canals of some teeth, with some calcified tissue deposited along the walls. Cellular cementum extended into the canal as far as connective tissue migrated. None of the teeth with positive cultures developed any degree of apical closure, and no tissue identifiable as dentin was observed in any of the experimental teeth. Failure to observe consistent revitalization in the experimental groups may be the result of positive bacterial cultures before filling the canals, the use of inadequate coronal restorative materials, or the use of caustic medications in the canal.
Torneck et al. also examined the effects of debridement and disinfection in infected teeth (Torneck et al. 1973). Eight immature teeth in monkeys were exposed to the oral environment to induce pulp and periapical disease. The canal walls were filed and irrigated with saline. Camphorated parachlorophenol was then placed and sealed in the canal with amalgam for intervals of up to two months before obtaining histological samples. The presence of residual pulp and inflamed tissue was noted at the apical end. There was deposition of hard tissue in and about the apical part of the root canal despite severe inflammation (Fig. 6.1). The authors concluded that either the mechanical instrumentation reduced the cell populations that offer potential for regeneration, or the irritating medicament impeded the cellular activity of the pulp. They did not report on how inadequate disinfection may have impacted their results.
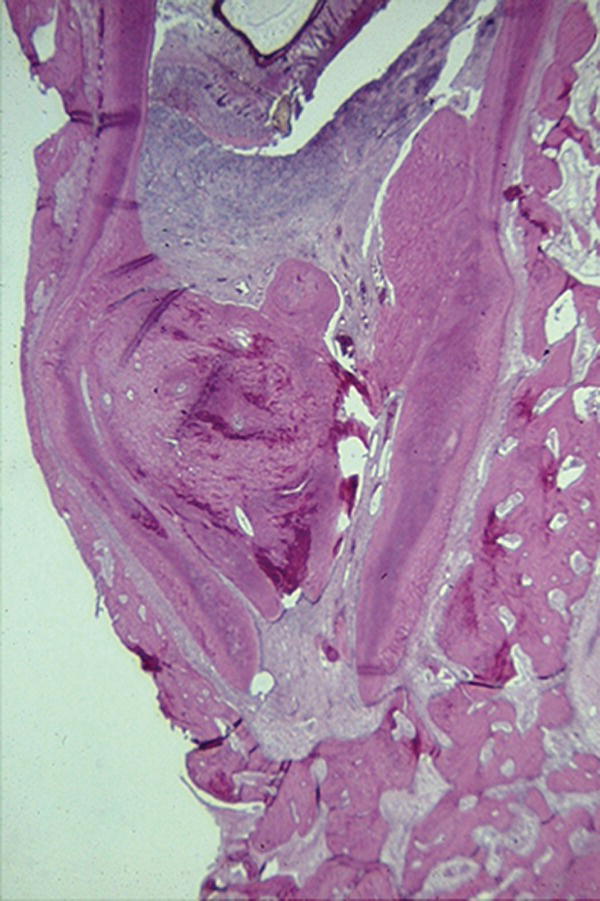
Fig. 6.1 Histological section of a tooth in which the pulp was removed and the root canal was contaminated and then sealed. Despite the presence of an abscess at a lower level, some of Hertwig’s epithelial root sheath survived and produced a dentin-like tissue at the end of the root canal. Courtesy of Dr Calvin Torneck.
Myers and Founatin, in 1974, also infected the pulps of teeth in monkeys. The root canals were then biomechanically cleaned by instrumenting 2 mm beyond the apex and irrigated using 5.25% sodium hypochlorite irrigation (Myers & Fountain 1974). They filled the pulp space with blood only, blood and gelfoam, or nothing, and sealed the coronal access only with Cavit. Histological specimens were examined at intervals up to six months. Very limited in-growth of tissue was observed at the apical extent. The authors reported that the majority of cases demonstrated periapical inflammation, and colonies of microorganisms could be found in the canals. Some of the teeth developed fulminate infections that were reopened to drain. The authors suggested that the lack of successful revitalization might have been due to coronal leakage.
In 1976, Nevins et al. exposed the root canals of rhesus monkeys to oral contamination for one week after removing their dental pulps in order to induce endodontic infections (Nevins et al. 1976). The teeth were then instrumented and initially sealed with a cotton pellet and IRM only. Despite obtaining positive bacterial cultures in all teeth, an experimental collagen phosphate gel was introduced into all of the canals 3 days later. Histology was observed after 12 weeks. The teeth treated with calcium hydroxide showed apexification as anticipated. Fifteen of the 21 teeth filled with the collagen-calcium phosphate gel were considered successfully revitalized with apposition of cementum-like tissue onto the canal walls narrowing the lumen and apical foramen. Teeth that were revitalized successfully showed tissues characteristic for cementum, bone, and reparative dentin lining the wall of the root canal for most of its length (Fig. 6.2). Lacerated remnants of pulpal tissue appeared to be actively producing reparative dentin. The authors concluded from their findings that cementoblasts appeared to proliferate and secrete cementum in a coronal direction in close proximity to the root canal dentin. They also postulated that mesenchymal cells within the apical periodontal ligament might have proliferated and differentiated to form hard tissue within the root canal space.
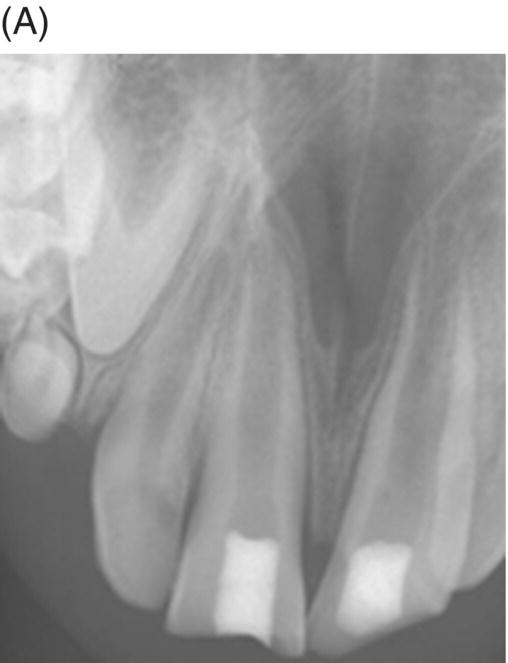
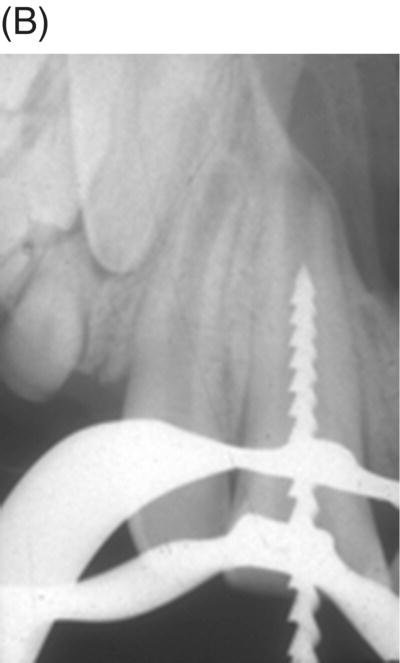
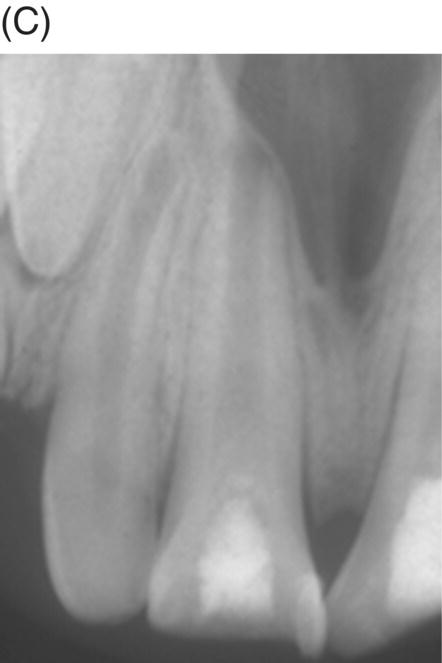
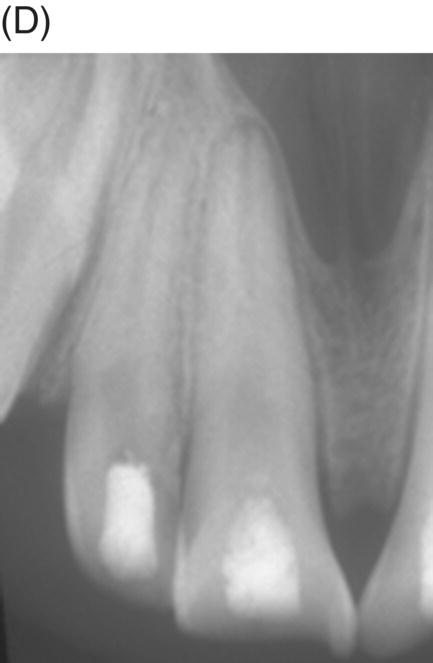
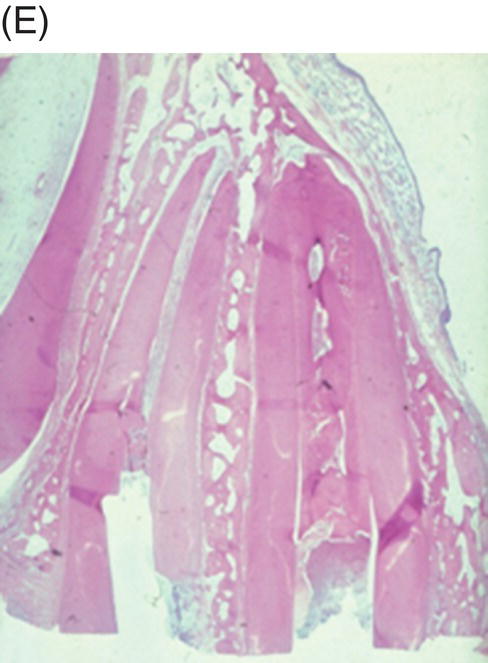
Fig. 6.2 (A), After removing dental pulps in monkeys and contamination of their root canals for a few days (B), they were then biomechanically instrumented (C), filled with an experimental collagen-phosphate gel or calcium hydroxide and then sealed with IRM (D). Radiographic examination of these teeth 6 months later shows closure of the apecies and thickening of the root canal walls (E). Histological examinations of these samples show apexification teeth treated with calcium hydroxide (right incisor) and formation of cementum, bone and reparative dentin lining the wall of the root canal of the left central incisor filled with experimental collagen-phosphate gel. Courtesy of Dr. Alan Nevins
Das and colleagues removed the pulps from 22 teeth in baboons and allowed the canals to remain open to the oral cavity for 60 days (Das et al. 1997). The teeth were then cleaned by conventional filing or with broaches only. After cleaning, half of each group was treated either with formocresol or tetracycline-saturated paper points. After one week, the points were removed, and the coronal portion was sealed with IRM and amalgam. The animals were euthanized at six months. Seven of the nine tetracycline-treated teeth reached root completion, compared to only three of the 10 treated with formocresol. Three of the nine filed teeth reached complete maturation, while seven of the 13 nonfiled teeth reached complete maturation. These findings supported the hypothesis that some tissue remnants may persist that contribute to organizing the in-growth of tissue, and that mechanical shaping or the use of caustic chemicals might remove these important cells.
Thibodeau et al. conducted a study involving immature dog teeth with preexisting necrotic pulps and apical periodontitis (Thibodeau et al. 2007). They sealed oral plaque into their pulp chambers until there was radiographic evidence of apical periodontitis in 48 contaminated canine teeth. The teeth were re-accessed, irrigated with 1.25% sodium hypochlorite without any mechanical instrumentation, and a triple antibiotic paste was placed into the canal. One group of teeth was sealed coronally with MTA and amalgam and received no further treatment. Teeth in the other groups were re-accessed and irrigated to remove the antibiotics, and filled with an induced blood clot, a collagen solution, or a combination of the collagen solution and a blood clot before being sealed coronally. Radiographic and histological evaluation was performed up to three months post-operatively to assess for apical closure, root wall thickening, and the presence of vital tissue in the canals. Overall, the authors found thickened walls and apical closure in 49% and 55% of teeth respectively. Vital tissue was observed in 29% of the teeth. Although no statistical differences between treatment groups were reported, the authors reported that the inclusion of a blood clot in the canal favored revitalization.
Wang et al. later histologically assessed the type of tissues that had grown into the canal space in the study by Thibodeau (Wang et al. 2010). Similar to previous animal studies, cementum-like and bone-like tissue were noted to be present inside the canal spaces. It was found that the cementum-like and bone-like tissues were responsible for the thickening of the canal walls. The cementum deposition at the apex was responsible for the increase of the root length (Fig. 6.3). They concluded that the tissues found were not of pulpal origin, and that the outcome of the revitalization process may not be tissue regeneration, but instead be tissue repair.
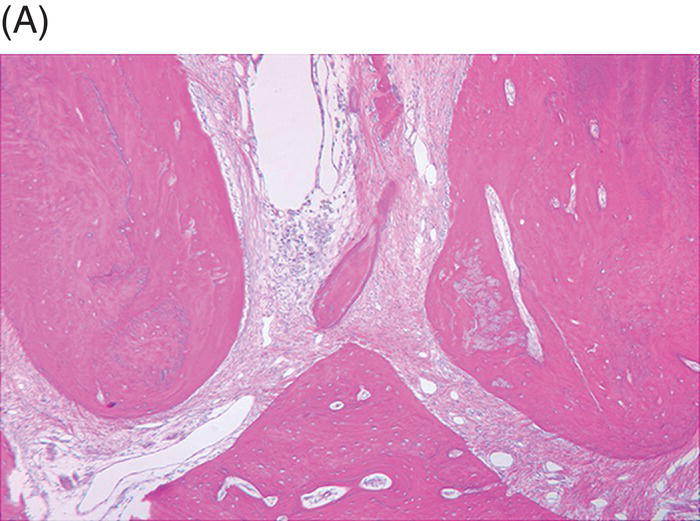
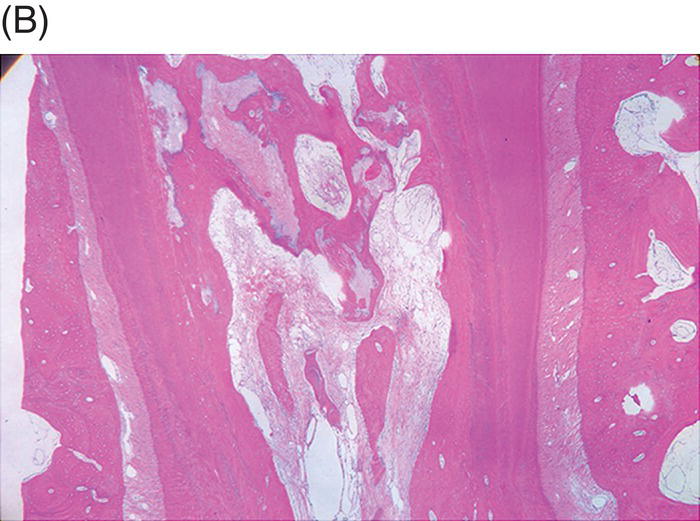
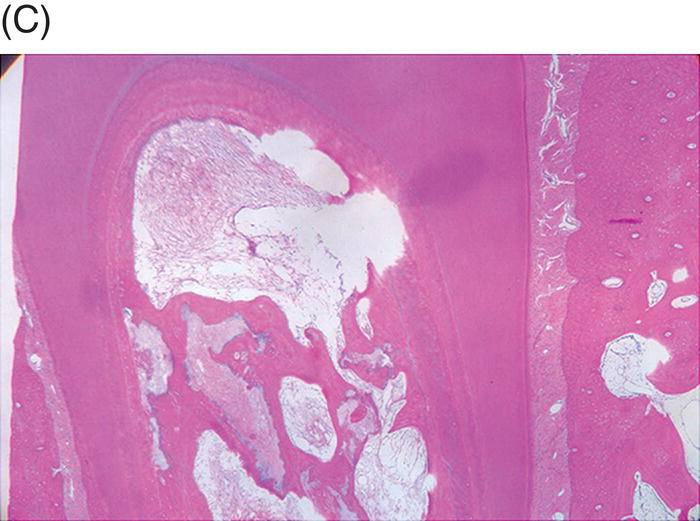
Fig. 6.3 After induction of apical periodontitis in immature dog teeth, they were disinfected using triple antibiotics. After removing the antibiotics, blood clot, a collagen solution, or a combination of the collagen solution and a blood clot were used as scaffolds. The teeth were then sealed coronally with MTA and amalgam. Histological examinations of the specimens three months later show an in-growth of bone into the (A) apical, (B) middle and (C), coronal sections of the root canal space. Courtesy of Dr B Thibodeau.
Zuong and associates compared root development and periapical healing between revitalization and apexification of immature dog teeth after inducing periapical periodontitis (Zuong et al. 2010). Revitalization and apexification were done in three teeth each, and radiographic examination was done at 1, 4, and 8 weeks postoperatively. After 8 weeks, the animals were euthanized. The revitalization group showed more consistent periapical healing. Apical closure was apparent with no significant change in dentin thickness in comparison to the apexification group. Histologically, the revitalized roots contained granulation tissue with large amounts of calcification on the canal walls.
da Silva and colleagues investigated the amount of inflammation and degree of repair of periapical tissues in infected immature dog teeth using two different techniques (da Silva et al. 2010). In their study, 56 roots were accessed and irrigated with hydrogen peroxide and sodium hypochlorite. In 28 roots, apical negative pressure irrigation using the EndoVac system was done with sodium hypochlorite. In 28 other roots, positive pressure irrigation with sodium hypochlorite followed by triple antibiotic dressing for 2 weeks was done. All canals were irrigated with sterile saline, dried with paper points, and the coronal accesses were restored with a double seal of MTA and amalgam. Histological assessment was carried out after 3 months and the authors reported that the negative pressure group showed significantly less inflammatory infiltrate at the periapical region compared to those samples treated with the positive irrigation technique. They also found out that there was an in-growth of connective tissue of periodontal origin containing fibroblasts and blood vessels inside the canals of both groups. They suggested that the EndoVac system may be an alternative to triple antibiotic paste for the revitalization process in necrotic immature teeth with apical periodontitis.
Yamauchi et al. treated 64 teeth in six dogs with apical periodontitis using a revitalization procedure (Yamauchi et al. 2011). Four different materials were left in the canals: blood clot, blood clot and collagen scaffold, blood clot with exposure of dentin matrix from ethylenediaminetetra-acetic acid (EDTA) treatment, and blood clot with collagen matrix and EDTA. All teeth showed increased wall thickness with resolving apical pathosis after 3.5 months. In the groups containing the collagen scaffold, more apical closure and a statistically significant amount of mineralized tissue was observed in comparison to groups not containing a collagen scaffold. When EDTA was used, histological evidence of hair-like projections from the dentin-associated mineralized tissue appeared to embed into the dentin wall. This illustrates the effects of EDTA, which promotes adherence of the mineralized tissue to the root canal walls.
Scarparo and colleagues compared the root development between teeth with vital pulp tissue, teeth with necrotic pulps, and teeth with necrotic pulps that subsequently underwent revitalization treatment procedures in immature rat molars (Scarparo et al. 2011). Initially, 36 teeth were accessed, their pulps were removed and left open to the oral cavity for 3 weeks to produce apical lesions. Eighteen teeth were left open for the remainder of the experiment. The remaining 18 teeth were then instrumented only in the cervical third of the canals, irrigated with sodium hypochlorite, dressed with a triple antibiotic paste, and sealed with a cotton pellet and an amalgam restoration. Histological samples were harvested after 3, 6, and 9 weeks. The results show that the control teeth with vital pulp showed no inflammation and normal signs of root development as expected. The control teeth with necrotic pulp showed periapical lesions and reduced root length and wall thickness. The teeth that were treated using revitalization procedures showed reduced periapical lesions with increased root length and wall thickness. About half of the revitalized group showed formation of cementum-like tissue in the apical portion of the roots while other samples in the same group showed an ingrowth of connective tissue into the root canal.
In 2011, Buhrley et al. evaluated the healing response in noninfected immature ferret cuspid teeth after extirpation of the pulp tissues followed by induction of a blood clot (Buhrley et al. 2011). Histological samples obtained three months after the procedures illustrated an ingrowth of bone that was contiguous with surrounding periapical bone (Fig. 6.4).
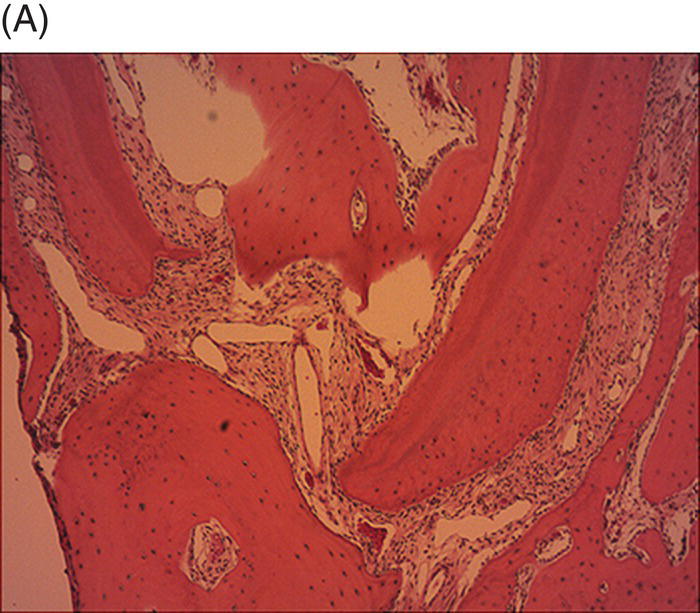
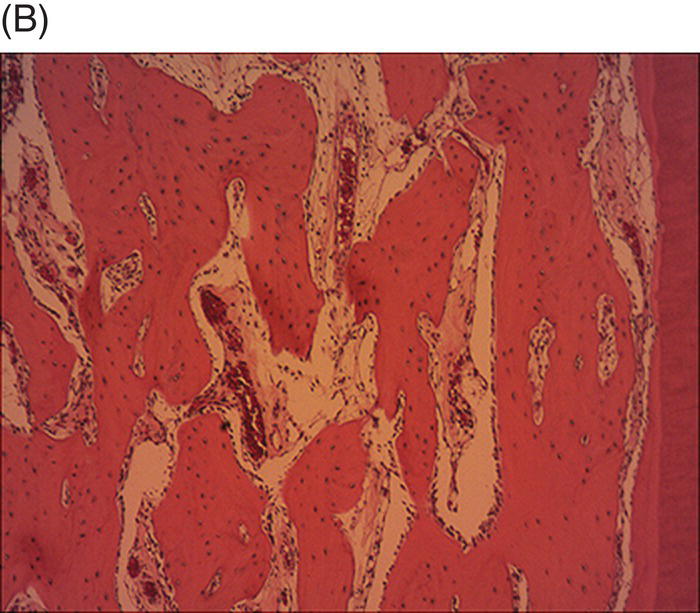
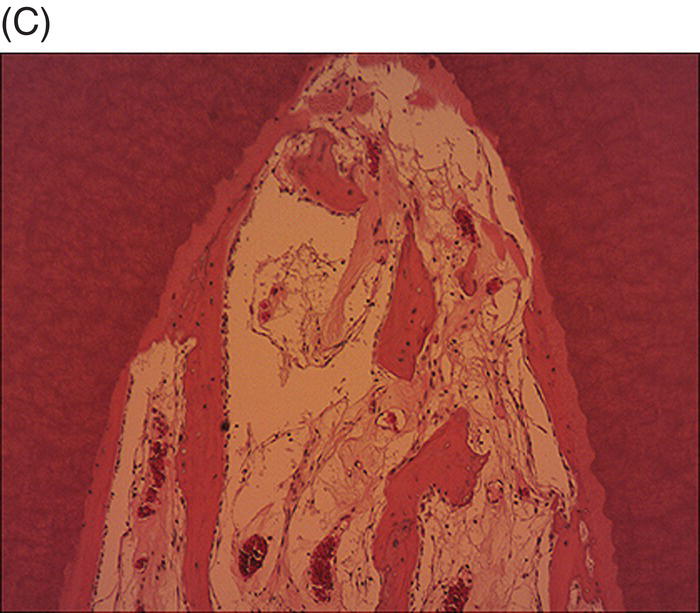
Fig. 6.4 After pulp extirpation in immature ferret cuspid teeth, blood clots were formed and covered with MTA and permanent filling material. Histological samples after 3 months illustrated an in-growth of bone into the (A), apical, (B), middle and (C), coronal sections of the root canal space.
Based on the available literature, there appears to be compelling evidence that revitalization is possible despite pulpal necrosis and apical periodontitis. From previous studies, the evidence regarding the phenotypes of tissues has been found to consist of tissues similar to bone, cementum, and connective tissue. Much of the histological evidence available is from studies that were conducted several decades ago, with some of the studies reporting known bacterial contamination or the use of materials that are inadequate to provide an effective resistance to coronal leakage. Other limitations in the previous studies include the use of standard instrumentation to debride the canals, and the use of caustic medicaments and disinfectants, both of which could be detrimental to the tissues that potentially provide the genesis for revitalization.
Although the radiographic appearance of apical closure and canal wall thickening can give the impression that a normal, functional pulp has regenerated, the present histological evidence does not support this assumption, and more research is needed to incorporate current protocols.
CLINICAL EVIDENCE FOR REVITALIZATION IN NONVITAL-INFECTED TEETH IN HUMANS
Several case reports and series have demonstrated the potential for a nonvital, necrotic and infected immature tooth to revitalize, as evidenced by radiographic root thickening, apical closure, or a return of sensibility to thermal and electric pulp testing. In 1966, Rule and Winter presented a case report of continued maturation of a nonvital, immature mandibular premolar with a sinus tract stoma (Rule & Winter 1966). The tooth was accessed without anesthesia and cleaned to the level of bleeding tissue, then dressed with a polyantibiotic paste. Two weeks later, the canal was re-accessed, the antibiotics removed, and resorbable iodoform was introduced before double sealing with zinc oxide–eugenol (ZOE) and amalgam. The root showed continued thickening and apical closure 3 years post-operatively.
Nevins and colleagues reported on a child with a traumatically intruded maxillary lateral incisor that had developed symptoms of irreversible pulpitis and apical periodontitis (Nevins et al. 1977). Endodontic access was made, purulence was observed in the pulp space, and the canal was mechanically debrided while using saline irrigation, removing all but the apical stump of pulp tissue. Collagen-calcium phosphate gel was introduced into the pulp space and the canal was sealed. The child remained asymptomatic; apexogenesis was observed to continue at 7 weeks, and was more pronounced at longer follow up periods (Fig. 6.5).
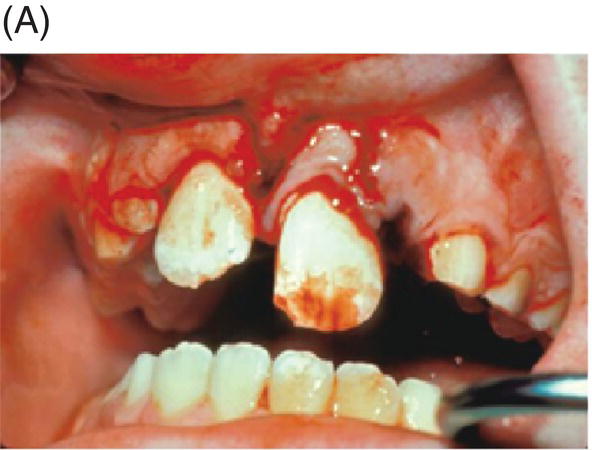
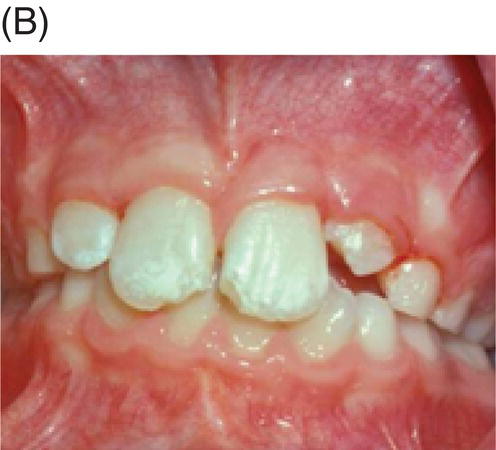
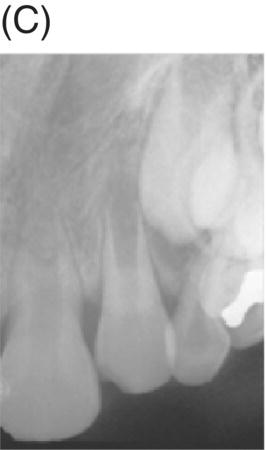
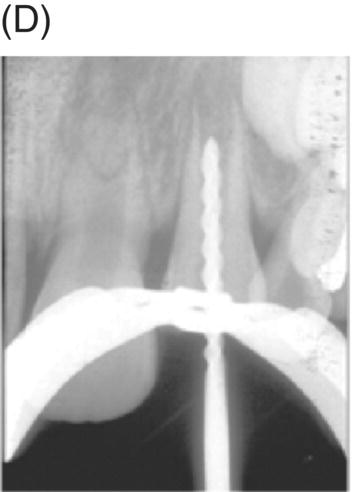
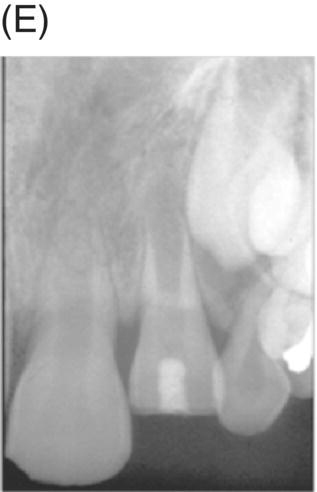
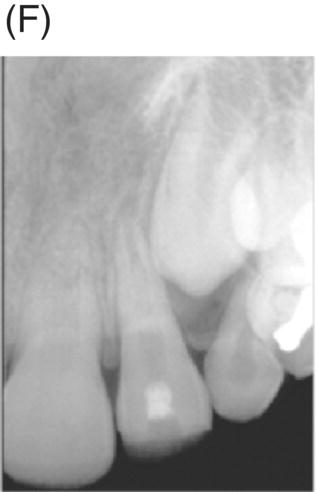
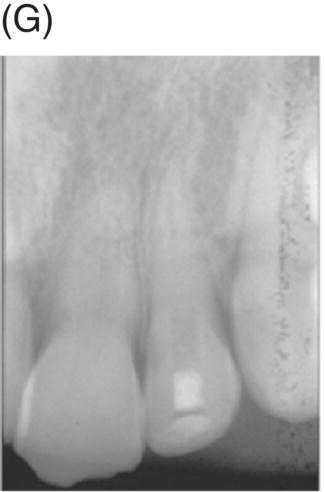
Fig. 6.5 (A) During an accident, a child developed an intruded maxillary lateral incisor. (B) Eight weeks after the tooth re-erupted. (C) Developed symptoms of irreversible pulpitis and apical periodontitis. (D) The tooth was instrumented. (E) The collagen-calcium phosphate gel was placed in the canal. (F) and (G) A radiograph taken 1 year and 3 years later shows closure of the apex and thickening of the tooth walls. Courtesy of Dr. Alan Nevins.
Iwaya et al. reported on a case of a child with an acute apical abscess in a mandibular bicuspid with a necrotic and immature apex that had an apparent fractured occlusal dental tubercle (Iwaya et al. 2001). Purulent exudate was found upon initial access. The tooth was managed over multiple visits with sodium hypochlorite, hydrogen peroxide, and intracanal placement of antibiotics. When vital tissue was observed 5 mm apical to the orifice, the coronal access was sealed with calcium hydroxide and a bonded restoration. Healing of the periapical abscess and complete closure of the apex were observed 2.5 years postoperatively.
Banchs and Trope presented a similar case of a child with a necrotic immature lower premolar secondary to an apparent broken dental tubercle (Banchs & Trope 2004). Associated swelling and sinus tract were observed, and purulent exudate was discovered upon entry into the pulp. They irrigated the canal with sodium hypochlorite and Peridex, without any attempt to mechanically clean the walls. A triple antibiotic paste was placed into the canal for 1 month. After removing the paste 1 month later, a blood clot was induced in the canal and the access was closed with MTA and a resin restoration. The patient was recalled periodically over 2 years. The tooth showed continued development radiographically and was responsive to cold at the 2-year recall.
Chueh and Huang presented four similar cases of children with necrotic mandibular premolars with broken dental tubercles (Chueh & Huang 2006). The teeth were managed with irrigation with sodium hypochlorite and application of calcium hydroxide. All four cases showed continued apical development. The authors attributed lack of coronal deposition of hard tissue in these cases to the placement of calcium hydroxide deep into the canals.
Cotti et al. reported a case of a child with traumatized immature necrotic permanent maxillary incisor with a sinus tract (Cotti et al. 2008). Upon entry, necrotic tissue was debrided from the canal with a spoon excavator, irrigated with sodium hypochlorite, and filled with calcium hydroxide. After 15 days, the sinus tract disappeared, and the calcium hydroxide was removed, bleeding was stimulated from the apex, and it was finally restored with MTA and final restoration. At the 2.5 year recall, there were radiographic signs of continual root development and dentinal wall thickening.
Jung et al. presented a case series of children with immature permanent teeth having necrotic pulps and apical periodontitis (Jung et al. 2008). In four teeth with necrotic pulps, the canals were irrigated with sodium hypochlorite, medicated with triple antibiotic paste, followed by evoking of bleeding to form a blood clot, sealed with MTA, and final restoration was placed. In five other cases where there were remnants of vital tissue inside the canal, the same protocol was followed except there was no evoking of bleeding. Apical closure, dentinal wall thickening, and increase in root lengths were observed in all of the cases.
In a case series conducted by Shah et al., 14 nonvital, immature teeth with periapical periodontitis were followed up to three and a half years after attempting revitalization procedures (Shah et al. 2008). Teeth were accessed, irrigated (after minimal filing) with hydrogen peroxide and sodium hypochlorite, and dressed with intracanal formocresol. Subsequently, bleeding was initiated from the periapical tissue, and the tooth was restored with glass ionomer cement with no MTA. They reported no signs and/or symptoms of pathosis in 11 out of 14 cases, periapical healing in 8 out 14 cases with thickening in dentinal wall, and an increase in root length in 10 out of 14 cases.
Ding et al. followed 12 cases of children having necrotic, immature teeth with or without clinical signs of periapical pathology (Ding et al. 2009). The root canals were irrigated with sodium hypochlorite and triple antibiotic paste was used as intracanal medication. After 1 week, bleeding was induced by manipulation with endodontic file, and the teeth were restored with MTA and composite restoration. Due to unresolved symptoms at recall, six teeth required further treatment using an apexification procecure. Three patients were not available for recall. Only three remaining teeth became symptom-free, showed complete root development, and tested positively to vital pulp testing after 15 months after treatment. The authors mention that one of the reasons for failure was inability to induce bleeding after canal disinfection.
Bose et al. compared the degree of continual root development and dentinal wall thickening between 48 teeth that underwent revitalization procedures with 40 teeth that underwent either apexification or regular nonsurgical root canal treatment (Bose et al. 2009). Those that went through revitalization were necrotic, immature permanent teeth with or without evidence of periapical pathology. The canal infections were managed by triple antibiotic past/>
Stay updated, free dental videos. Join our Telegram channel

VIDEdental - Online dental courses


