6
Erosion
6.1 Introduction
Dental erosion is an often painless, progressive, irreversible loss of hard tooth tissue, first the enamel and denuded cementum and subsequently the exposed dentine. Erosion occurs due to chemical dissolution of these tissues by mechanico-acidic or idiopathic processes.
In material science, erosion is viewed as wear caused by flow of fluid with abrasive particles.243 “Corrosion” is a physicochemical or electrochemical process.116 117 Hence, erosion is also called “corrosive wear”.192
Enamel dissolves in distilled water,169 but not in saliva, which, at pH 7, is supersaturated with respect to the teeth, unless chelation takes place,75 i.e. organic molecules bind calcium ions bivalently.47 At a pH ∼5.5 (or <6)305 saliva is undersaturated and dissolves the enamel: the more acidic the environment, the greater is the enamel surface loss and softening.
Rinsing with 2% citric acid (pH 2.1) makes the saliva undersaturated for 2 minutes. As the acid is washed away and neutralised by the saliva216 the oral environment returns to a state of supersaturation.27
Erosion Versus Caries (Figure 6.1)
Under the plaque layers, bacterial acid production leads to carious white spots (Chapter 5). As with composite etchants,118 292 the amount of minerals dissolved by the strong exogenous acids from the superficial tooth surface layer is too large to be adequately replaced with materials from the deeper subsurface layers; in vitro studies show that the enamel tissue loss increases linearly with time.139 303 The pellicle covering the teeth, regardless of its age,125 protects the enamel somewhat against acids, as does the plaque, which helps to neutralise the acids with calcium and phosphate.142 209 288 The thickness of plaque varies between individuals after a short time of its initiation and is inversely related to the degree of erosion.10 Eroded enamel does not have plaque and high-erosion patients accumulate less plaque than low-erosion individuals.155 In contrast with the dull surface that is produced after etching, eroded enamel is usually shiny, because the softened enamel wears away.65
Figure 6.1 Diagrammatic comparison of the reactions occurring on the tooth surface in caries and enamel.
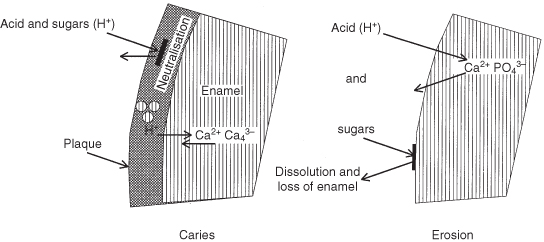
Erosion and Mechanical Factors
Miller (1907) attributed erosion to tooth brushing,215 but later research showed that acidic dissolution initiates erosion (Figure 6.1) rather than wear and salivary characteristics.231 Acidic dissolution leads to loss of a thin surface layer and makes the demineralised, and thus softened, superficial enamel more susceptible to wear,26 80 in particular, immediately after the exposure to the acids.65 116 188 Surface loss through tooth-to-tooth contact and tooth brushing (Figure 6.2) are co-responsible for further development of the erosive lesion,4 28 65 75 195 205 258 as is repeated contact with the lips, cheeks and tongue,112 192 and also prosthetic materials.43 258 At the bottom of the erosive lesion is a zone of highly softened enamel, which is a few micrometers deep.
Figure 6.2 Defect in the cervical part of the root with undermining of the enamel, a consequence of the combined effect of tooth brushing, the toothpaste and, presumably, acid.
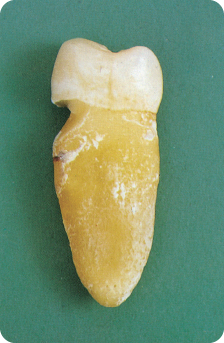
Concerning wear, some studies show interesting findings.
- In an in vitro study, more enamel wear was seen in neutral than under acidic conditions, owing to a smoothing and flattening effect of erosion on the contacting surfaces (but periodontal stress relief and salivary lubrication of the teeth were lacking).79
- When enamel slabs demineralised with citric acid were fixed to the teeth and exposed to saliva for 0, 30 or 60 minutes and then brushed, there was loss of 0.26, 0.22 and 0.20 µm of the superficial layer, respectively:144 that is, the longer the enamel was exposed to the saliva, the lower was the amount lost. The loss due to brushing was 10–27 times greater than that from the control enamel.144 184 Demineralised dentine is also more susceptible to abrasion.18
- The shape of the occlusal lesions in teeth of people living in the Medieval times caused by severe wear associated with a coarse diet closely resembles the defects produced by acids in individuals consuming a contemporary “normal” diet: however, whether a lesion was caused by erosion or wear was not distinguishable on the basis of the lesion shape.95
- In another in vitro study, at pH 5 maintained over a period of several days, the undersaturated environment with a low fluoride concentration was associated with softening of the enamel surface. A somewhat higher fluoride concentration in a more saturated environment led to formation of white spot lesions.304
Role of Saliva
In erosion, there is insufficient dilution, buffering and washing away of the acids by the saliva. Stimulated saliva has a higher buffering capacity than unstimulated saliva. Van Nieuw Amerongen reported that the buffering capacity and the stimulated salivary flow rate seemed normal in 75% of a sample of erosion patients, but in some patients the small amount of the stimulant citric acid lowered the oral pH to less than 5.5.316
Saliva stored in experimental conditions was found to protect the enamel to a greater degree against an erosive challenge than deionised water. The salivary protection in vivo is greater with a greater quantity of saliva and with the deposition of an organic pellicle on the specimen, and varies with different chemical compositions of saliva.123
6.2 Aetiology
6.2.1 Acid Erosion
Several studies show erosion is associated with exogenous acids.8 14 64 74 76 82 89 119 128 134 135 149 172 174 204 207 210 235 254 281 321 341 Variations in effects of acids in rats have been attributed to differences in individual characteristics.135 Tests for determining the validity of these findings are being carried out.
Laboratory Techniques
Several techniques are used to assess the effects of acids on the enamel.23
Scanning electron microscopy 114 and digital image analysis226 are used to measure erosion in vitro.
Microindentation reveals the hardness of the enamel surface before and after contact with acid. The greater the indentation produced by a diamond upon loading, the softer is the enamel.23 In another study, the elasticity of enamel and dentine was not reduced after 10 minutes of immersion in erosive beverages.191
Nanoindentation is sensitive to the initial stages of erosion. The displacement of the registering tip as a function of the applied load is continually monitored and enables calculating the enamel hardness and the plastic and elastic deformation. The technique has been applied to determine reduction in the erosive potential of fluids after addition of calcium and phosphate.23
Profilometry is performed with a stylus that scans a surface partly exposed to an acid. The surface is scanned before and after the exposure. The amount of material lost and the erosive potential of agents are determined from the scan trace.23 210 More recently, laser profilometry has also been used.
Microradiography. A beam of X-rays is directed to an enamel block. The degree of blackening on a photographic plate is used to record the amount of penetrating radiation; the mineral density of the block is mapped.
Chemical analysis measures the concentrations of dissolved calcium and phosphate.23
6.2.2 Dietary Erosion
Fruits
The pH of citrus fruits is in the range of 2.0–3.8.4 8 32 76 306 307 Citric acid in fruits has greater capacity to chelate and demineralise enamel than other acids,83 including dietary phosphoric and hydrochloric acids.330 Citric acid with a pH 5.9 is erosive and malic acid with a pH 5.9 is not.210 Chelation is irrelevant at pH 2, but not at a higher pH.17 Most fruits, which have a pH 3–4, contain tannic, tartaric and other organic acids.318 Oranges contain 75% citric and 17% malic acid, apricots 55% citric and 35% malic acid, apples 95% malic acid and 24 other acids, and grapes contain 50% tartaric acid.119 Enamel immersed for 6 minutes in fruit juices was found to show differing “etching” depths:216
- 5.5 µm in apricot juice
- 3 µm in grape juice
- 1 µm in apple juice.
Commercial fruit juices are six to eight times more erosive than homemade juices. Children drinking fruit juices from nursing bottles are easily exposed to acids for 6 minutes or for more time.119
Drinks
The pH of a fluid is exponentially related to its erosive potential.169 Meurman et al. found that the pH of commercial sports drinks was low and all of them dissolved enamel,210 although another study found that dental erosion in athletes (37%) did not differ with the use of different sports drinks.201 Moreover, 85% of a sample of cyclists using sports drinks had palatal erosion.224 The pH of most sport drinks is <4.210
Frequent consumption of wine, cider, acid herbal teas, iced teas with fruit aroma, carbonated beverages, fruit juices and other acidic drinks is associated with erosion.14 74 110 134 152 221 252 317 Carbon dioxide in carbonated beverages forms carbonic acid.78 Ascorbic acid is present in many (sport) drinks and candies.330 Some phosphoric and citric acid is present in nearly all soft drinks, including (<0.1%) colas.257 226 330 The consumption frequency of carbonated drinks and (diluted) fruit juices has been shown to be significantly higher in children with erosion than in those without.239 Frozen fruit juices “on a stick” (Popsicles) are worse, as they are sucked slowly.308 Cider (pH 3–4) seems to be as erosive as orange juice.252 Wines may be very erosive110 and induce a greater loss of tooth minerals than Coca-Cola and apple juice.337
Nowadays, large quantities of soft drinks and fruit juices are consumed, for instance in the UK (160 L/person/year in 1996),234 274 326 and Saudi Arabia.153 The consumption by children and adolescents in particular, has increased in the past 50 years from 2 cans/week to 2 cans/day. Presently, Dutch teenagers consume daily, on average, 7 glasses of acidic drinks, some 100 g of fruit, and 3 glasses of dairy-based drinks.320
The desire for a healthy life influences the diet.192 Slimming and health diets may be dentally unhealthy because the consumption of fruit drinks is encouraged.185 Substitution of sugar-containing drinks by fruit juices fortified with vitamin C prevents caries but makes the teeth vulnerable to erosion.280
Other Food
There are “hidden” acids in commercial food products, masked by sugar. For instance, sweets such as sourballs,286 salad dressings, yoghurts and tomato ketchup are acidic.
In some tribes (Burma) the teeth are coloured black with a tree resin, citron juice and iron (Chapter 10).
Factors Modifying the Effects of Dietary Causes of Erosion
Modifying factors include the amount of drink consumed, frequency and manner of consumption,154 155 dietary constituents, tooth brushing, use of chewing gum, fluoride exposure, the quantity and quality of the saliva, the pellicle and the plaque.123 219 352
Holding a cola-type drink in the mouth for some time rather than swallowing immediately was found to lower the pH to ∼4; a less pronounced pH drop, but lasting for 20 minutes, was recorded with slow sipping. Gulping decreased the pH minimally. The method of drinking strongly determines the risk of erosion.154 A patient with severe erosion had a habit of swirling every swig of cola throughout the mouth. Abandoning the habit arrested the process.127 Swishing acidic drinks (agitation) dissolves more enamel than just holding in the mouth.143 202 238 As elsewhere, wines in Sweden are tested (wine tasting) before they are marketed. Within a few hours 20–50 wines are swirled through the mouth. Sessions take place four to five times/week. In one study, three-quarters of about 20 tasters demonstrated erosion, against 5% of controls.336 Other studies have also shown that frequent wine tasting is associated with extensive erosion.56 110 269
The moment of consumption has an impact: about 60% of a sample of children with severe erosion drank fruit juice just before sleeping, versus about 30% with modest and around 15% with minimal erosion.217 In contrast, tooth wear in the deciduous dentition was not found to be related to the consumption of fruit juices and soft drinks.328
The adherence of different soft drinks to the enamel is different. Coca-Cola and (low sugar) blackcurrant drinks are retained longer on the enamel than distilled water and saliva.143 Desorption of the salivary proteins from the enamel reduces their protective ability and renewal of the protein coat takes time. Sipping is therefore more damaging than gulping.353
Studies have found that contact between the teeth and a cola for 1 hour softened the enamel, which became harder again after 5 minutes of chewing cheese (Cheddar, Edam). However, these findings should be viewed with caution because the chewing action may have resulted in loss of the softened superficial enamel layer. Drinking milk also hardens the enamel. Chewing on paraffin, to stimulate the salivary flow, lacks a beneficial effect.97 98 99
Titrability
The total amount of titrable acid is important.29 32 Titrability signifies the hydrogen ion availability, i.e. the acid’s erosive potential,349 and is a measure of the degree of resistance against a pH rise. Acids that resist buffering act for a longer time and are more destructive than fluids that are quickly neutralised. The buffering capacity of soft drinks has a greater effect than their initial pH.118 The type of acid and temperature are also co-factors. Organic acids are able to maintain the pH below the dissolution threshold for a long time because of their buffering capacity.326
Precipitation of brushite prevents saturation of the eroding solution with dissolved tooth minerals and maintains the enamel dissolution as long as the pH is low.169
Table 6.1 shows the pH of and the amount of the alkaline sodium hydroxide (NaOH) needed to buffer some beverages.188 Other studies have confirmed these values.78 261
Table 6.1 Acidity (pH) and buffering capacity of some beverages188
| Beverage | pH | Volume of base (NaOH, 0.1 N) required for neutralisation (mL) |
| Coca-Cola (fresh) | 2.4 | 3.9 |
| Coca-Cola (old) | 2.4 | 0.9 |
| Isodrink | 3.0 | 10.0 |
| Apple wine | 3.2 | 8.6 |
| Grapefruit juice | 3.2 | 22.0 |
| Rivella Marathon | 3.4 | 4.0 |
| Orange juice | 3.6 | 12.4 |
| Isostar | 3.7 | 4.0 |
| Isostar Light | 3.7 | 4.0 |
Acidic drinks requiring lower amounts of NaOH for buffering are associated with less enamel hardness changes.186 Therefore, “old” Coca-Cola is, despite its lower pH, less erosive than grapefruit juice.188 However, others have found pure fruit juices to be most erosive. Fruit-based carbonated drinks lead to more tooth erosion than carbonated drinks. Plain mineral waters are harmless. Parry et al. found very low dissolution with still and sparkling mineral waters and slightly higher dissolution with carbonated waters, but this was still 100 times less than with orange juice and cola drinks.245 Regular and diet versions of the same drink show fewer differences in erosive potential than different brands.78 “Light” drinks with sugar substitutes may be as erosive as the sugar-containing regular versions,315 but in view of titrability, Diet Coke is less erosive than regular Coca-Cola.257 261
Erosion caused by fruit drinks correlates with pH, but not titratable acidity.354 Fruit particles in citrus drinks enhance the buffering capacity.29 The erosive potential of soft drinks is co-determined by their composition, such as the calcium concentration.186 354 Acidic drinks with calcium and phosphate do not erode if they are supersaturated with respect to apatite.169
Most wines (pH 3–3.6)336 and beers (pH ∼4) are relatively easily neutralised.314 Orange and blackcurrant concentrates used for preparing drinks demonstrated, in a variety of dilutions, higher titrability than citric and hydrochloric acid and were neutralised faster as the dilutions increased.49 Vinegar and salad dressing (pH 3.5) required remarkably large amounts of NaOH for neutralisation.314 Grapefruit and kiwi fruit juice require smaller, yet also substantial, amounts.185 The pH of most vegetables is >5.5. Asparagus, beets, cabbage (sauerkraut) carrots, olives, rhubarb, spinach, and tomatoes are more acidic.318
Table 6.2 shows the weight lost from enamel blocks immersed for 14 days in a range of beverages. Different amounts of enamel was lost in some soft drinks of almost equal pH. Colas caused 50 times higher enamel weight loss/cm2 than tap water; some non-cola drinks dissolved much more mineral. Bearing in mind the salivary buffering capacity, this 14-day immersion period can be considered equivalent to 13 years of normal soft drink consumption.326
Table 6.2 Acidity (pH) of some soft drinks and the loss in weight of enamel from enamel blocks immersed in the beverages for 14 days (% and mg/cm2)326
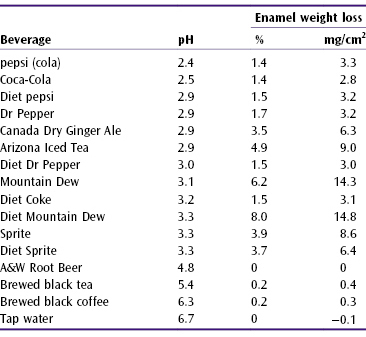
Contact with acids does not have to last long to soften the enamel, but the contact time differs by acid, for instance lemons erode faster than grapefruit.4 Erosion does not require very frequent contact with acids. A comparison of patients with and without erosion showed the risk to be increased:
- 37 times when citrus fruits were consumed more than twice daily
- 10 times when apple vinegar was used weekly
- 4 times when soft drinks were consumed daily
- 4 times when sport drinks were consumed weekly.149
Dietary Groups at Risk
- People with diabetes consuming large amounts of fruit juices are at higher risk.86
- Antihypertension (and other) medicaments cause xerostomia,325 which increases consumption of acidic sweets (and possibly acidic foods) and acidic drinks.29
- Some sports, occupations and environments leading to dehydration promote the consumption of (sports) drinks in large quantities. Although intended for athletes for rehydration, electrolyte replacement and energy replenishment, children and adolescents consume these drinks as well. Moreover, dehydration through strenuous exercise reduces the salivary flow and buffering capacity.349
- Ecstasy use combined with vigorous dancing results in life-threatening hyperthermia. Xerostomia is exacerbated by dehydration. Thirst is relieved by soft drinks, but the sugar enhances drug absorption.72
- Cocaine causes erosion.158 164
Cocaine is synthesised from coca paste (Bazooka) and contains hydrochloride, which dissolves in the saliva and lowers the pH to 4.5.164 The occlusal and buccal surfaces of cocaine addicts are glass-like and smooth.34 Regular application of cocaine to the buccal gingiva results in necrotising ulcerative periodontitis and erosions.158 Nasal insufflation erodes the whole dentition.164 Cocaine introduces the risk of cardiovascular complications with local anaesthetics and retraction cords, which requires postponing the dental treatment for 6–24 hours after application of the cord.34
Medicaments
Acidic medicines are erosive, for instance iron medications taken for longer periods,147 regular chewing of aspirin44 105 298 and vitamin C,63 and hydrogen peroxide rinses.
Enamel placed in a 1% acetylsalicylic acid solution (pH 2.40) became rough within 30 seconds and the superficial enamel disappeared within 15 minutes.126 Chewing aspirin tablets, 90–130 mg/day/kg body weight, for juvenile rheumatoid arthritis (250 000 children in the USA) causes severe occlusal erosion; immediate swallowing of the tablets does not.298 If not fully swallowed, aspirin taken just before sleeping causes the formation of a mucosal white slough that gradually rubs off, leaving red, ulcerated and painful lesions.199
Dissolution of 500 mg vitamin C in saliva lowers the pH to 4.4, lasting 5 minutes or longer.129
Hydrochloric acid medications for achlorhydria (in patients with lack of gastric acid production) are erosive7 200 (but non-erosive substitutes exist293), as was the acid formerly used to dissolve small renal calculi.141
Studies have found that children with asthma had more erosion than controls,7 203 272 273 and explanations included more frequent vomiting,7 279 heartburn,7 reduced salivary flow after prolonged use of medicaments, altered salivary composition, and pH of the medication,35 203 273 although the latter seems unlikely.305 Asthma medicaments are smooth muscle relaxants that act on the oesophageal sphincter and are associated with gastro-oesophageal reflux and xerostomia in the long term. The drugs may promote consumption of acidic drinks.273
Dry powder inhalers have a pH of 5.06 and pressurised metered inhalers a pH of 6.45; the titrability of the former is twice that of pressurised inhalers, but Tootla et al. found that neither the salivary nor the plaque pH dropped below 6 with their use.305 Other studies have shown that the (un)stimulated salivary pH and buffering capacity of children with erosion is lower than in controls,238 as is the case in children using asthma medicines, who also secrete less salivary proteins.260 However, these findings have not been substantiated.7
Mouthcare products are not medicaments.349 The pH of some mouthrinses used against halitosis (offensive breath) is <5.5, but neutralisation requires only a small amount of NaOH. The alcohol content varies from 0% to 27%.30 In vitro, alcohol is cytotoxic to the fibroblasts.296 Frequent rinsing introduces the risk of oral cancer30 91 and softened the surface of composite restorations.121 In bedridden patients with insufficient self-care, the mouth is sometimes cleaned with cotton swabs impregnated with citric acid (pH ∼2.7).212
Industrial and Environmental Erosion
Industrial workers are exposed to, for instance, hydrochloric or sulphuric gases in the electrolytic zinc galvanising, battery, fertiliser, munitions and beverage industries.117 The concentration, exposure time and mouth breathing, which is prompted by the fumes, determine the lesion severity.44 193 280 Other factors, such as not using a commercial toothbrush (e.g. using dattun sticks instead), also have an effect.309
Swimmers, especially competition swimmers, swimming in water disinfected with chlorine may exhibit extensive erosions if hydrochloric acid is formed under the influence of light.
In chlorinated (Cl2) water, the disinfectant hydrochlorous acid (HOCl) is formed. Hydrochloric acid (HCl) is an unwanted by-product. Supplementation with calcium or sodium carbonate (Na2CO3) neutralises the acid. Some 40% of a sample of swimmers training more than 2 hours/day were found to exhibit erosion.54
Disinfection with sodium hypochlorite (NaOCl) results in a high pH, at which little HOCl is formed, and which is neutralised with sulphuric acid. At pH >7 the teeth of competitive swimmers are yellow-brown in colour, because the disinfecting chloride (or bromide) products denature the salivary proteins on the surface of the teeth.355 356
An Israeli study found that the pH in swimming pools varied from 3.6 to 7.8. Teeth immersed for 1–2 hours in water with pH 3.6 became etched.90 In the USA, the pH of water of a swimming pool treated with chlorine was 2.7. In the Netherlands, of 3600 samples of swimming pool water, five had a pH <5.5.182
Jockeys occasionally try to lose body weight by frequent vomiting (and use of laxatives): in the last 2 days before a race, up to 10 kg weight may be lost. Severe erosion is a consequence.33
Endogenous Erosion (Perimylolysis)
Regurgitated gastric acid in the mouth is erosive. Vomiting once a week is as erosive as consuming 2 citrus fruits/day. In one-quarter of a sample of erosion patients, gastric juice was the cause of the erosion, in another quarter it was exogenous acids and in half it was endogenous plus exogenous acids.149
In chronic regurgitation, the gastric acid that is regurgitated has a pH 1–1.5. Peristalsis clears the oesophagus, followed by salivary neutralisation. The pH of vomit is ∼3.8. Vomiting caused the extensive lesions shown in Figures 6.3, 6.4 and 6.5.
Figure 6.3 Labial view of the dentition of a p/>
Stay updated, free dental videos. Join our Telegram channel

VIDEdental - Online dental courses


