Composites, Glass Ionomers, and Compomers
Chapter Objectives
On completion of this chapter, the student will be able to:
1. Describe the various types of composite resin restorative materials.
2. Discuss the uses, advantages, and disadvantages of each type of composite resin.
4. Describe how fillers affect the properties of composites.
5. Explain why incremental placement of composite resin is recommended.
6. Describe the factors that determine how long an increment of composite resin should be light-cured.
7. Discuss the procedural differences between direct and indirect composite restorations.
KEY TERMS defined within the chapter
Direct-Placement Esthetic Materials tooth-colored materials that can be placed directly into the cavity preparation without being constructed outside of the mouth first
Composite Resin tooth-colored material composed of an organic resin matrix and inorganic filler particles
Organic Resin (Polymer) Matrix thick liquids made up of two or more organic molecules that form a matrix around filler particles
Inorganic (Silica) Filler Particles fine particles of quartz, silica, or glass that give strength and wear resistance to the material
Silane Coupling Agent a chemical that helps bind the filler particles to the organic matrix
Pigments coloring agents that give composites their color
Self-Cured Composite composite that polymerizes by a chemical reaction when two resins are mixed together
Light-Cured Composite composite that polymerizes when a chemical is activated by light in the blue wave range
Macrofilled Composite an early generation of composite that contained filler particles ranging from 10 to 100 µm
Microfilled Composite composite that contains very small filler particles averaging 0.04 µm in diameter
Hybrid Composite composite that contains both macrofill and microfill particles to obtain the strength of a macrofill and the polishability of a microfill
Flowable Composite a light-cured, low-viscosity composite resin that contains fewer filler particles
Packable Composite a light-cured, highly viscous, heavily filled composite resin for dentists who use a placement technique with composite that is similar to that of amalgam
Glass Ionomer Cements a self-cured, tooth-colored, fluoride-releasing restorative material that bonds to tooth structure without an additional bonding agent
Hybrid (Resin-Modified) Glass Ionomer a glass ionomer to which resin has been added to improve its physical properties
Compomer composite resin that has polyacid, fluoride-releasing groups added
Indirect-Placement Esthetic Materials tooth-colored materials that are used to construct restorations outside of the mouth in the dental laboratory or at chairside on replicas of the prepared teeth. They are later cemented to the teeth
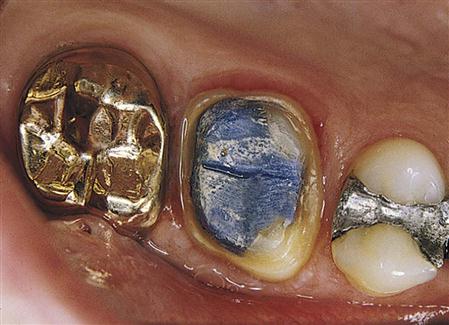
For the first half of the 20th century, amalgam and gold were the primary restorative materials for posterior teeth. Some anterior teeth also had metal restorations that were visible when the patient smiled in the form of gold margins of three-quarter crowns, class III gold foils, or class V gold inlays or amalgams. In the latter half of the 20th century, a variety of direct-placement tooth-colored restorative materials were introduced. Initially, unfilled acrylic resins were used, but they leaked, wore down quickly, and became discolored. Silicate cements were also used, but they were relatively soluble in the mouth and washed out over time. In the 1960s, composite resins were introduced, and they have been continually improved upon ever since by making them more durable, esthetic, and color stable. Other materials such as glass ionomer cements and compomers have also been developed, providing the dental team with a wide selection of esthetic materials for the restoration of carious or damaged teeth and for cosmetic enhancement. Choosing the type of material depends, in part, on the extent of damage to the tooth, the stresses that will be placed on the restoration, and the esthetic requirements of the patient. Because of the superior properties of the other esthetic materials, acrylic resin has been relegated primarily to use for denture bases and teeth (see Chapter 16) and in the fabrication of temporary or provisional restorations (see Chapter 17).
Now, with the capability of bonding restorative materials to tooth structure, advances in esthetic materials and techniques have improved the ability of the dental team to deliver the esthetic results that patients demand. The dental team must keep current with the rapid changes that occur with materials and techniques. Good listening skills are needed to determine the types of esthetic services the patient is requesting so that the dental team and the patient are working in concert toward the same goal. Esthetic materials must be carefully selected so that their properties are compatible with the patient’s oral condition and occlusion. Dental hygienists and dental assistants must understand the properties of these materials, so that as important members of the dental team they can help the dentist to assess the performance of the restorations and can alert the dentist when they perceive that a restoration may be failing. They need to be familiar with the physical properties of the materials so that they do not damage the restorations during routine oral hygiene, coronal polishing, and preventive procedures. Dental assistants need to know the handling characteristics of the esthetic materials so that they can assist the dentist in their placement or can perform steps in their placement as permitted by state dental practice acts.
This chapter describes the physical properties, clinical applications, and shortcomings of directly placed esthetic materials. These materials include composite resins, glass ionomer cements, resin-modified glass ionomer cements, and compomers. Indirectly placed composite resin restorations are discussed as well. Guidelines for selection of the shade of these materials to obtain satisfactory cosmetic results also are discussed.
Composite Resin and Other Direct-Placement Esthetic Restorative Materials
Direct-placement esthetic materials are those that can be placed directly into the cavity preparation or onto the tooth surface by the clinician without first being constructed outside of the mouth. Esthetic materials are those that are tooth colored. The direct-placement esthetic materials used most commonly are (1) composite resin, (2) glass ionomer cement, (3) resin-modified glass ionomer cement (also called hybrid ionomer), and (4) compomer. These are listed in their chronologic order of development.
Composite Resin
A composite is a mixture of two or more materials with properties superior to any single component. Composite resins are tooth-colored materials that are used in both the anterior and posterior parts of the mouth. They are composed mainly of an organic resin (polymer) matrix and inorganic (silica) filler particles joined together by a silane coupling agent that sticks (adheres) the particles to the matrix. Also added are initiators and accelerators that cause the material to set and pigments that give color to the material and match tooth colors. Composite resins are commonly called composites and also can be referred to in the dental literature as resin composites.
Components
Resin Matrix
The most commonly used resin for the matrix of composites is bis-GMA, produced by reacting glycidyl methacrylate with bisphenol-A. Another resin that is used for the composite matrix is urethane dimethacrylate (UDMA). These resins are thick liquids made up of two or more organic molecules called oligomers. To reduce viscosity and allow loading with filler particles, a low molecular weight monomer, TEGDMA, is added. (Monomers are molecules with double carbon bonds that are linked together to form a resin or polymer.)
Filler Particles
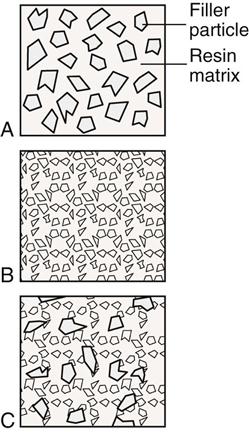
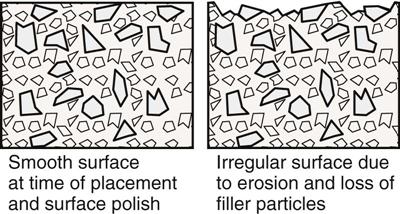
The addition of filler particles makes the organic resin stronger and more wear resistant. Fillers are also added to control the handling characteristics of the composite resin and to reduce the shrinkage that occurs when the resin matrix polymerizes, or sets. The fillers used in composite resins are inorganic silica particles. Silica may be used in crystalline form such as quartz or noncrystalline form such as glass. Fillers may be modified with ions to improve their characteristics. To make the composite resin restoration show up on radiographs (appear radiopaque), ions of barium, boron, zirconium, or yttrium may be added to the filler particles. The most commonly used filler is a modified glass. Important factors for the durability of the composite resin are the size of the filler particles and the ratio or weight of the filler to the matrix. As a general rule, the higher the filler content, the stronger the restoration and the more wear resistant it will be. The amount of filler in a composite resin usually is reported by the manufacturer as the percent of filler by weight (weight %) in the resin. Composites can be classified by the size of the filler particles they contain (Figure 6-1). Wear of the composite is related to the filler particle size, the amount of filler in the resin, and the amount of resin between particles. Large filler particles tend to get pulled (called plucking) from the resin matrix at the surface when the restoration is under function or abraded by food and tooth brushing, resulting in wear of the remaining resin matrix and a rough surface. Smaller particles are not as easily plucked from the resin and therefore cause fewer voids that contribute to wear. Smaller particles can be packed closer together, thereby exposing less of the resin matrix to wear (Figures 6-2 and 6-3) and increasing the number of filler particles that can be added to the resin matrix. The smaller the particles, the smoother the surface of the composite will be after finishing and polishing.
Coupling Agent
To provide a stronger bond between the organic fillers and the resin matrix, a coupling agent is used. This coupling agent is silane, which reacts with the surface of the inorganic filler and with the organic matrix to allow the two to adhere to each other. Good adhesion of the two is necessary to minimize loss of filler particles and to reduce wear.
Pigments
Inorganic pigments are added in varying amounts to develop a variety of colors that approximate the basic colors of teeth.
Polymerization
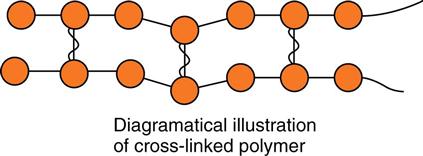
Polymerization is the chemical reaction that occurs when low molecular weight resin molecules called monomers join together to form long-chain, high molecular weight molecules called polymers. For composite resins, activation of the polymerization process can be chemically activated or light activated or a combination of the two (dual-cured). In the polymerization for either method, an activator (chemical or light) causes an initiator molecule to form free radicals (highly charged molecules that have unpaired electrons). The monomers (called dimethacrylates, i.e., bis-GMA) have carbon-to-carbon double bond (C=C) functional groups. The free radicals break one of the carbon-to-carbon double bonds to form a single bond and another free radical. That free radical can cause the same reaction with another monomer to add to the polymer chain (called addition polymerization). As the monomers link together into chains, the volume of resin decreases, so the net result is shrinkage (called polymerization shrinkage). Polymer chains have small groups of atoms hanging off their sides. When side groups of adjacent polymer chains share electrons, they form covalent bonds that link (called cross-linking) the chains together (Figure 6-4). Cross-linking of polymers produces a much stronger, stiffer material than is formed with single-chain polymers. (See Chapter 16 for a more detailed description of polymer formation and properties.)
Chemical Cure
Chemically cured composite resins, or self-cured composite resins, are two-paste systems supplied in jars, syringes, or cartridges. One paste, called the base, contains composite and benzoyl peroxide as an initiator. The other paste, called the catalyst, contains composite and a tertiary amine as an activator. Equal parts of these two pastes are mixed together, and the polymerization reaction begins. The reaction could go to completion very quickly, but chemicals called inhibitors are added to each paste to slow down the reaction. The operator has a limited amount of time (working time—usually about 2 minutes in the mouth) to place the restoration before it becomes too stiff to manipulate. Once the initial set occurs, the material should not be manipulated or the properties of the restoration will be degraded. Disposable mixing sticks are usually supplied with the composite contained in jars or syringes. Because the two pastes must be manually mixed, air can be incorporated into the material, causing voids or porosity in the restoration. Composites in cartridges come with mixing tips that automatically mix the two pastes as they are extruded from the cartridge (see Figure 6-7). This “automixing” greatly reduces the introduction of air into the mixed composite. The composites most commonly found in cartridges are those used as core materials for crowns. The core materials also come in jars or syringes.
Light Cure
Light-cured composite resins are the most common type of composite resin used in private practice. Many clinicians prefer the light-cured composite resin, because it requires no mixing and the operator can control the working time by deciding when to apply the curing light. An intense visible light in the blue wave range activates these materials. Blue light with a wavelength about 470 nanometers (nm) activates an initiator (camphoroquinone) that, in the presence of an accelerator (an organic amine), causes the resin to polymerize. These components are both present in the composite but do not react until the light triggers the reaction. Inhibitors are also present to reduce the effects of the operatory light on a premature setting. However, some manufacturers’ materials are still sensitive to direct operatory light. The operator may choose to turn the operatory light away from the mouth when placing the composite. The ability of the light to cure the composite depends on the accessibility of the composite to the light, the thickness of the composite, the light’s intensity, and the color of the composite. If the composite resin is placed in too thick an increment, the light might not penetrate completely, and the composite may not cure all the way to the bottom. In general, it is recommended that the composite be placed in increments no thicker than 2 mm. Newer, more powerful curing lights might be able to cure greater thicknesses of material. Interproximal areas may need additional time to cure completely because of the more difficult access of the area to the direct path of the light. It is a good practice to cure the interproximal composite restoration again from both facial and lingual surfaces after the metal matrix band is removed to ensure complete curing in the bottom of the box form of the preparation. Darker shades also require a longer curing time, because the light is more readily absorbed by the dark color and does not transmit through the material as readily as through lighter-colored materials.
Dual Cure
Dual-cured composite resins are two-paste systems that contain the initiators and activators of both light-activated and, to a smaller extent, chemically activated materials. The advantage is that when the two pastes are mixed together and placed in the tooth, the curing light is used to initiate the setting reaction, and the chemical setting reaction continues in areas not reached by the light to ensure a complete set. This dual-cure process is very helpful when one is building up an endodontically treated tooth and placing composite core material part way into the canal space. The curing light might not reach the material in the canal, but the composite material will cure chemically on its own.
Classification of Composites by Filler Size
Composite resins have undergone a steady progression in their development to improve their properties. Over time, the filler particle size has become smaller and smaller, the number of filler particles in the resin has increased, and polymerization shrinkage has decreased. As a result, composite restorations have become more durable, leak less, polish better, and match the teeth better. Composites have been classified according to the size of the filler particles they contain.
Macrofilled Composites
The first generation of composite resins used relatively large particles as fillers, ranging in size from 10 to 100 microns (µm). These composites are called macrofilled composites. The large particles make these composites difficult to polish, and they become rough as filler particles are lost at the surface under function or the resin wears, exposing the large particles. They are generally stronger than composites with smaller particles. Because of their roughness and rapid wear, macrofilled composites are no longer widely used. They have been replaced by materials with fillers about 10 µm in size (midi composites).
Microfilled Composites
Microfilled composites were developed to overcome the problems that arose with larger particle size. They became commercially available in the late 1970s. As the name implies, microfilled composites have fillers that are much smaller than those in macrofilled composites. Filler particles average about 0.04 µm in diameter and range in size from 0.03 to 0.5 µm. Several small particles have a larger total surface area than one large particle of similar weight. It is difficult to load a large volume of microfillers in the resin matrix because of this large surface area. Therefore the volume of filler in microfilled composites is only 35% to 50%, as opposed to 70% to 85% with many other composites. A lower filler volume results in a composite with poorer physical properties (i.e., weaker, with greater polymerization shrinkage, and less wear resistant). To help overcome these shortcomings, some manufacturers mix microfillers into a resin, polymerize (cure) it, and grind the hardened material into particles ranging from 10 to 20 µm. Then they use these particles (consisting of pre-polymerized resin and microfillers) as the filler so that they can get more microfillers into the resin and improve its physical properties. Alternative methods to increase the numbers of microfillers that can be loaded into the resin include clumping the microfillers together by heating them or by condensing them into large clumps.
When polished, the microfilled composites produce a very smooth, shiny surface, unlike the rougher macrofilled composites. However, because of their poorer physical properties, they are not suitable for class I, II, and IV (incisal edge repair) restorations.
Hybrid Composites
In the late 1980s, the next generation of composites was introduced. They are called hybrid composites, because they contain both macrofillers and microfillers with filler particles ranging from 0.1 to 3 µm. The combination of the two filler sizes produces a strong composite that polishes well. The filler content is 70% to 80% by weight. They are universal in application in that they can be used well in both the anterior and posterior parts of the mouth.
The hybrids were improved upon by the use of even smaller particles. These hybrids are called microhybrids, because they contain a mixture of small particles (0.5 to 3.0 µm) and microfine particles (0.04 µm). Microhybrids can contain high filler content (70% by volume), because microfine particles fill in spaces between small particles. The next improvement was the introduction of the mini-microhybrids with a particle size of 0.1 to 1 µm. These were rapidly followed by nanohybrids with particle sizes of 0.005 to 0.020 µm. The ability to add increased numbers of filler particles reduces the amount of resin. With less resin, these composites shrink less when polymerized. Shrinkage has been reduced from roughly 3% with earlier composites to less than 1% with some of the nanohybrids. They are strong composites that can be polished to a high shine, and they retain that shine better than earlier composites. Each generation of composite represents some improvement in physical or chemical properties, handling characteristics, polishability, or ability to match the teeth. Research on other methods to improve the properties of the composite resins includes the use of fibers embedded in the resin to reinforce it and the use of crystals to increase strength.
Flowable Composites
Flowable composites are low-viscosity, light-cured resins that may be lightly filled (about 40%) or more heavily filled (up to 70%). Initially, the particle size was in the range of those for hybrid composites. However, nano-sized fillers are being used in the flowable composites also. These composites flow readily and can be delivered directly into cavity preparations by small needle cannulas attached to the syringes in which they are packaged (see Figure 6-7). Because of their low viscosity, they adapt well to cavity walls and flow into microscopic irregularities created by diamonds and burs. They are well suited for use in conservative dentistry (i.e., preventive resin restorations), where they readily flow into the narrow preparations created with small burs and diamonds or air abrasion. Many dentists use them in place of conventional pit and fissure sealants. Because their filler content is higher than that of most lightly filled sealants, they are more wear resistant. They are useful as liners in large cavity preparations because they adapt to the preparation better than more viscous materials such as hybrid and packable composites. Their low elastic modulus allows them to cushion stresses created by polymerization shrinkage or heavy occlusal loads when they are used as an intermediate layer under hybrid and packable composites. (The lower the elastic modulus, the more flexible the material; the higher the elastic modulus, the stiffer the material.) They are useful for restoration of class V noncarious lesions caused by toothbrush abrasion, acid erosion, or occlusal stresses, such as bruxing, that lead to flexing of the tooth (abfraction lesions). For toothbrush abrasion lesions, the patient should have the heavy toothbrushing habits corrected first. Otherwise, the flowable composites may wear too rapidly if the patient continues to brush too hard. The lightly filled flowable composites shrink more when polymerized (about 5% to 7%) than the hybrid composites (<3%), wear more readily, and are weaker. However, the flowable composites too are being improved upon to make them stronger and more durable with less shrinkage.
Pit and Fissure Sealants
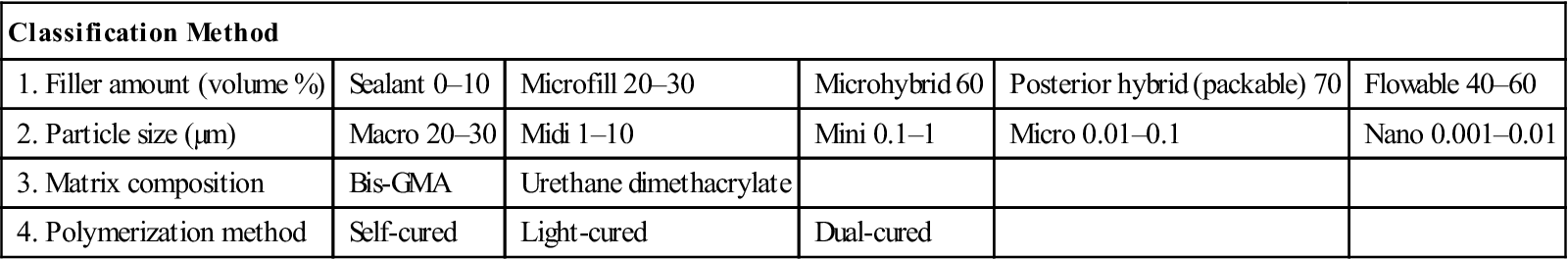
Pit and fissure sealants are low-viscosity resins that vary in their filler content from no filler to more heavily filled resins that are essentially the same as flowable composites. They are used to prevent dental caries in pits and fissures of teeth (see Chapter 7). See Table 6-1 for classification of composites by four different criteria.
TABLE 6-1
Four Classification Methods for Composites
< ?comst?>
| Classification Method | |||||
| 1. Filler amount (volume %) | Sealant 0–10 | Microfill 20–30 | Microhybrid 60 | Posterior hybrid (packable) 70 | Flowable 40–60 |
| 2. Particle size (µm) | Macro 20–30 | Midi 1–10 | Mini 0.1–1 | Micro 0.01–0.1 | Nano 0.001–0.01 |
| 3. Matrix composition | Bis-GMA | Urethane dimethacrylate | |||
| 4. Polymerization method | Self-cured | Light-cured | Dual-cured | ||
< ?comen?>< ?comst1?>
< ?comst1?>
< ?comen1?>
Adapted from Marshall GW, Marshall SJ: Dental composites. In Biomaterials science for restorative dentistry (teaching syllabus), San Francisco, 2000, University of California.
Packable Composites
Packable composites are highly viscous resins that contain a high volume of filler particles (about 70%), which gives them a stiff consistency and makes them less likely to stick to the composite placement instrument. They are used for restoration of posterior teeth in areas of high function (class I and II restorations), because they are stronger and more wear resistant (about 3.5 µm/year) than most hybrids that contain less filler. They are marketed as substitutes for amalgams. Because of their stiffness, they handle more like amalgam than the hybrid composites. They are sometimes referred to as condensable composites, but they cannot truly be condensed (made denser). More correctly they can be “packed” into the cavity preparation. When polymerized, they shrink less than less heavily filled composites because there is less resin and more filler. Common brands include Prodigy Condensable (Kerr Co., Orange, CA), Pyramid (Bisco, Inc., Schaumburg, IL), and Filtex 60 (3M/ESPE, Dental Products, St. Paul, MN).
“Smart” Composites
The idea of restorative materials that would react to the oral environment to combat recurrent caries resulted in the introduction in 1998 of a “smart” composite resin. This composite releases fluoride, calcium, and hydroxyl ions when the acidity of the area around the restoration increases. If bacteria in plaque release acid as a by-product of their metabolism of sugars or cooked starches, the restoration is “smart” enough to release ions that will counteract the effects of the acid and remineralize the tooth. The effectiveness of this material has not been confirmed by clinical studies.
Core Buildup Composites
Core buildup composites are heavily filled composites used in badly broken-down teeth needing crowns. They replace missing tooth structure lost from dental caries or tooth fracture so that there is adequate structure to retain a crown. These composites can be light-cured, self-cured, or dual-cured. They often contain pigments that colorize them so that they can be easily differentiated from natural tooth structure (Figure 6-5). Dentin-colored core materials are used when all-ceramic crowns are to be used. Amalgam would create an esthetically unacceptable dark discoloration under the all-ceramic crown as light passes through the porcelain and reflects off the amalgam. Core composites are strong and can be bonded to tooth structure to minimize bacterial leakage and increase retention. The tooth can be prepared immediately after the composite core is placed and polymerized. The materials are packaged in jars, compules, syringes, syringes with automixing tips, and cartridges with automixing tips similar to impression materials (see Figure 6-7).
Provisional Restorative Composites
Composites for provisional restorations are used in place of acrylic resins for the construction of provisional onlays, crowns, and bridges. They are more expensive than the acrylic resins, but they wear less and shrink less and release less heat as they cure. They can be repaired easily with flowable composites to add to contact areas and margins. They are more brittle than the acrylic resins and tend to break more easily with longer-span bridges (see Chapter 17). Common brands include Protemp Garant (3M/ESPE) and Integrity (Dentsply International, York, PA).
Physical Properties of Composites
Important physical properties of composites include biocompatibility, strength, wear, polymerization shrinkage, thermal conductivity, coefficient of thermal expansion, water sorption, elastic modulus, and radiopacity.
Biocompatibility
Newly placed composite resins can release chemicals that, in deep cavity preparations, could pass through the dentinal tubules into the pulp, causing an inflammatory reaction. When the tubules are sealed by dentin bonding agents or protected with a base or liner, there is no problem. Polished composites are well tolerated by surrounding soft tissues. They are not known to cause any systemic disorder. A very few individuals may be allergic to one or more of the components of the material, and for these individuals another restorative material must be chosen.
Strength
Most of the composites commonly used today are similar in compressive strength. They are not as strong in compression as amalgam but are stronger than glass ionomers.
Wear
Composites wear faster than amalgams. Recent improvements have made the latest generation of composites more wear resistant than early composites, and they are beginning to approach the wear rate of amalgams under normal function. Filler content has an effect on the wear rate. Composites with a lower volume of filler (microfills and flowables) wear faster than more heavily filled materials. Wear can result from abrasion by foods or toothbrushing or by contact with opposing teeth during eating or bruxing. The wear from the o/>
Stay updated, free dental videos. Join our Telegram channel

VIDEdental - Online dental courses