Dental Amalgam
Chapter Objectives
On completion of this chapter, the student will be able to:
1. Discuss the safety of amalgam as a restorative material.
2. List the main components in dental amalgam.
3. Describe the advantages of high-copper amalgams over low-copper amalgams.
4. Describe the role of the gamma-2 phase in corrosion of the amalgam.
6. Define creep, corrosion, and tarnish.
7. Discuss the effect of mixing time on the strength and manipulation of amalgam.
8. Describe the advantages and disadvantages of bonded amalgam restorations.
Key Terms defined within the chapter
Alloy a mixture of two or more metals
Amalgamation reaction that occurs when silver-based alloy is mixed with mercury
Dental Amalgam restorative material composed of silver-based alloy mixed with mercury
Lathe-Cut Alloy irregularly shaped particles formed by shaving fine particles from an alloy ingot
Spherical Alloy alloy particles produced as small spheres
Admixed Alloy mixture of lathe-cut and spherical alloys
Gamma-2 Phase a chemical reaction between tin in the silver-based alloy and mercury that causes corrosion in the amalgam
Tarnish oxidation affecting a thin layer of a metal at its surface. Not as destructive as corrosion
Creep gradual change in the shape of a restoration caused by compression from occlusion or adjacent teeth
Triturator (Amalgamator) mechanical device used to mix the silver-based alloy particles with mercury to produce amalgam
Historically and currently, the most widely used material in restorative dentistry is metal. Dental amalgam is a combination or amalgamation of metals, mostly silver alloy powders and mercury. Its ease of manipulation, good clinical durability, and low cost have made it a popular material that has been used for hundreds of millions of restorations for over a century and a half. Its use has been gradually diminishing in the Western world as more esthetic materials such as composite resin and ceramic restorations have improved in their physical properties and handling characteristics. It is essential that dental clinical personnel have an understanding of the characteristics of the various amalgam alloys, so they can correctly mix, place, and carve them. Additionally, knowledge of good mercury hygiene measures is important for health and safety reasons. Dental assistants and hygienists will be asked questions by patients about the mercury content of amalgams and the health risks. Patients need to be provided with accurate information about this issue. Some states mandate that patients be provided with a dental materials fact sheet listing pros and cons of the materials. This chapter covers the properties and handling of amalgam and mercury hygiene.
Dental Amalgam
In the United States, dental amalgam has been used successfully for the restoration of teeth for over 170 years. Approximately 50 million amalgam restorations are placed each year. Amalgam has been studied and tested more than any other restorative material. Although composite resins are being used with increasing frequency for posterior restorations and are being requested more often by patients instead of amalgam, amalgam is still a widely used direct-placement material for the posterior region of the mouth. No other direct restorative material has the durability, ease of handling, and good physical characteristics of amalgam. Its wear resistance and compressive strength are superior to composite resin and glass ionomer cement. Clinical studies indicate a typical life expectancy of about 15 years for class I and II amalgams. Many can last much longer, with some amalgams documented as lasting 30 to 40 years. The safety of amalgam has been called into question in recent years, but a study conducted by the National Institutes of Health (NIH) from 1991 to 1993 concluded that amalgam is safe for human use. Additionally, the U.S. Public Health Service, the Food and Drug Administration (FDA), the Centers for Disease Control and Prevention, and the World Health Organization all consider amalgam to be a safe material. Less than 0.01% of people have an adverse reaction to components of the amalgam. However, a combination of concerns by patients about its safety and its lack of esthetics has prompted many patients to request tooth-colored restorative materials. Insurance carriers have seen a reduction in the use of amalgam for posterior restorations by approximately 40% in the past 15 years.
Alloy Versus Amalgam
An alloy is a mixture of two or more metals. The alloy used to produce dental amalgam is predominantly composed of silver but also contains copper and tin. A variety of other metals, such as palladium, indium, or zinc, may be added in much smaller quantities to produce specific properties in the alloy. When the silver-based alloy is mixed with mercury, a liquid metal, the reaction that occurs is called amalgamation and the material that is produced is called dental amalgam.
Silver-Based Amalgam Alloy Particles
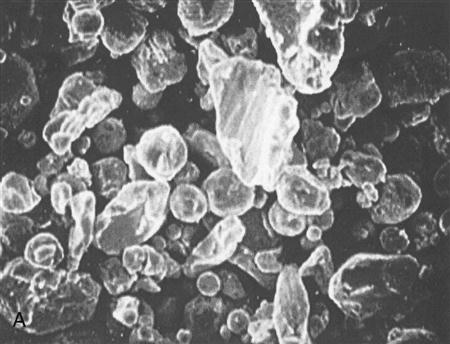
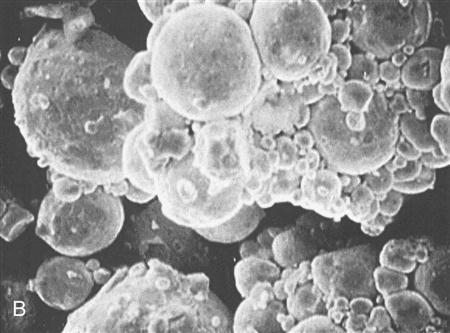
Silver-based amalgam alloys are classed according to the shape of the particles in the powder as irregular, admixed, or spherical (Figure 9-1). Each of these particle shapes contributes certain handling characteristics to the amalgam, and to some degree the amalgam type is selected by the dentist according to these characteristics. Irregularly shaped particles are formed by shaving fine particles off an ingot of the alloy with a lathe (lathe-cut alloy). The particles are sifted to separate them into fine and ultrafine particles. Spherical particles are produced by spraying molten alloy into an inert gas (spherical alloy). Spherical particles are formed as the atomized droplets cool. Admixed particles consist of a mixture of lathe-cut and spherical particles (admixed alloy).
Composition
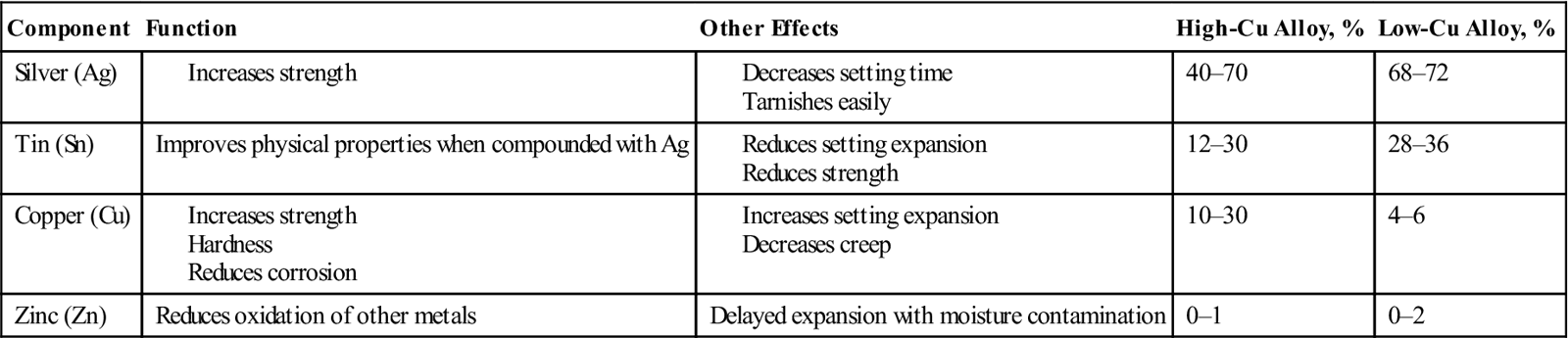
Modern dental alloys are considered to be high copper in content (13% to 30%) compared with their predecessors, which had 4% to 6% copper by weight (Table 9-1). They generally contain 40% to 70% silver and 12% to 30% tin. They are mixed with mercury 43% to 50% by weight. Spherical alloys require less mercury to wet the particles and generally set faster than lathe-cut particles. Manufacturers may also add indium (1% to 4%), palladium (0.5%), and zinc (0.01% to 2%). Zinc may inhibit corrosion by reducing the oxidation of the other metals in the amalgam. In the low-copper amalgams, zinc was responsible for gradual expansion of the amalgam over time (delayed expansion) when moisture contamination was present during placement. Contact of moisture with the zinc caused the formation of hydrogen gas within the amalgam, which caused it to expand. Delayed expansion could cause the restoration to expand beyond the cavity walls, causing cracking in the adjacent enamel. Because of this undesirable property, most high-copper alloys contain no zinc or very low levels of zinc and are called zinc-free alloys.
TABLE 9-1
Main Components of Amalgam Alloy
< ?comst?>
< ?comen?>< ?comst1?>
< ?comst1?>
< ?comen1?>
Setting Transformation
When the alloy in powder form is mixed with the liquid mercury, a chemical reaction occurs. The mixture has a putty-like consistency that can be packed into the cavity preparation. Over the next several minutes, it gradually becomes firmer. During the first part of this firming phase, the amalgam can be carved (during the working time or time available to manipulate the amalgam) to the anatomic shape of the tooth. Once it reaches its initial set, it can no longer be carved and is firm but is not fully reacted. It is relatively brittle at this point, and the patient is advised not to bite on it for several hours. Many of the high-copper spherical amalgams gain approximately 50% of their compressive strength in the first hour, but it takes up to 24 hours for most amalgams to gain their maximum strength. Once fully set, they are hard, strong, durable restorations.
Setting Reactions
The chemical reaction that occurs when the alloy and mercury are mixed has three phases. The first phase, called the gamma phase (γ), is the silver alloy phase. It is the strongest phase and has the least corrosion. The second phase is the gamma-1 phase (γ1) consisting of mercury reacting with the silver. It is strong and corrosion resistant, although not as resistant as the gamma phase. The third phase, the gamma-2 phase (γ2), consists of the reaction of mercury with tin. Gamma-2 is weak and corrodes readily. Tin is used to control the rate of set of the amalgam. Both silver and tin dissolve into the liquid mercury until the solution becomes saturated with them, and they also absorb mercury. Newly formed particles begin to precipitate (crystallize) out of the mercury until there is no more mercury left to react. This process may take up to 24 hours to go to completion. The low-copper amalgams had much more corrosion because of the chemical reaction of tin and mercury (gamma-2 phase). Copper reacts with the tin to keep it from being available for the gamma-2 phase. High-copper amalgams do not have a gamma-2 phase and are superior in their clinical performance, displaying reduced corrosion, higher strength, and margins that hold up better.
Tarnish
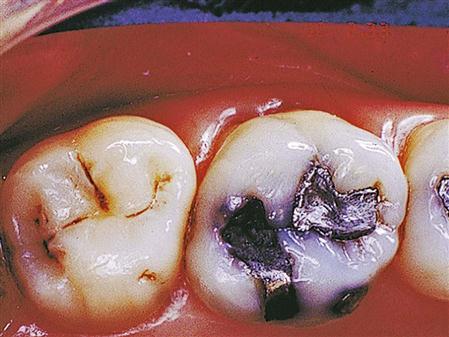
Tarnish is an oxidation that attacks the surface of the amalgam and extends slightly below the surface. It results from contact with oxygen, chlorides, and sulfides in the mouth. It causes a dark, dull appearance, but it is not very destructive to the amalgam (Figure 9-2). The rougher the surface, the more it tends to tarnish. Metals such as palladium are sometimes added to help reduce the tarnish. Polishing of the restoration can also reduce the tarnish. Polishing of amalgams is best done after the restoration has set for a period of 24 hours or longer. Some clinicians have advocated polishing fast-set amalgams in as little as 20 minutes after placement. However, amalgams polished this soon after placement usually do not achieve a high shine. High-copper amalgams have a smoother surface after carving than low-copper amalgams and tend to tarnish less. Polishing is not as critical to their longevity as with low-copper amalgams. Because of this fact, controversy exists among dental educators and clinicians as to whether high-copper amalgams need polishing if they are well carved and contoured at the time of placement. Generation of excessive heat during polishing can cause a release of mercury from the silver-mercury phase that results in a mercury-rich surface that will corrode more readily and deteriorate at the margins.
Corrosion
Corrosion can occur from a chemical reaction between the amalgam and substances in saliva or food, resulting in oxidation of the amalgam. It can also occur when two dissimilar metals interact in a solution containing electrolytes (saliva is such a solution). An electrical current is generated between the metals (much like a battery) in a process called galvanism. The result of the galvanic reaction is oxidation of one of the metals. This oxidation is responsible for the corrosion of the amalgam. Corrosion also takes place within the amalgam through interaction of its metal components. It weakens the amalgam over time, can stain surrounding tooth structure as corrosion products enter the dentinal tubules, and can lead to deterioration of the margins (Figure 9-2). The high copper content of newer alloys eliminates the formation of the gamma-2 reaction product that caused weakening of the amalgam. High-copper alloys have virtually replaced low-copper alloys, because high-copper amalgams are more durable with less deterioration at the margins (better marginal integrity), less corrosion, and higher strength.
Clinically, a galvanic reaction may occur when a newly placed amalgam contacts another metal restoration such as a gold crown. The patient feels a mild electrical shock and may experience a metallic taste. This problem may persist until the amalgam completes its setting reactions, until enough oxides build up on one of the metals to stop the electrical current, or until the offending restoration is replaced with a nonconducting restoration such as composite or with a restoration of a metal similar to the one next to it. Corrosion, however, can occur within an amalgam without the patient ever being aware of the process.
Creep
Creep in dental amalgams refers to the gradual change in shape of the restoration from compression by the opposing dentition during chewing or by pressure from adjacent teeth. It was a phenomenon associated with the gamma-2 phase seen with low-copper alloys (about 6%) and resulted in deterioration of the margins. High-copper alloys exhibit far less creep (less than 0.5%) and have superior marginal integrity.
Dimensional Change
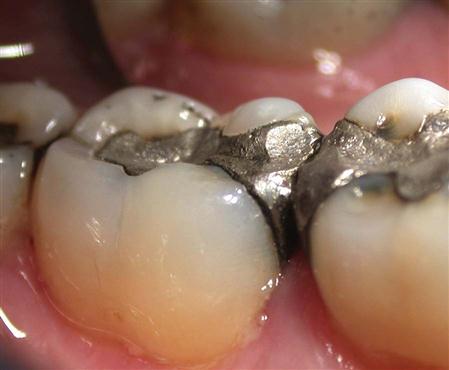
Ideally, the dimensions of a newly placed amalgam should not change. If it contracts excessively, it will open gaps at the margins, contributing to leakage of fluids and bacteria and causing sensitivity. If it expands excessively, it can put pressure on the cusps and cause pain with biting pressure or may result in fracture of the cusps. Some expansion and contraction occur during the setting reaction of the amalgam. It is the net effect of these two processes that is important. The composition of the alloy particles, the ratio of the mercury to alloy powder by weight, and salivary/moisture contamination are other factors that contribute to dimensional changes. The low-copper amalgam, especially those containing tin, were prone to expansion over time if they were exposed to moisture during placement. This gradual expansion after placement is called delayed expansion (Figure 9-3).
Strength
Amalgam is among the strongest of the directly placed restorative materials. It has the ability to resist the strong forces of the bite repeatedly over many years when it is properly placed. Amalgams are stronger in compression (approximately 400 to 450 MPa) than composites or glass ionomers. However, they are relatively weak in tension (about 12% of compressive strength) and shear and therefore require adequate bulk to resist breaking. If thin excesses of amalgam are left over the cavosurface margins, they will chip away over time, creating an irregular margin that tends to collect plaque and contribute to recurrent caries. Generally, high-copper amalgams have a higher early compressive strength (1 hour) than low-copper amalgams. This is advantageous because it helps resist breakage if the patient inadvertently bites on a newly placed amalgam. Some high-copper amalgams gain approximately 80% of their strength in the first 8 hours. Low- and high-copper amalgams are comparable in compressive strength once they have completely set at about 24 hours. See Table 9-2 for American National Standards Institute/American Dental A/>
Stay updated, free dental videos. Join our Telegram channel

VIDEdental - Online dental courses