Chapter 5
Regenerative Periodontal Techniques
Aim
This chapter aims to provide a historical overview of the principles underlying guided periodontal tissue regeneration (GTR) and to illustrate the different surgical techniques that may be employed to regenerate lost periodontal tissues. It will allude to the limited evidence base for efficacy of some of these techniques and also demonstrate the core principles, which may also be applied to guided bone regeneration (GBR).
Outcome
Having read this chapter, the reader should be familiar with the biological basis for, and principles underlying, GTR and, most importantly, be conversant with their limitations, indications and contraindications.
Introduction
Immediately following non-surgical or surgical RSD, the pocket space and connective tissues lying adjacent to the root surface are occupied by a thin blood clot. The principles of wound healing follow those for secondary intention healing, in that the clot initially “organises” and is then replaced by fibrous repair tissue. The pioneering research of Tony Melcher in the 1960s and 1970s led to the classic paradigm that “the first cells to repopulate a root surface will dictate the nature and quality of tissue that forms there” (Melcher, 1976). Of those cell types that have the potential to repopulate the postoperative wound, the fastest dividing cells are the oral epithelial cells (Fig 5-1), and hence the majority of new root surface attachment formed is epithelial in nature – the aptly named long junctional epithelium (LJE) (Caton et al., 1980).
The classical biological processes that contribute to pocket-depth reduction post-therapy are:
-
LJE formation (one week)
-
Resolution of gingival inflammation (two weeks)
-
Remodelling of the gingival connective tissues and subsequent shrinkage owing to collagen maturation by cross-linkage (two to three months).
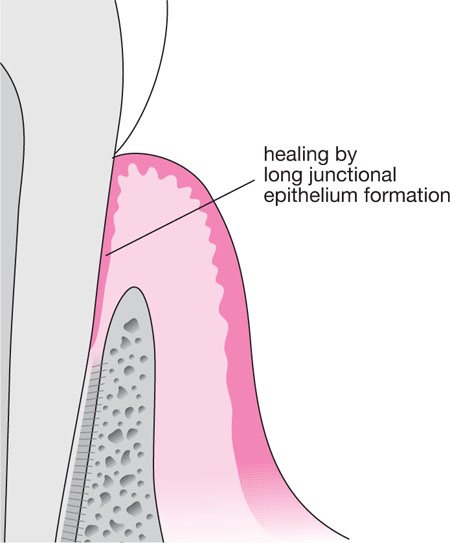
Fig 5-1 Longitudinal section of treated pocket, demonstrating formation of a new long junctional epithelial attachment to the treated root surface.
Hence, reprobing within three months of therapy is generally not recommended. Despite the LJE formation, Rosling et al. (1976) demonstrated that new bone will also form at the base of infrabony defects (Fig 5-2a,b).
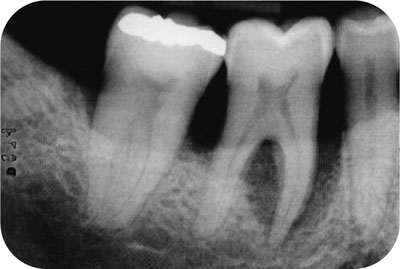
Fig 5-2a Mandibular molar tooth with furcation involvement and bone loss close to the apex of the mesial root pre-non-surgical RSD.
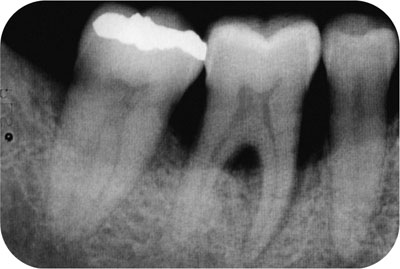
Fig 5-2b The same tooth as in Fig 5-2a, 12 months following non-surgical RSD. Note the bony infill at the base of the vertical bone defect (whether an epithelial layer exists between the root surface and the new bone in this case is unknown).
Despite this, histological studies have demonstrated that epithelium may be interposed between the new alveolar bone and root surface in such cases (Caton and Greenstein, 1993). Classical periodontal regeneration, however, requires the formulation of a new connective tissue attachment to the root surface, achieved by selectively guiding undifferentiated stem cells from the periodontal ligament and adjacent alveolar bone to repopulate a treated root surface. This process should lead to new cementum formation, followed by new periodontal ligament and adjacent alveolar bone formation. True periodontal regeneration should also recreate the overlying gingival epithelium and connective tissues; this is, however, not yet technically possible. This latter failure is crucial to understanding the limitations of contemporary GTR, because complete periodontal regeneration is rarely achieved and thus recession is a component of healing even post-GTR in the “real world”. It is important that patients are advised of this as part of the informed consent process.
Whilst the LJE is relatively stable, it is far less robust than the original periodontal connective tissue attachment and hence supportive care programmes involving regular (3–4 monthly) recalls for professional reinforcement of oral hygiene and professional prophylaxis remain the mainstay of successful traditional periodontal therapies. The ultimate goal of periodontal therapy is to replace the supporting apparatus that has been destroyed by the disease process with cells and tissues identical to those that were lost – this is true regeneration.
Terminology
Repair – is the restoration of new tissue that does not replicate the structure and function of the lost tissues (Melcher, 1969).
Regeneration – is the biological process by which the architecture and function of the lost tissue is completely restored (Melcher, 1969).
Connective tissue re-attachment – is the reunion of connective tissue with a root surface on which a viable periodontal ligament exists, for example following a surgical flap for peri-radicular surgery where there was no underlying periodontal disease (Isidor, 1985).
Connective tissue new attachment – is the union of connective tissue with a root surface that has been deprived of its periodontal ligament (Isidor, 1985).
It is important to appreciate that a new attachment may be formed by epithelium (new epithelial attachment) or by connective tissue (new connective tissue attachment) and it is the latter that GTR aims to achieve.
Biological Principles Underpinning GTR
The dentogingival anatomy immediately post-therapy, with a blood clot sitting adjacent to the debrided root surface, is illustrated in Fig 5-3. The natural history of healing is that the oral epithelial cells migrate onto the root surface and attach via hemi-desmosomal junctions to form the LJE. If the epithelial down-growth is blocked using a physical barrier then the next cell type to reach the root surface would be the gingival fibroblasts and the connective tissues they form. The gingival connective tissues are dynamic, and collagen replacement is affected by collagenase production by gingival fibroblasts. In areas where the RSD has denuded the root surface of cementum, the liberation of collagenases may resorb the root dentine organic matrix, thus creating external root resorption (Fig 5-4). This is undesirable; therefore any barrier membrane should also isolate the cleaned root surface from the gingival connective tissues, thereby leaving two remaining potential sources of cells to re-populate the wound:
-
osteoblasts from the adjacent bone
-
undifferentiated stem cells (derived from the embryonal neural crest) within the periodontal ligament (PDL).
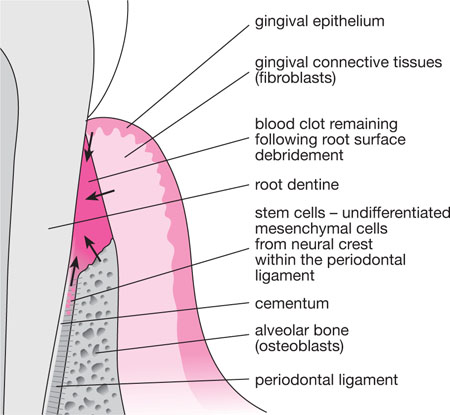
Fig 5-3 Longitudinal section of treated pocket, demonstrating (by arrows) the cells with the potential to colonise the root surface. The oral epithelial cells are the first to reach the root surface, forming a new long junctional epithelial attachment at the coronal aspect.
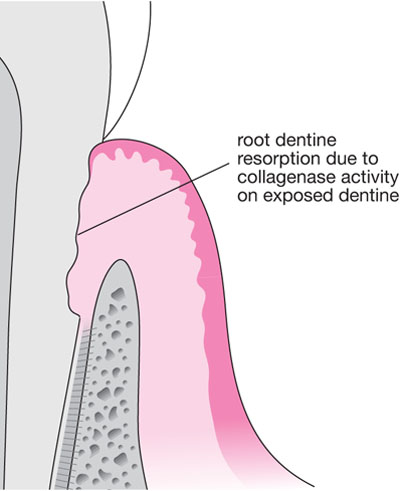
Fig 5-4 If the oral epithelium is physically blocked from reaching the treated root surface, the next cells to contact are those of the gingival connective tissues. Collagenases produced by gingival fibroblasts were shown in animal models to resorb the root surface where denuded of cementum by instrumentation.
Osteoclasts are more slowly dividing than the PDL stem cells; therefore it is largely academic to speculate as to what their contact with the root surface would achieve. In experimental models it was demonstrated that the collagenases produced by osteocytes/clasts within bone resorbed the root dentine (as with the gingival connective tissues), and since bone is a dynamic and vital tissue, bony ingress to the resorbed dentine spaces arose. The result was ankylosis (Fig 5-5), an undesirable outcome for natural teeth, but a very desirable one for titanium implants, where the term “osseointegration” is now employed to describe the resulting tight union between bone and the implant surface.
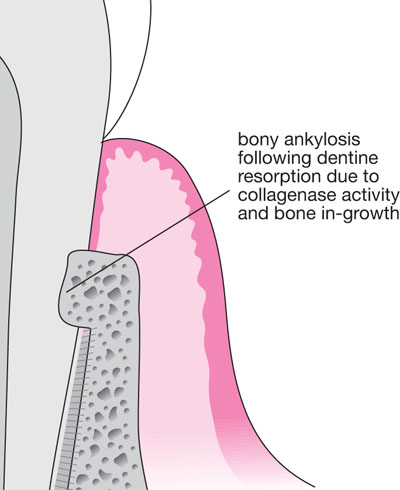
Fig 5-5 In animal models, where bone was allowed to reach the root surface, resorption also took place (as with Fig 5-4), but new bone formed into resorption lacunae, creating an ankylosis.
The potential for periodontal tissue regeneration was demonstrated in a series of elegant histological studies (Nyman et al., 1982). Following surgical flap procedures, a buccal and approximal bone window was created; all exposed cementum was then curetted prior to placement of a 0.22 mm Millipore filter over the buccal fenestration and flap closure. Six months later, new cementum with inserting connective tissue fibres had formed, alongside significant (up to 3.9 mm) bone growth. A further study utilised Millipore and Goretex filters placed over surgically induced defects, where cementum was denuded from non-periodontitis affected roots (Gottlow et al, 1984). Six months later, and after exposure to long-term plaque accumulation, block dissected biopsies demonstrated new cementum formation with new PDL fibre insertion. Nyman and colleagues (1982) also demonstrated new cementum and bone formation around a human mandibular incisor with advanced periodontitis in a 47-year-old male, using a Millipore filter and the now standard surgically induced root surface notch as a reference point from which to measure new connective tissue attachment.
Subsequent studies (e.g. Gottlow et al., 1986) demonstrated that the surgical placement of a barrier membrane made from Goretex (expanded polytetrafluoroethylene – ePTFE) fibres tied around the cervical margin of the tooth and draped beneath a mucoperiosteal flap and onto the alveolar bone surface (Fig 5-6) was capable of preventing both the down-growth of oral epithelial cells and the trans-growth of gingival fibroblasts onto the cleaned root surface. The result was that undifferentiated stem cells from the PDL migrated into the wound, and sequential differentiation into cementoblasts and fibroblasts led to the formation of new cementum and new PDL fibres inserting into new alveolar bone (Fig 5-7). These studies established the principles of periodontal regeneration and verified Melcher’s theories; GTR was born, and the benefits of stem cell biology were realised for the first time in periodontology.
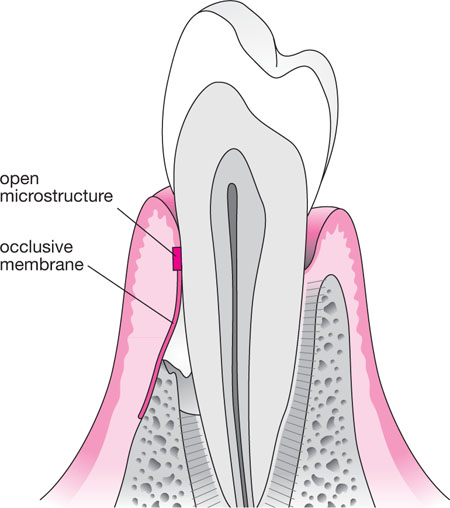
Fig 5-6 The placement of an ePTFE membrane. The open micropore surface is designed to slow down the migration of large (relative to fibroblasts) epithelial cells. The occlusive “skirt” draped over the alveolus had a smaller micropore structure designed to prevent slender gingival fibroblasts from traversing the membrane onto the root surface.
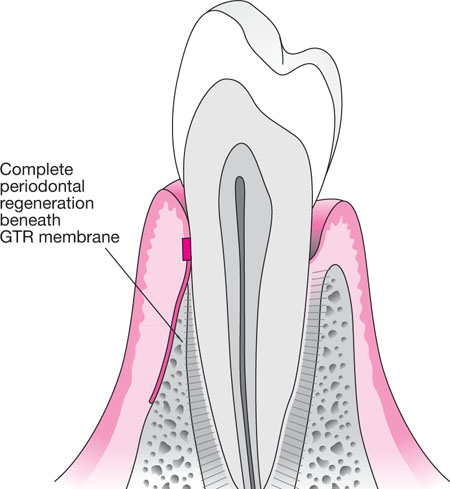
Fig 5-7 An illustration of “theoretical” complete regeneration following Goretex membrane placement. In reality at six weeks, when membranes were removed, the regenerated tissue was a red gelatinous tissue which did not resemble architecturally the definitive regenerated structures: this process takes approximately 12 months.
Surgical Planning for Membrane GTR
Indications
-
Most Class II furcations.
-
Early Class III furcations.
-
Two- or three-walled bony defects.
-
Circumferential “moat defects”.
Contraindications
-
Poor oral hygiene – failure more likely.
-
Patient attitude, e.g. expectations, compliance – technically challenging procedures.
-
Economics – expensive.
-
Multiple infrabony defects – cost, time and logistics.
-
Class III furcation defects – owing to a failure of coronal bone growth.
-
One-walled bony defects – owing to a lack of vertical bony walls to supply osteoblasts.
-
Horizontal bone loss – coronal bone growth did not occur.
-
Lack of soft tissue to cover space coronally – membrane exposure, plaque accumulation and recession.
-
Smoking.
-
Medical history.
What Went Wrong in Practice and Why?
The majority of membranes became exposed between the first surgical stage and their surgical removal six weeks later. Many claimed this did not arise in their cases, but those with experience recognised that this was either “luck” or due to the fact that those operators had not performed a sufficient number of procedures to justify authoritative comment! Exposure was simply because the ePTFE membranes were literally “non-stick” and there was no initial fibrinous union between the adjacent flap and membrane surface; thus muscular pull from the circum-oral musculature drew the flap apically and exposed membranes, resulting in recession. Other complications included:
-
any complications for routine flap surgery
-
failure of procedure
-
additional therapy needed, so costly
-
infection/abscess formation
-
exfoliation of graft/membrane
-
Stay updated, free dental videos. Join our Telegram channel

VIDEdental - Online dental courses


