Chapter 5 Pathogenesis of microbial disease
General aspects of infection
Communicable diseases
Natural history of infectious disease
An acute infection generally progresses through four stages:
Pathogenesis of bacterial disease
Determinants of bacterial pathogenicity
Transmission
There are four important portals (or gates) of entry of pathogens (Table 5.1):
| Portal of entry | Pathogen | Disease |
|---|---|---|
| Skin | Clostridium tetani | Tetanus |
| Hepatitis B virus | Hepatitis B | |
| Respiratory tract | Streptococcus pneumoniae | Pneumonia |
| Neisseria meningitidis | Meningitis | |
| Haemophilus influenzae | Meningitis | |
| Mycobacterium tuberculosis | Tuberculosis | |
| Influenza virus | Influenza | |
| Rhinovirus | Common cold | |
| Epstein–Barr virus | Infectious mononucleosis | |
| Gastrointestinal tract | Shigella dysenteriae | Dysentery |
| Salmonella typhi | Typhoid fever | |
| Vibrio cholerae | Cholera | |
| Hepatitis A virus | Infectious hepatitis | |
| Poliovirus | Poliomyelitis | |
| Genital tract | Neisseria gonorrhoeae | Gonorrhoea |
| Treponema pallidum | Syphilis | |
| Human immunodeficiency virus (HIV) | Acquired immune deficiency syndrome (AIDS) | |
| Candida albicans (fungus) | Vaginitis |
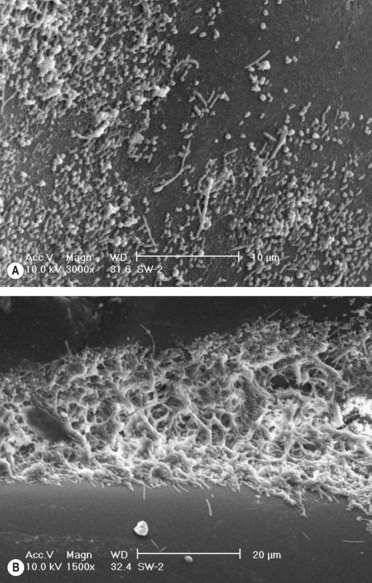
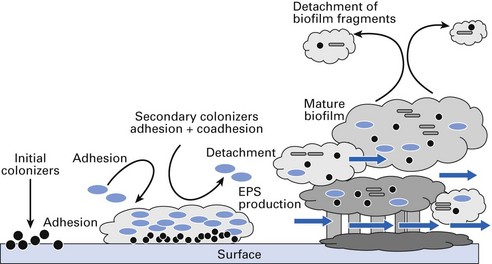
Biofilm formation
As mentioned, biofilms are intelligent communities. Structurally, they are not flat and compressed but comprise a complex architecture with towers and mushroom or dome-shaped structures with water channels that permit transport of metabolites and nutrients (Figs 5.1–5.3). Bacteria in biofilms maintain the population level by constantly secreting low levels of chemicals called quorum-sensing molecules (e.g. homoserine lactone), which tend to repulse incoming bacteria or activate the communal bacteria to seek new abodes. Further, specific gene activation may lead to production of virulence factors or reduction in metabolic activity (especially those living deep within the matrix).
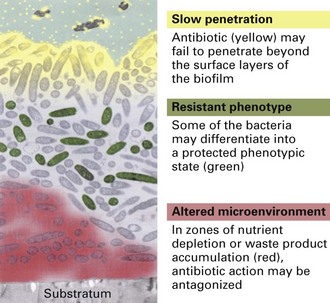
It is now known that infections associated with biofilms are difficult to eradicate as sessile organisms in biofilms exhibit higher resistance to antimicrobials than their free-living or planktonic counterparts. The reasons for this appear to be (Fig. 5.4):
Invasiveness
Table 5.2 Examples of surface virulence factors that interfere with host defences
| Organism | Virulence factor | Used in vaccine |
|---|---|---|
| Bacteria | ||
| Streptococcus pneumoniae | Polysaccharide capsule | Yes |
| Streptococcus pyogenes | M protein | No |
| Staphylococcus aureus | Protein A | No |
| Neisseria meningitidis | Polysaccharide capsule | Yes |
| Haemophilus influenzae | Polysaccharide capsule | Yes |
| Klebsiella pneumoniae | Polysaccharide capsule | No |
| Escherichia coli | Protein pili | No |
| Salmonella typhi | Polysaccharide capsule | No |
| Mycobacterium tuberculosis | Mycolic acid cell wall | No |
| Fungi | ||
| Cryptococcus neoformans | Capsule | No |
Stay updated, free dental videos. Join our Telegram channel

VIDEdental - Online dental courses