Calculus and Other Disease-Associated Factors
• Describe the role of dental calculus and other disease-associated factors in the initiation and perpetuation of gingival and periodontal disease.
• Describe the formation and attachment of supragingival and subgingival calculus in the oral environment.
• Describe the distribution of calculus deposits.
• Compare the composition, distribution, and attachment of supragingival and subgingival calculus.
• Explain how anticalculus agents work in reducing calculus formations in humans.
• List the variety of factors that are linked to periodontal disease.
• Describe hygienic restorations.
• Explain the role of the dental hygienist in the recognition and provision of care for patients with disease-associated factors.
A thorough understanding of calculus, what it is and why it is important, places calculus removal in perspective in dental hygiene practice. A good deal of practice time is spent removing calculus and preventing its formation. Calculus removal has an important place in dental hygiene care because its presence tends to accelerate the progression of disease (frequently in localized areas), and it is a readily modifiable factor associated with improved periodontal health.1 In addition, other irritating factors related to tooth placement and tooth replacement can also present complications in dental hygiene care by providing areas where plaque biofilms accumulate and are hard to control. These factors are very important in dental hygiene practice, and dental hygienists spend much of their work time addressing them.
Calculus
Tartar
Tartar is the common name, often used by patients, for dental calculus. Although introduced in the sixteenth century, and wrongly attributed with causation of malady,2 the use of the term is likely to continue because it is often used in advertising. Products referred to as tartar control toothpaste and rinses are popular and reinforce the use of this term.
Significance
Calculus is formed by the deposition of calcium and phosphate salts present in bacterial plaque. It has long been thought to be the cause of periodontal diseases due to its association with gingival infections and the improved gingival health observed following its removal. A thoughtful review by Mandel and Gaffar3 reported that 11% of sites with calculus also had gingivitis, whereas 75% of tooth surfaces with plaque had gingivitis. This finding suggested that calculus may be a result of disease rather than the cause and that more strongly identified plaque bacteria biofilm is a causative factor.
Calculus does contribute to the development of disease, serving as a reservoir for bacterial plaque biofilm, the etiologic agent. Calculus has been shown to have nonmineralized areas appearing microscopically as channels that contain bacteria and other debris.4 Colonies of bacteria inside the calculus are impossible to remove by any oral hygiene procedure and provide sheltered areas in periodontal pockets that keep plaque in close proximity to the tissues.
It is also clear that even in young people, the presence of calculus is associated with increased levels of gingival disease. A study of Thai children aged 11 to 13 years showed a significant association among gingivitis, plaque status, and calculus accumulation, but no association between calculus status and caries.5 An evaluation of data from 1285 young people aged 13 to 20 years who participated in the 1986-1987 national survey of the oral health of U.S. adolescents indicated that subgingival calculus is associated with both attachment loss and aggressive forms of periodontitis. The young people with measurable attachment loss had significantly more gingival bleeding and subgingival calculus than matched control subjects. Importantly, the aggressive periodontitis group had significantly more subgingival calculus than those with attachment loss, but no aggressive disease, suggesting an association between gingival disease and the presence of subgingival calculus.6 (See Chapter 7 for a discussion of aggressive periodontitis.)
Definition
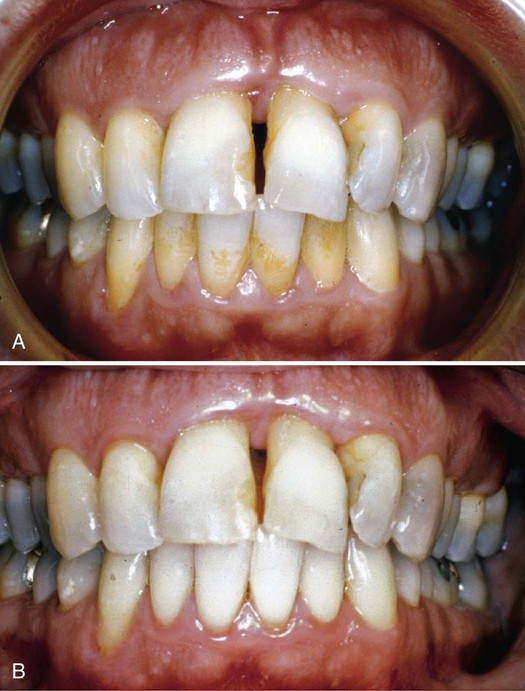
Dental hygienists commonly describe patients as light, moderate, or heavy calculus formers, depending on the amount of supragingival calculus that forms between recall visits. This distinction is useful in terms of estimating treatment time, but not much else. Patients may have light, moderate, or heavy deposits of subgingival calculus that forms below the gingival margin, supragingival calculus alone, or both. Figure 5-1 highlights the effects of dental hygiene care on a patient with heavy deposits of subgingival calculus. Tissue tone and color improve and the calculus and plaque biofilm removal results in reduced inflammation, causing tissue shrinkage.
Supragingival Calculus
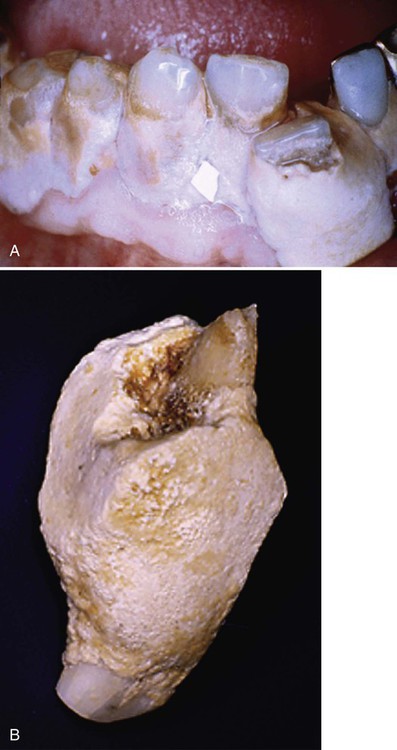
Supragingival calculus is found on the clinical crowns of the teeth, above the margin of the gingiva.7 It is readily visible as a yellowish-white accumulation, although it may darken with age. Figure 5-2 shows an extreme deposition of supragingival calculus.
Formation and Components
In supragingival calculus formation, mineral crystals are deposited in an organic matrix of plaque microorganisms, glucans, glycoproteins, and lipids. The calculi (calcified nodules) are stratified, suggesting that supragingival calculus is deposited in layers. Inorganic mineral content makes up about 80% of supragingival calculus. The minerals are primarily calcium phosphate (75.9%), calcium carbonate (3.1%), traces of magnesium,8 sodium, and potassium, and trace elements of fluoride, zinc, and strontium.2
The main crystal types in calculus are as follows:
• 58% hydroxyapatite (Ca10[PO4]6 · OH2)
• About 21% octacalcium phosphate (Ca8[HPO4]4)
Brushite appears in large proportions, up to 50%, in young, recently deposited calculus. After a few weeks or months, the predominant crystal type becomes hydroxyapatite.2
The organic component of calculus makes up to 15% to 20% of the dry weight of calculus. Half is protein from the bacterial cells, but it also includes carbohydrates and lipids from bacteria and saliva.2 Carbohydrates make up 1.9% to 9.1% of the organic component of supragingival calculus, salivary proteins account for 5.9% to 8.2% of the mass, and there are trace amounts of lipids. Dental calculus, salivary duct calculus (calculi that can form and block the openings of major and minor salivary ducts, referred to as sialoliths, or salivary stones), and calcified dental tissues are all similar in organic composition, but subgingival calculus lacks the quantity of salivary protein.8
Attachment to the Tooth Structure
Attachment to the tooth surface occurs in relationship to bacterial plaque biofilm. Acquired pellicle is a layer of glycoprotein deposited from salivary and crevicular fluids that are strongly bound to the tooth surface.9 Bacterial plaque biofilm formation begins on the pellicle with gram-positive coccoid organisms and then calcification occurs. After about 5 days, the plaque becomes filamentous and resembles decalcified mature calculus.2 Pellicle can grow into microscopic pores and openings and form dendritic (finger-like) structures in the cementum. These structures are thought to increase the tenacity of calculus attachment.2 Mechanical locking of calculus into tooth surface irregularities and close adaptation to tooth shape are also forms of attachment that can add to its tenacity.8
Mineralization in plaque can begin within 4 to 8 hours. Plaque may become 50% mineralized in 2 days and up to 90% mineralized after 12 days. Mineralization occurs more rapidly in some individuals and more slowly in others.7 Supragingival calculus can form on occlusal tooth surfaces when the teeth are out of occlusion or in crossbite, as well as on both fixed and removable prosthetic devices.
Subgingival Calculus
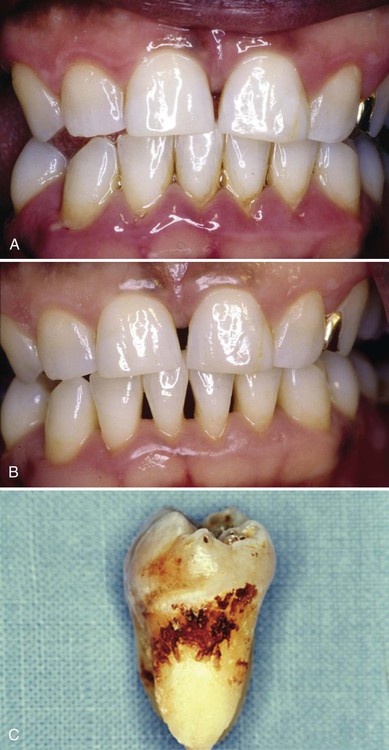
Subgingival calculus forms on root surfaces below the gingival margin and can extend into the periodontal pockets. Subgingival calculus is likely to be tenacious and is typically dark green or black, probably because of the organic matrix products of the subgingival plaque. The subgingival matrix of plaque biofilm contains blood products associated with subgingival hemorrhage. These matrix products have a dark pigment and differ from supragingival calculus, where the organic matrix components come primarily from saliva and do not contain blood.9 Subgingival calculus is commonly deposited in rings or ledges on root surfaces, but it may also appear on veneers. Subgingival calculus is also called submarginal calculus because of its location or serumal calculus because of the source of the mineral content.7 Figure 5-3 shows a patient with subgingival calculus.
Formation and Components
The organic components of subgingival calculus are similar to those of supragingival calculus, but contain more calcium, magnesium, and fluoride because of the higher concentrations of these minerals in crevicular fluid.2 There is no salivary protein present in subgingival calculus and, interestingly, the sodium content of subgingival calculus increases with the depth of the periodontal pockets.8
The pocket contents are composed of subgingival calculus adjacent to the root surface, attached plaque biofilm, and loosely attached or unattached bacteria. Bacterial cells become mineralized in the calculus, but do not form a solid mass; there are channels in which bacteria survive so that calculus is filled with bacteria that perpetuate the disease process.4 As with supragingival calculus, there is a layer of attached plaque biofilm covering the calculus and loosely adherent bacteria next to the tissue in the pocket. Figure 5-4 shows the contents of the periodontal pocket, including calculus.
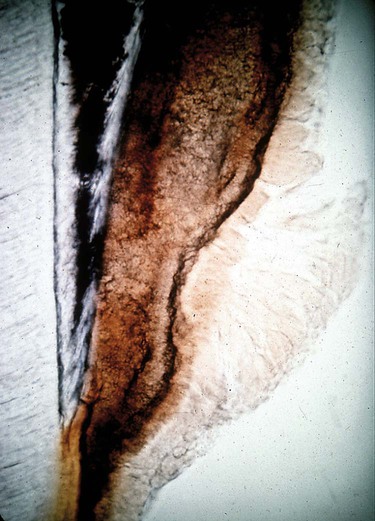
The calculus is immediately adjacent to the tooth root and crown and appears dark red. The enamel surface is dark blue and the dentin is light blue and appears striated because of the presence of dentinal tubules. Organized bacterial plaque biofilm is attached to the surface of the calculus and the unattached plaque is apparent as the less organized bacterial masses. There is no epithelium in the section. (Courtesy of Dr. Edward Green.)
Attachment to the Tooth Structure
Due to multiple means of attachment, subgingival calculus is tenacious and more difficult to remove than supragingival calculus. Pellicle attachment to cementum is the primary mechanism of subgingival calculus adherence. Deposits also form in and around microorganisms on the more irregular cemental surfaces, possibly caused by the loss of Sharpey’s fibers, resorption lacunae, or arrested caries.2 Crystals can grow deep into cemental irregularities. They appear morphologically similar to cementum (termed calculocementum)8 and exhibit intercrystalline bonding.2 Figure 5-5 shows a scanning electron micrograph of subgingival calculus adapted to a cemental surface and Figure 5-6 illustrates the microscopic appearance of calculus embedded in a root surface.
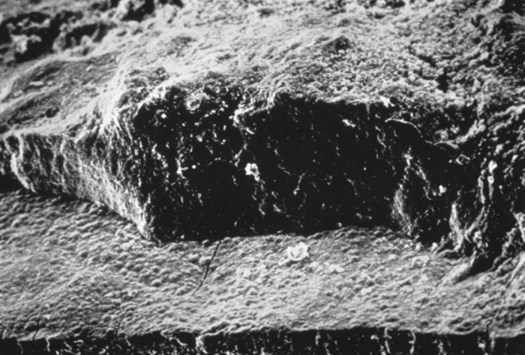
The cementum is the flat surface on the lower portion of the micrograph. (Courtesy of Dr. John Sottosanti.)
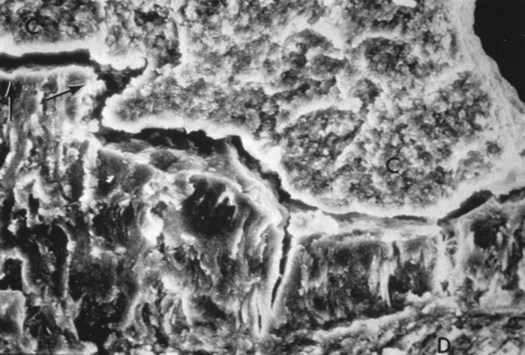
The cementum surface is the lower portion of the micrograph, calculus above. (Courtesy of Dr. John Sottosanti.)
Unlike supragingival calculus, the location of subgingival calculus is not site-specific.10 It is found anywhere throughout the mouth, and both radiographic examination and explorer detection are required to evaluate the extent and location of deposits. Radiographs alone tend to show mesial and distal deposits and underestimate the amount of calculus present because of technique-related artifacts. Overangulation of film, cervical burnout, and poor processing techniques limit the reliability of radiographic detection of subgingival calculus. Explorer detection provides the dental hygienist with buccal and lingual estimates of deposits, along with proximal estimates, but it is also subject to individual technique. It is advisable to use both radiographs and tactile skill to evaluate the amount of subgingival calculus present.
Distribution.
Supragingival calculus appears most commonly on the mandibular incisors and maxillary molars, especially the first molar, and is related to the openings of the major salivary ducts. Subgingival calculus is more evenly distributed throughout the mouth.10 It is a common mistake to assume that no calculus deposits occur around the maxillary anterior teeth. Although less frequent than other locations, both subgingival and supragingival calculus can occur there in as many as 10% of patients. Figure 5-7 shows the distribution of calculus found in a large population of patients.11
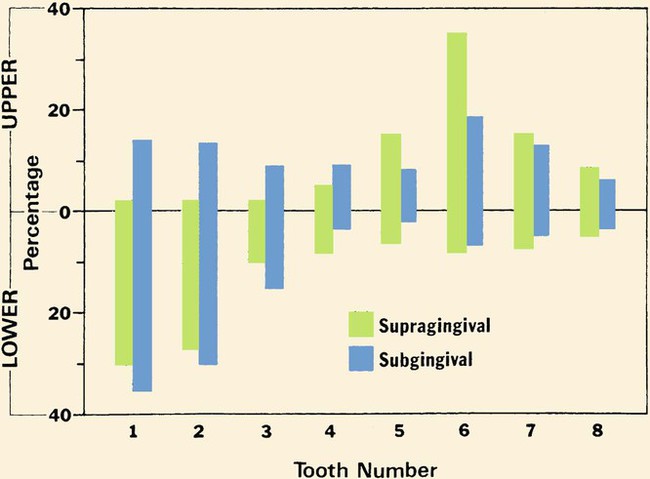
The percentage of individual teeth with supragingival and subgingival calculus is shown by tooth number and arch. Tooth #1 is the central incisor and tooth #8 is the third molar in each quadrant. Subgingival calculus forms on all teeth in the mouth, with greater formation in the mandibular anterior area. Supragingival calculus is most prevalent in the mandibular anterior and maxillary posterior areas but can also form on any tooth. (Adapted with permission from Schroeder HE. Formation and Inhibition of Dental Calculus. Vienna, Austria: H. Huber; 1969.)
The course of calculus formation and its distribution in humans has also been described. An important study by Anerud and colleagues12 compared 480 Sri Lankan male tea laborers, 14 to 31 years old, over a 15-year period with Norwegian men and boys, 16 to 30 years old, over a 20-year period. The researchers described the natural history of calculus formation with these data. Findings from Sri Lanka, a country with no dental care and in which toothbrushing was not performed, were compared with the clinical course of calculus formation in Norwegian subjects, who had optimum preventive dental care. Natural history refers to the course of calculus formation over time without any intervention, as in the case of the Sri Lankan subjects. The Norwegian subjects had ample opportunity to receive regular care in school clinics and private dental practices and represented the course of calculus formation tempered by regular, modern dental treatment. These data, summarized in Table 5-1, clearly showed the importance of calculus removal for long-term gingival health and emphasized the importance of maintenance therapy, including thorough calculus removal by the dental hygienist.
TABLE 5-1
Calculus Formation in Adult Male Populations on the Basis of the Findings of Anerud, Löe, and Boysen12
| CALCULUS | SUPRAGINGIVAL | SUBGINGIVAL | TOBACCO EFFECTS | LOSS OF ATTACHMENT | |
| Natural history: Sri Lankan tea workers with no dental care | All had some; amount increased over years | Formed early in life on maxillary molars and mandibular anterior teeth; continued to grow until age 25 or 30 years; was present on all teeth | Formed 6-8 years after eruption; continued to increase in extent and severity; leveled off at age 30 years | Smokers and betel nut chewers had more calculus than nonsmokers or nonchewers | Subgingival calculus associated with more attachment loss than teeth with no subgingival calculus |
| Clinical course: Norwegian men with regular dental care | Generally low levels over years | Found on maxillary molars and mandibular anterior teeth but at low levels; rarely found in other locations; did not increase in amount with age | Seen at low levels around age 20-29 years; no predilection for specific teeth; increased somewhat with age | Not associated with increased amounts of calculus | Accumulation of supragingival and subgingival calculus that were removed regularly had no effect on attachment loss |

For reasons that are not completely understood, there is tremendous individual variation in calculus formation rates. Plaque has shown evidence of mineral precipitation occurring in 1 to 14 days, but it can occur as quickly as within 4 hours. Calcifying plaques may become up to 50% mineralized in 2 days and 60% to 90% mineralized in 12 days.8 Heavy calculus formers have higher levels of calcium and phosphorus (from the saliva) in the plaque within a few days after a prophylaxis. In addition, differences in diet and in the composition of the microbial flora may contribute to formation rates. Mandel2 reviewed a number of characteristics that may be related to an increased rate of calculus formation:
Stay updated, free dental videos. Join our Telegram channel

VIDEdental - Online dental courses