Periodontal Diseases
• Describe the pathogenesis of periodontitis.
• Define periodontal disease activity.
• List and describe the American Academy of Periodontology categories of periodontal diagnosis.
• Define clinical attachment loss and its relationship to periodontitis.
• Compare and contrast the following forms of periodontitis as to demographics and clinical and microbiologic characteristics:
 Rapidly progressing periodontitis
Rapidly progressing periodontitis
 Necrotizing ulcerative periodontitis
Necrotizing ulcerative periodontitis
 Periodontitis as a manifestation of systemic disease
Periodontitis as a manifestation of systemic disease
• Identify systemic diseases and genetic factors associated with periodontal disease.
• State the role of systemic antibiotic treatment, locally delivered controlled-release antibiotic treatment, and enzyme suppression treatment in periodontitis.
• Describe the role of the dental hygienist in treating periodontal disease.
Periodontitis is a set of diseases. It is characterized by inflammation of the supporting tissues of the teeth, specifically the periodontal ligament, cementum, and alveolar bone.1 Unlike gingivitis, which is limited to the epithelium and gingival connective tissues, periodontitis results in loss of connective tissue attachment to the cementum on the tooth root. Loss of attachment creates a deepening of the gingival sulcus to form a periodontal pocket by migration of the junctional epithelium along the root surface. This is an inflammatory response that results in bone loss, recession, or both. Left unchecked, periodontitis continues to worsen and causes a weakened periodontium characterized by mobile teeth that are susceptible to abscess formation. The disease can be uncomfortable and unsightly. Teeth do not function effectively and are lost; they simply fall out, or they are extracted if they become acutely infected or untreatable by periodontal or restorative dental methods.
Periodontal Pocket
Pathogenesis of Periodontal Pockets
Probing pocket depths can increase for two reasons:
1. Coronal movement of the gingival margin through swelling or deepening of the sulcus. The resulting pockets that form are classified as gingival enlargements and periodontal pockets. Gingival pockets reflect tissue enlargement often related to inflammation and systemic conditions and are not associated with bone loss.2
2. Periodontal pockets reflect a progressive deepening of the sulcus through tissue destruction and are associated with bone loss.3
Biofilm → gingival inflammation → pocket formation → more biofilm formation2
Contents of the Pocket
The periodontal pocket contains subgingival plaque biofilm with many virulent bacteria, including motile forms that can survive in the sheltered pocket environment. It also contains metabolic products from the biofilm, which can be quite toxic, copious amounts of gingival fluid from the proliferating capillaries, and many cells. The pocket also contains calculus that has formed in the matrix of the plaque biofilm and adheres strongly to the teeth. Pus (accumulations of large numbers of dead cells and serum products) can also often be seen in periodontal pockets. The contents of the pocket are so virulent that they have been demonstrated to cause toxic effects in experimental animals even when the bacteria themselves have been filtered out.3
Pathogenesis of Periodontitis
Pockets deepen because of the breakdown of collagen fibers in the gingival connective tissue by enzymes such as collagenase, which are released by some of the plaque bacteria and the host’s inflammatory cells. The junctional epithelium elongates and then separates from the root surface at the coronal end. The plaque bacteria release a number of chemotactic substances that increase the flow of neutrophils into the gingival sulcus. The neutrophils react with bacteria, producing the suppuration (pus or exudates) often seen in progressing periodontal disease. Figure 7-1 shows the histologic events that occur in the periodontal pocket and crest of the bone.
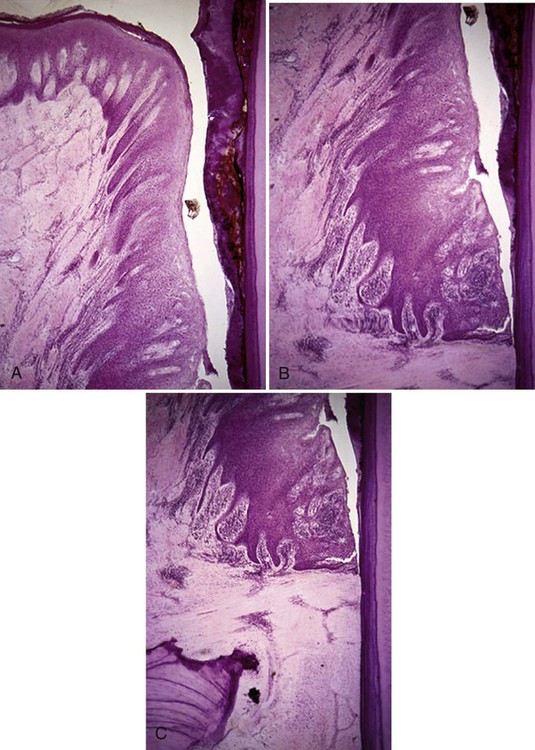
A, The coronal portion of the periodontal pocket is adjacent to a plaque biofilm and calculus-covered root surface. The sulcular epithelium is thickened, with exaggerated rete pegs extending into the underlying connective tissue. There is a dense inflammatory cell infiltrate under the sulcular epithelium and surrounding the blood vessels. B, The base of the same periodontal pocket shows the apical termination of the thickened junctional epithelium, which has separated from the root surface during specimen preparation. The epithelium precisely follows the contours of the root surface and calculus (right). A large amount of inflammatory cell infiltrate is seen within the thickened junctional epithelium. Microulcerations appear on the epithelium lining the pocket, and the dense infiltrate has replaced the collagen fibers in the connective tissue. C, A lower power view of the same specimen shows the crest of the alveolar bone and the periodontal ligament space. The inflammatory cells have migrated along the vascular channels into the periodontal ligament, spreading the infection. The structure on the lower left side is the crest of the alveolar bone. There is a single osteoclast seen in its resorption bay at the crest of the bone; the cell appears pale. (Courtesy of Dr. Gary C. Armitage.)
Periodontitis begins with apical migration of the junctional epithelium and loss of alveolar crest bone. Bone is an active tissue undergoing continuous resorption and formation, so when bone resorption exceeds apposition, a net decrease in the amount of bone occurs. Loss of crestal alveolar bone through the inflammatory process is called periodontal bone loss. There is limited evidence that systemic bone thinning, such as seen in osteoporosis,4 is related to increased alveolar bone loss and tooth loss.
When periodontal disease becomes established, both plasma cells and lymphocytes are present in the periodontal tissues. Plasma cells are important in antigen-antibody reactions, a major activity of the immune system. Antigen-antibody reactions activate a cascade of events that attract additional inflammatory cells to the periodontal tissues. These cells produce active molecules that cause additional destruction of the collagen fibers in the periodontal connective tissue. Lymphocytes, when stimulated by the bacteria in the plaque biofilm, release lymphokines, another class of active proteins. Lymphokines have many effects on the inflammatory system, including the production of chemical factors that activate osteoclasts and thus increase osseous resorption.4 For a more complete description of the host response, review Chapter 2.
Other products of the interaction of bacterial plaque with inflammatory cells include prostaglandins, which stimulate bone resorption. In combination, these and other factors, such as activated complement, appear to cause bone loss in progressing periodontitis. Interestingly, understanding the mechanisms of bone resorption may provide a clue to new methods of periodontal disease treatment. For example, nonsteroidal anti-inflammatory drugs inhibit prostaglandins and current research suggests that these drugs may inhibit some of the bone loss seen in periodontitis.5 However, in combination, these processes result in varying degrees of deepened pockets, tissue inflammation, bone loss, exposed furcations, and mobile teeth seen in periodontitis.6
Disease Activity
It is also important to know that periodontal disease is site specific. It does not occur in all teeth at any given time, and some teeth in the mouth may be untouched by disease. This is likely related to differences in the pathogenicity of plaque biofilm in various sites around the mouth. Site specificity of disease activity can result in new disease sites, increased tissue damage in existing sites, or both.7
Classification of Periodontitis
Periodontitis occurs in a variety of forms. The American Academy of Periodontology has classified periodontitis into a number of general categories on the basis of etiology, clinical presentation, pathogenesis, progression, and response to therapy.1 Seven categories of periodontitis are recognized, but they probably represent many different bacterial infections with similar symptoms. An additional category describes developmental or acquired deformities that are related to periodontitis. The classification categories are presented in Box 7-1.8
Chronic Periodontitis
Chronic periodontitis may have its preclinical onset in adolescence and, unless halted by therapy, it appears to progress for the life of the individual. This disease is characterized by bone resorption that progresses slowly and predominantly in a horizontal direction. It is not usually significant clinically until about the age of 35 years; however, clinically significant pocketing and bone loss can occur at any age. Epidemiologic evidence suggests that periodontitis may occur more commonly among U.S. men than women, but this is not a consistent finding throughout the world. It may be explained by the fact that women visit the dentist more regularly in this country and therefore may benefit from more prevention and earlier treatment (see Chapter 3).
The severity of chronic periodontitis is directly related to the accumulation of plaque biofilm and calculus on the surfaces of the teeth. The rate of periodontal destruction varies depending on disease activity and the patient’s resistance (host response). Chronic periodontitis is not associated with systemic disease or abnormalities in host defense.9
Chronic periodontitis is a prevalent disease that may be either localized or generalized and progresses intermittently until the teeth are lost by exfoliation or extraction. The progression of periodontitis appears to occur in episodic bursts of activity that cause attachment loss.7 The disease progresses in the presence of pathologic dental plaque biofilm and attachment loss occurs when collagen fibers in the local pocket are destroyed. Disease activity halts when the host resistance controls the disease process through therapy or natural defenses. The severity of disease can be characterized as described in Table 7-1.
TABLE 7-1
| SEVERITY | CAL MEASUREMENT |
| Slight | 1-2 mm |
| Moderate | 3-4 mm |
| Severe | ≥5 mm |
Clinical Attachment Loss
The defining element for classifying periodontal disease is not probing depth, but the level of attachment loss from the cementoenamel junction, which indicates bone loss. This concept is important for the dental hygienist to remember. Probing pocket depth (also called probe depth, probing depth, or pocket depth) is significant because the patient’s ability to adequately clean deepened pockets is greatly reduced. In addition, deepened periodontal pockets usually require instrumentation of complex root anatomy, including flutes, grooves, and furcation areas. Cleansing of the root surfaces, both through instrumentation and patient home care practices, is more difficult, and the prognosis for the tooth may be altered. However, the degree of periodontal disease severity is directly related to the amount of periodontal ligament destroyed and the amount of bone lost, which indicate loss of physical support for the tooth. In the healthy state, the crest of alveolar bone does not begin exactly at the cementoenamel junction but 1 to 2 mm apical to it. Viewing radiographs is therefore an indispensable aid in assessing the severity of bone loss in periodontal disease. Figure 7-2 highlights the significance of clinical attachment loss in evaluating periodontal disease compared with probe depth measurements and Figure 7-3 shows the long-term effectiveness of dental hygiene care in maintaining teeth with significant attachment loss.
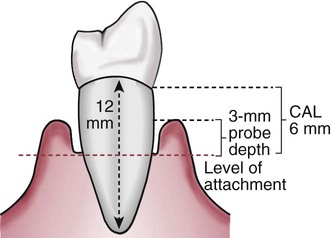
The typical root length is 10 to 12 mm, although there is significant variation among the teeth in the arch. Probing depth in the presence of attachment loss represents a more complete assessment of loss of support for the tooth than probing depth alone.
Plaque Biofilm
Chronic periodontitis is considered a multibacterial disease. Subgingival plaque biofilms associated with periodontal pockets at diseased sites contain high levels of gram-negative anaerobic and motile organisms. Elevated levels of spirochetes are also present. Of the primary pathogens associated with periodontal diseases, Porphyromonas gingivalis, one of the red complex bacteria, is perhaps the most common species identified.10 Other species commonly found in higher levels in chronic periodontitis include Tannerella forsythensis, Treponema denticola, Prevotella and Fusobacterium species, and Actinomyces actinomycetemcomitans. Our understanding of this extremely complex microbiology is growing through exciting research that will lead to improvements in both diagnosis and treatment for periodontal disease. For a further understanding of bacteria associated with plaque biofilms and periodontal disease, review Chapter 4.
The presence of specific pathogens is not always associated with the clinical presentation of chronic periodontal disease. A significant number of cases of chronic periodontitis have been reported without detection of P. gingivalis or other forms of periodontal pathogens in the pockets.11 Thus, diagnosis of periodontal diseases through the analysis of bacterial plaque biofilm remains an adjunctive technique. Until it is better understood, clinical rather than bacteriologic diagnosis remains the primary method of classifying periodontal disease.
Epidemiology of Chronic Periodontitis
Most patients with periodontal diseases have chronic adult periodontitis. It affects 20% or more of the adult population in the United States (see Chapter 3). Although this is a very complex disease, the 1999 Consensus Report of the World Workshop8 (sponsored by the American Academy of Periodontology) identified the following characteristics of chronic periodontitis:
• Most prevalent in adults, but it may appear in children and adolescents
• Periodontal destruction consistent with the amount of local factors
• Subgingival calculus frequently found
• Slow to moderate progression, with periods of rapid progression
• Classified on the basis of extent and severity
• Associated with local predisposing factors
Treatment
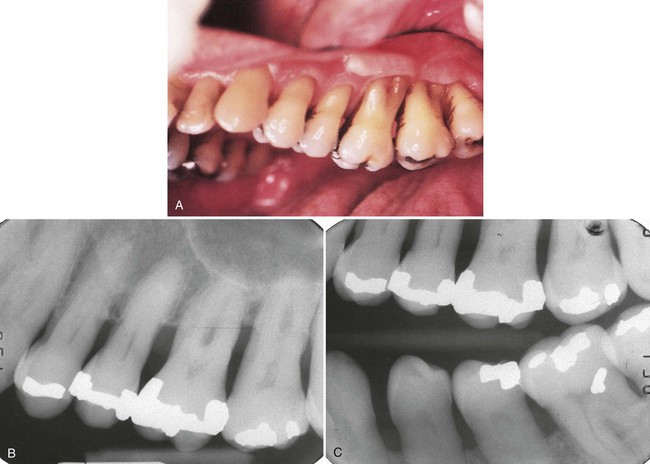
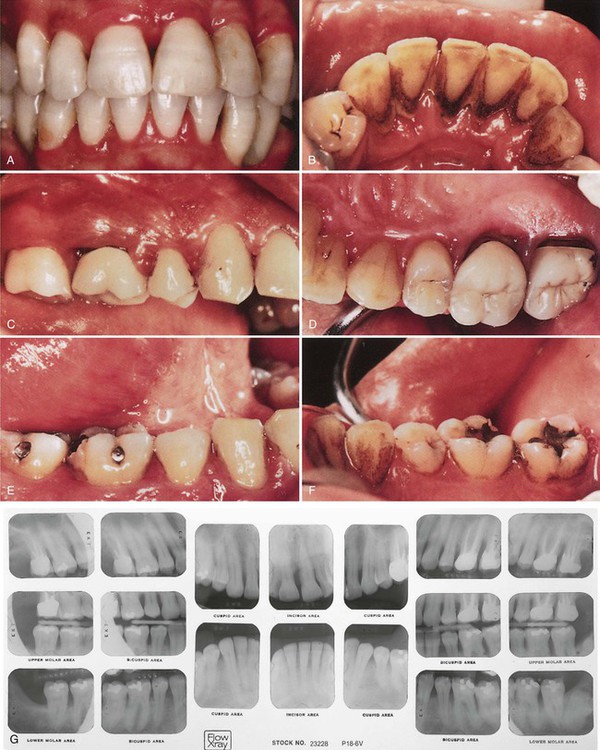
Patients with a diagnosis of slight to moderate chronic periodontitis are often treated in the general dental practice, with the dental hygienist taking a major role in initial therapy. However, patients with a diagnosis of advanced periodontitis are best treated in a specialty periodontics practice. Although an important part of the treatment may be performed by the dental hygienist in the periodontist’s office, the patient should be referred to a specialist for evaluation before any periodontal treatment is begun. With this approach, all signs of the disease will still be evident to assist the periodontist in complete diagnosis and treatment planning. Figures 7-4 to 7-6 show examples of slight, moderate, and severe periodontitis. A complete description of the dental hygiene treatment protocols for adult periodontitis is found in Chapter 10.
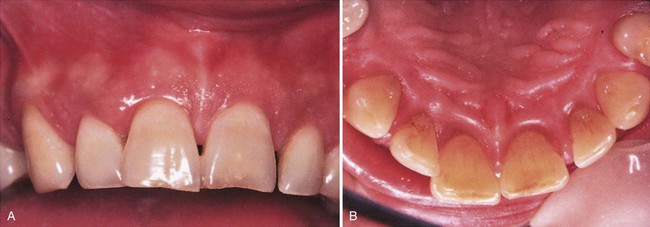
A, The clinical appearance of the maxillary anterior shows normal-appearing gingiva, with little obvious attachment loss. B, The clinical appearance of the lingual view suggests inflamed gingiva, but attachment loss must be assessed by careful probing around the teeth.
Stay updated, free dental videos. Join our Telegram channel

VIDEdental - Online dental courses