Clinical Assessment
Cheryl A. Cameron, Gina D. Evans and Dorothy A. Perry
• Describe the connection between patients’ overall health and their oral health.
• Define the aspects of clinical assessment in the dental hygiene process of patient care.
• List and describe the indices that measure:
• Describe the intrinsic and extrinsic dental stains and their associated causes.
• Compare and contrast normal and abnormal clinical presentation of the periodontium and dentition.
• Identify the radiographic changes seen in periodontal diseases.
• Compare and contrast normal and abnormal clinical and radiographic presentation of the periodontal structures surrounding dental implants.
The information and procedures essential to the assessment of a patient’s periodontal status include the chief complaint, medical and dental histories, clinical examination, and radiographic examination.1 The value of the assessment process is enhanced when data collection follows a comprehensive and systematic protocol. This chapter describes the elements of an assessment protocol for dental hygiene practice. An overview of the assessment process is presented in Box 8-1.
Patient History
Medical History
Medical history questionnaires vary in length, content, and format.2,3 It is important that the medical history survey remains current with evolving connections between overall health issues and dental and periodontal health, and that it gathers information critical to ensuring patient safety, health, and well-being (e.g., bisphosphonate use for conditions such as osteoporosis and associated dental implications).4 Regardless of which questionnaire is used, it is critical to supplement the information by interviewing the patient.5 All patient responses should be documented in the patient record and there should be no unanswered questions on the medical history form. Supplementing the medical history through consultation with other treating health care providers may also be necessary to fully address the goal of patient safety, health, and well-being. For example the patient’s orthopedic surgeon may need to be contacted regarding the need for premedication in the case of joint replacement.6
Patient vital signs must be taken and documented in the medical history. This includes blood pressure, pulse, and respiration. Although the taking of blood pressure may seem time-consuming, the increased understanding of the connection among hypertension, cardiovascular disease, and periodontal disease provides important support for its regular administration.6 Vital signs should be noted at the initial interview and all subsequent medical history updates. It is also critical that the medical history be dated and signed by both the patient and the dental hygienist to validate the patient’s responses.
Dental Health History
Patients should be asked to identify their chief complaints or reasons for seeking oral health care. Chief complaints and problems and possible oral health connections are described in Table 8-1. Patients should also describe any pain they may be experiencing. Common sources of oral pain are described in Table 8-2.
TABLE 8-1
Common Chief Complaints and Their Possible Oral Health Connections
| CHIEF COMPLAINT | POSSIBLE ORAL HEALTH CONNECTIONS |
| Bleeding gums (gingival inflammation) | Pathogenic bacteria, drug-induced bleeding (e.g., aspirin, anticoagulant therapy), drug-induced gingival hyperplasia (e.g., antihypertensive and antiseizure medications), and systemic disease–induced gingival inflammation and hyperplasia (e.g., diabetes, HIV) |
| Loose teeth (mobility) | Loss of periodontal support, bruxism, traumatic tooth injury, and root fracture |
| Spreading of the teeth with the appearance of spaces where none existed before (tooth migration) | Loss of periodontal support, missing teeth (e.g., lack of posterior dental support), myofunctional habits (e.g., tongue thrusting), and failure to use or maintain orthodontic retainers |
| Bad taste in mouth | Fungal infection, periodontal disease, smoking, and xerostomia |
| Bad breath (halitosis) | Chronic or acute periodontal disease, xerostomia, nasopharyngeal infection (e.g., sinus, tonsil, and throat infections), deep caries, and coated tongue |
| Food catching between teeth | Restoration defect resulting in a lack of interdental contact, tooth migration (e.g., high bite in dental restoration, bruxism, missing teeth), loss of periodontal support or interdental papilla, and marginal ridge defect |
| Rough spot on tooth | Supragingival calculus, chipped tooth, restoration defect, dental caries, wear facet, and recession |
| Dry mouth (xerostomia) | Medications (e.g., tricyclic antidepressants, antipsychotics, antihistamines, antihypertensive medications, diuretics), diseases of the salivary glands (e.g., Sjögren’s syndrome), head and neck radiation therapy, dehydration, smoking, and aging |
| Itchy feeling in gums | Chronic gingival inflammation, open contact, ill-fitting restoration (e.g., overhang) |
TABLE 8-2
Common Pain Descriptions and their likely Oral Health Connections
| CHIEF PAIN-RELATED COMPLAINTS | POSSIBLE ORAL HEALTH CONNECTIONS |
| Cannot open mouth wide enough or painful to open, or to brush or floss | Temporomandibular joint (TMJ) dysfunction, muscle fatigue associated with bruxism and clenching, muscle spasm, trauma associated with dental treatment or oral habit (e.g., dental restoration or extraction, chewing hard foods) |
| Headaches on the side of the head | TMJ, muscle fatigue associated with bruxism and clenching |
| Hurts to bite hard things | Tooth or root fracture, defective restoration, occlusal attrition or erosion, dental caries, bruised periodontal ligament (PDL) (e.g., high bite or traumatic injury) |
| Hurts to eat sugar or sweet things | Dental caries, defective restoration, tooth wear condition (i.e., attrition, abrasion, erosion, abfraction), dentin exposure from recession |
| Hurts to brush teeth at the gum line | Tooth wear condition, dentin exposure from recession, gingival inflammation, traumatic injury to the tooth and/or gingival tissue (e.g., toothbrushing technique, biting on hard foods, consuming acidic foods and beverages), acute manifestations of systemic and periodontal conditions (e.g., lichen planus, acute necrotizing ulcerative gingivitis [ANUG], herpes simplex) |
| Constant dull gnawing pain or dull pain after eating | Bruised PDL, pulpitis, periodontal disease, open contact with food impaction |
| Acute throbbing pain | Bruised PDL, pulpitis, food impaction (e.g., popcorn kernel), periodontal or endodontic abscess, deep dental caries, thermal sensitivity (hot or cold), systemic or localized infection (e.g., ANUG, primary herpes simplex, acute mucositis, infected oral piercing) |
| Sensitivity to hot and cold | Dental caries, pulpitis, recession, tooth wear condition, traumatic injury (e.g., recent restoration, malocclusion, blunt force tooth injury), bruxism, clenching, tooth fracture |
| Burning sensation in the gums | Fungal infection (e.g., candidiasis), xerostomia, chronic manifestation of systemic conditions (e.g., lichen planus), allergic reactions (e.g., sodium lauryl sulfate, cinnamon, palladium, nickel) |
A dental history should also include information about the patient’s previous dental experiences, current oral hygiene practices, and attitudes toward dentistry. Information about the patient’s family, social history, and habits (e.g., caffeine consumption, tobacco use, alcohol use, recreational drug use), are important elements of your assessment. Sometimes seemingly healthy lifestyle routines can have untoward effects on the dentition or oral health. For example, physical fitness and competitive sport activities can lead to high consumption of acidic sport drinks. The information gathered in the dental history may affect the plan and delivery of oral health care, as well as patient education. Always be aware that patients may not be willing to disclose health history information fully because they think it is not relevant to dental care or because of a concern for personal privacy.7
Clinical Examination
Extraoral and Intraoral Assessment
The head, neck, and oral cavity should be examined using visual and tactile techniques. This is the time to look for pathologic conditions, physical deviations, or modifications such as oral piercing. In addition, lymph chains and salivary glands should be palpated to identify swelling or masses. Additional visual adjunctive aids, such as autofluorescence and chemiluminescence, have been developed with the goal of early identification and intervention of high-risk oral lesions; however, their benefit to the dental practice has not yet been established.8,9
Findings from this assessment support the development of the dental and dental hygiene treatment plan and patient education plans. For example, when oral piercings are observed, the associated risks of gingival recession, tooth fracture, and infection should be discussed.10,11 The findings of the extraoral and intraoral assessment may also identify a need for dental specialty referral and for medical consultation and/or referral.
Oral Mucosa Assessment
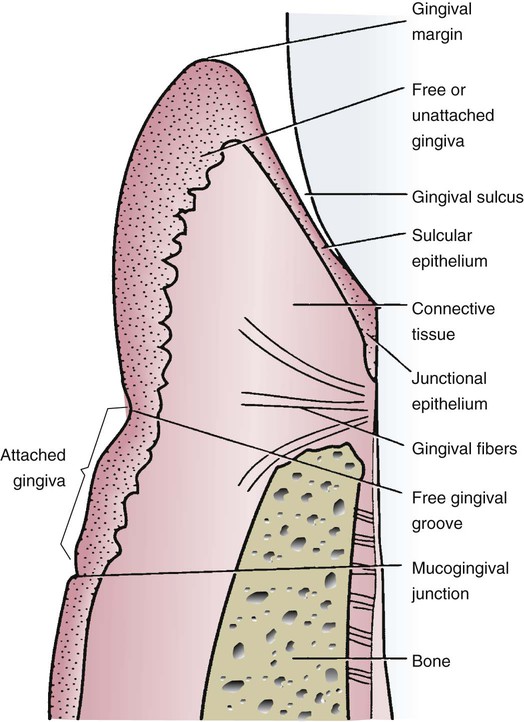
The oral mucosa assessment requires an understanding of the normal clinical features of the intraoral soft tissues. Three types of oral mucosa line the oral cavity: masticatory mucosa, specialized mucosa, and lining mucosa as described in Chapter 2. Figure 8-1 presents the anatomy of the normal gingiva adjacent to a tooth in diagrammatic form.
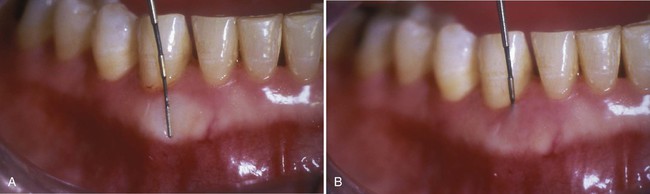
One clinically significant observation is noting the amount of attached gingiva extending from the free gingival groove to the movable alveolar mucosa. This junction is called the mucogingival junction. It is usually seen as a slightly scalloped line, as seen in Figure 8-2. The attached gingiva on the buccal aspect of the mandible and maxilla extends from the free gingival groove to the mucogingival junction and is continuous with the vestibule. The width of the attached gingiva is determined by subtracting the sulcus or pocket depth from the total width of the keratinized gingiva (from the gingival margin to the mucogingival junction). This measurement is performed by stretching the lip or cheek to demarcate the mucogingival line while the pocket is probed. The amount of attached gingiva is considered insufficient when stretching the lip or cheek induces movement of the free gingival margin. The attached gingiva on the lingual aspect of the mandible also extends from the free gingival groove to the mucogingival junction, but is continuous with the lining of the floor of the mouth. On the palatal aspect of the maxilla, the attached gingiva is continuous with the masticatory palatal mucosa. The amount of attached gingiva can be important for some restorative procedures.
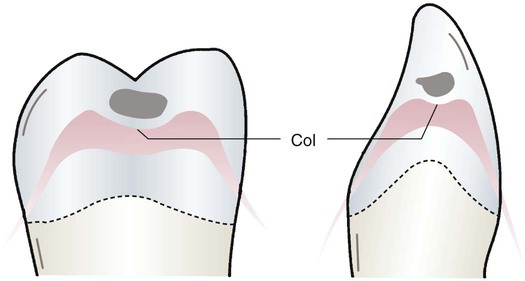
The interdental papilla is the tissue between two adjacent teeth. It consists of a facial or buccal papilla, a lingual papilla, and a col. The col is the valley, or depression, connecting the facial or buccal papilla and the lingual papilla. The col is shown in Figure 8-3. It is usually nonkeratinized and conforms to the interproximal contact area. The col is often absent in areas in which teeth are not in contact.
Frenum Attachments
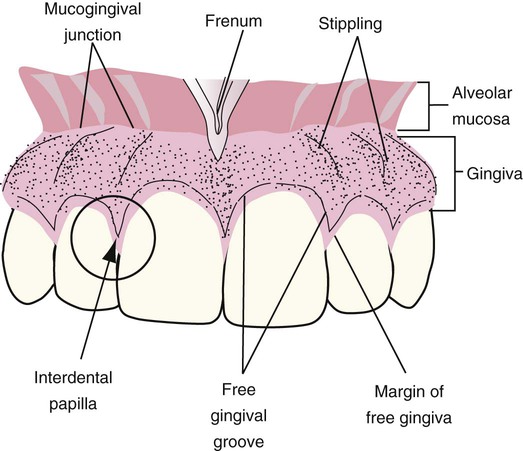
Frenum attachments are folds of mucosal tissues, often including muscle fibers, that join the movable mucosa to attached or specialized mucosa. Maxillary and mandibular anterior frenum attachments are located at the midlines of the maxillary and mandibular central incisors, as shown in Figure 8-4. The lingual frenum attachment is located on the underside of the tongue. Maxillary and mandibular buccal frenum attachments are located at the canines and premolars.
The assessment of the periodontal health of patients includes the description and documentation of the oral mucosal features. This documentation provides a baseline for planning dental and dental hygiene treatment and assessing the outcome of care. It is essential to describe clinical features, size, location, and severity clearly. Gingival observations may support the identification of underlying systemic, environmental, or behavioral contributing factors that may warrant further evaluation.12–15 Sample gingival descriptions are shown in Figures 8-5 and 8-6. Routine digital intraoral photographic images with supplemental narrative clinical observations are also useful for documenting and comparing the oral mucosa assessment at baseline and subsequent appointments, and for sharing with other members of the patient’s health care team. The electronic record is now commonly used and is a valuable resource to retain and compare images and other documentation over time.
Oral Hygiene Assessment
Plaque Biofilm
Plaque biofilm is a structured and functioning synergistic community of bacteria that support their combined metabolism and together enhance their overall bacterial virulence. It is described in detail in Chapter 4. Eighty-five percent of all dental treatment is related to the diagnosis, treatment, and prevention of plaque biofilm–related diseases. Periodontal disease and dental caries are the reason for 75% of all extractions.16
Identifying and documenting the location of plaque biofilm on the teeth can be used to educate patients on their specific oral hygiene prevention needs and provide a record over time. The plaque biofilm can be stained with a red disclosing solution to help patients see it or it can be shown to the patient when removed with an explorer or other instrument. The O’Leary Plaque Control Record provides a simple method of recording the presence of plaque biofilm at the gingival third of the buccal, lingual, mesial, and distal surface of each tooth.17 The plaque biofilm record quantifies the percentage of total tooth surfaces with plaque biofilm (e.g., 20 surfaces with recorded plaque biofilm ÷ 40 total surfaces = 50%). This method is particularly useful in research protocols requiring standardized recording of patient data.
In the clinical practice setting, the amount of plaque biofilm is more easily described as light, moderate, or heavy and the location is noted as generalized or localized, such as anterior, posterior, buccal, lingual, interproximal, and cervical or marginal. This documentation is kept in the patient’s permanent record and can be used to compare and evaluate the success of the plaque biofilm prevention program. An example of a plaque biofilm control record is presented in Figure 8-7.
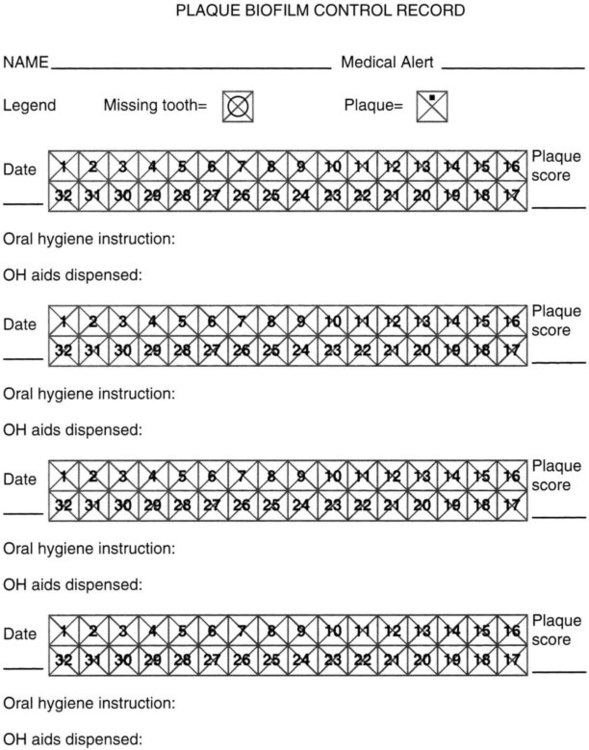
Four plaque scores can be recorded to provide the patient with a comparison over time, monitor improvement, and reinforce better plaque control. There is also space to record the oral hygiene (OH) instructions given at each session and to list the aids dispensed.
Calculus
The assessment of calculus is very important for developing the dental hygiene treatment plan. The presence of supragingival calculus can be observed directly. Light supragingival calculus deposits are more easily seen when dried with air. Subgingival calculus is detected using an explorer by carefully examining each tooth surface to the level of the periodontal attachment. Figure 8-8 shows the topography evaluated by explorer detection. Gentle blasts of air may be used to deflect the gingiva and permit some visualization of the calculus in the pockets.
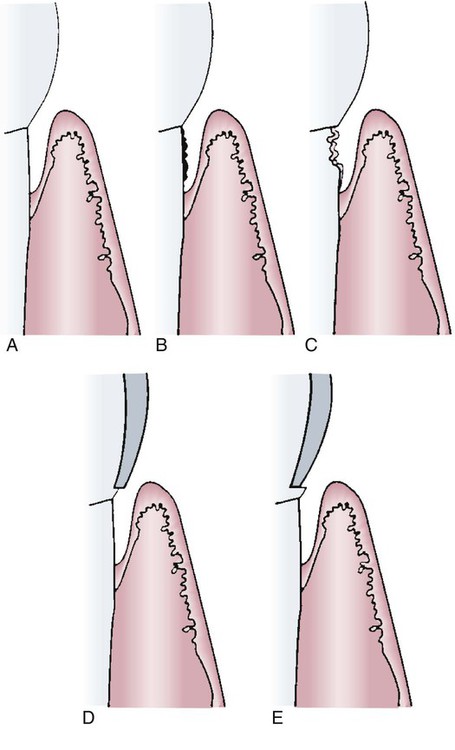
A, Pathologically deepened sulcus with normal topography. B, Calculus on the root surface. C, Caries on the root surface. D, Overhanging restoration margin. E, Undercontoured restoration margin.
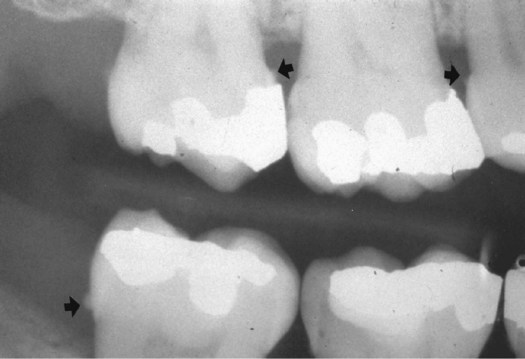
Radiographic images may show heavy interproximal calculus deposits, as seen in Figure 8-9. However, calculus is usually present in greater amounts on the teeth than can be seen on the two-dimensional images. Detection methods can be enhanced through the use of fiberoptic endoscopy, spectro-optical scanning, autofluorescence, ultrasound, and combined laser-autofluorescence technologies.18–20 These technologies are not routinely available in general dental practice so the combination of tactile, radiographic, and visual detection of subgingival calculus remains the general professional standard for the assessment of calculus.
Dental and Oral Tissue Stain
Extrinsic Stains
Brown Stain.
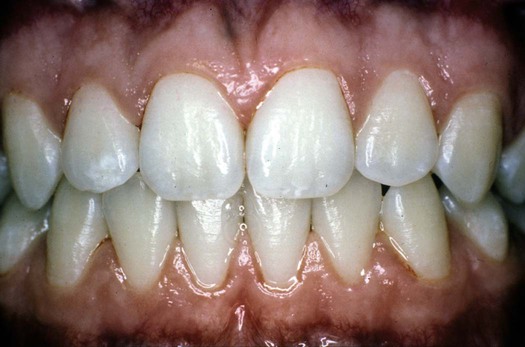
Brown stain is a thin, translucent, acquired, bacteria-free pigmented pellicle. It is frequently associated with poor oral hygiene and not using dentifrice. Brown stain is the most commonly occurring stain. It can be very diffuse and appear at varying intensities throughout the dentition. The discoloration is usually greater on irregular tooth surfaces and places that tend to concentrate pellicle, such as the gingival margins and interproximal areas. The brown staining is often caused by tannin, which has a denaturing effect on pellicular proteins and is found in coffee, tea, fruits, and red wine.21
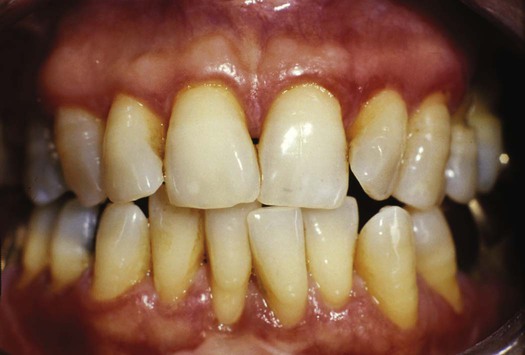
Chlorhexidine was introduced to U.S. dental practice in the 1980s as a general antiseptic with broad antibacterial action against gram-positive and gram-negative bacteria and yeasts. An undesirable side effect of continued use of chlorhexidine solution is that it imparts a yellow-brown to brown color to the tissues of the oral cavity. The staining also appears in the cervical and interproximal regions of the teeth, on restorations, in plaque biofilm, and on the surface of the tongue.22 Chlorhexidine also has a denaturing effect on proteins that may result in increased staining of the acquired pellicle by metal ions. There may also be increased binding of dietary chromogens after treatment with chlorhexidine.23 Chlorhexidine staining is shown in Figure 8-10.
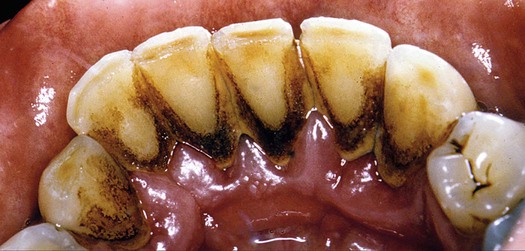
Note the formation of supragingival calculus, which is also stained.
The anticaries benefits of stannous fluoride have long been recognized. In addition, stannous fluoride is used for the control of plaque biofilm formation, gingivitis, and dentinal hypersensitivity. Stannous fluoride imparts a yellow-brown or golden discoloration of the tongue and teeth. It has been suggested that this stain is primarily stannous sulfide.24
Black Stain.
Black stain usually occurs as thin black lines on the facial and lingual surfaces of the teeth near the gingival margin and also as diffuse patches on proximal surfaces. It is firmly attached, tends to recur after removal, is more common in women, and may occur in mouths with excellent hygiene. The black stain that occurs in children is typically associated with a low incidence of caries. The microflora of black stain is dominated by chromogenic bacteria, which may be the cause of the black pigmentation.25 Another theory is that the black pigmentation is an insoluble ferric sulfide produced as a result of an interaction of hydrogen sulfide–producing bacteria and iron from the saliva and gingival fluid.26 Black stain tends to recur, and in some cases a 3- or 4-month recall program is required to respond to the patient’s aesthetic concerns.
Green Stain.
Green stain is a green-yellow stain, sometimes of considerable thickness. It is most often associated with children. The stain is considered to be stained remnants of the enamel cuticle, but this theory has not been substantiated. The discoloration has been attributed to fluorescent bacteria and fungi, such as Penicillium and Aspergillus.27 Green stain usually occurs on the gingival half of the facial surfaces of anterior teeth. It is associated with poor oral hygiene.28
Orange Stain.
Orange stain is less common than green or brown stain. It may occur on both the facial and lingual surfaces of anterior teeth. Orange stain is often associated with poor oral hygiene and is observed to form on loosely attached debris. Serratia marcescens and Flavobacterium lutescens have been suggested as the responsible chromogenic organisms.29
Preeruption Intrinsic Stains
Fluorosis.
The excessive ingestion of fluoride from sources such as toothpaste, fluoride supplements, and water can cause hypomineralization of enamel during development, known as fluorosis. Although the enamel remains decay-resistant, the aesthetic appearance is discolored and mottled. In its mildest form, fluorosis can appear as white flecks primarily on the cusp tips and facial surfaces of permanent dentition. The more severe forms of fluorosis, which result from source fluoride of a higher dosage or longer duration of exposure, may appear as white opaque areas or darkly stained and pitted enamel.30
Tetracycline can also cause tooth discoloration when used during tooth development. The appearance may range from yellow to gray-brown in color in teeth simultaneously developing during tetracycline exposure. This staining is extrinsic in the dentin so it cannot be removed. The discoloration takes on a banded appearance in the section of the tooth developing at the time of exposure, as seen in Figure 8-11.31
Other Intrinsic Stains.
In addition to drug-related intrinsic stains, tooth discoloration may occur as a result of metabolic disorders (e.g., hypothyroidism), inherited disorders (e.g., amelogenesis imperfecta, dentinogenesis imperfecta), systemic disorders (e.g., cystic fibrosis), or environmental factors (e.g., trauma).32
Posteruption Intrinsic Staining
Minocycline Stains.
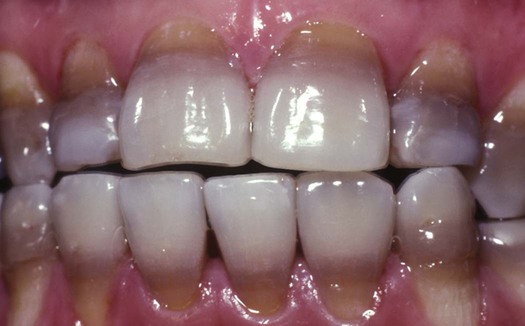
Minocycline, a commonly used medication with a broad range of clinical indications (e.g., acne, rosacea, rheumatoid arthritis, periodontal disease), has been shown to cause intrinsic dental and oral tissue discoloration, as seen in Figure 8-12. The onset of discoloration can occur within 1 month or many years after initiation of treatment.31 The discoloration ranges in color from green-gray to blue-gray.
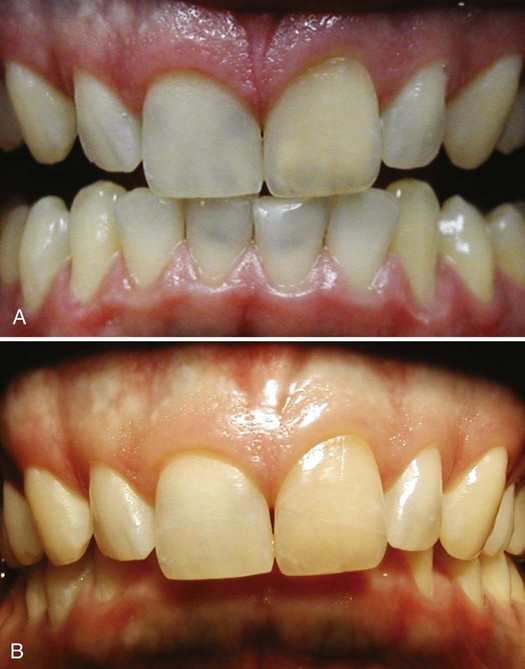
A, Intrinsic blue-gray dental staining of the upper and lower incisors. B, Intrinsic blue-gray oral tissue staining of the upper and lower attached gingival mucosa.
Periodontal Assessment
Periodontal assessment includes a clinical evaluation of the periodontium, looking for signs of inflammation and damage to periodontal tissues. The clinical detection of inflammatory lesions and damage to periodontal tissues is essential for diagnosis, treatment planning, and monitoring therapeutic effectiveness.34 The clinical assessments include the following:
• Measuring periodontal probe depths and clinical attachment levels
• Assessing gingival bleeding and suppuration
The assessment should follow an established sequence to ensure that it is comprehensive and complete. The diagnoses for periodontal diseases and conditions are defined by the American Academy of Periodontology’s Classification System for Periodontal Diseases and Conditions in Box 8-2.36 These are defined in detail in Chapters 6 and 7.
Stay updated, free dental videos. Join our Telegram channel

VIDEdental - Online dental courses