Later Stages of Development
Adolescence: The Early Permanent Dentition Years
Initiation of Adolescence
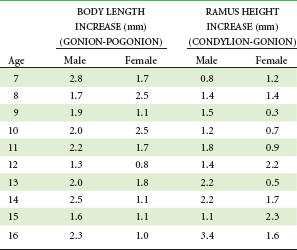
The first events of puberty occur in the brain, and although considerable research progress has been made in this area, the precise stimulus for their unfolding remains unknown. For whatever reason, apparently influenced both by an internal clock and external stimuli, brain cells in the hypothalamus begin to secrete substances called releasing factors. Both the cells and their method of action are somewhat unusual. These neuroendocrine cells look like typical neurons, but they secrete materials in the cell body, which are carried by cytoplasmic transport down the axon toward a richly vascular area at the base of the hypothalamus near the pituitary gland (Figure 4-1). The substances secreted by the nerve cells pass into capillaries in this vascular region and are carried the short distance to the pituitary by blood flow. It is unusual in the body for the venous return system to transport substances from one closely adjacent region to another, but here the special arrangement of the vessels seems made to order for this purpose. Accordingly, this special network of vessels, analogous to the venous supply to the liver but on a much smaller scale, is called the pituitary portal system.
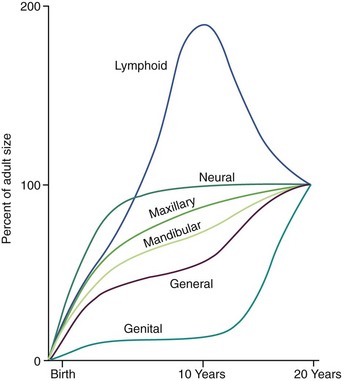
Under the stimulation of the pituitary gonadotropins, sex hormones from the testes, ovaries, and adrenal cortex are released into the bloodstream in quantities sufficient to cause development of secondary sexual characteristics and accelerated growth of the genitalia. The increasing level of the sex hormones also causes other physiologic changes, including the acceleration in general body growth and shrinkage of lymphoid tissues seen in the classic growth curves described in Chapter 2. Neural growth is unaffected by the events of adolescence, since it is essentially complete by age 6. The changes in the growth curves for the jaws, general body, lymphoid, and genital tissues, however, can be considered the result of the hormonal changes that accompany sexual maturation (Figure 4-2).
Timing of Puberty
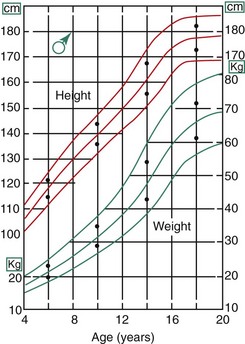
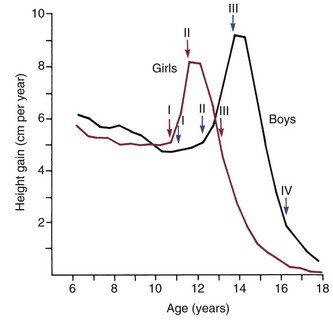
There is a great deal of individual variation, but puberty and the adolescent growth spurt occur on the average nearly 2 years earlier in girls than in boys (Figure 4-3). Why this occurs is not known, but the phenomenon has an important impact on the timing of orthodontic treatment, which must be done earlier in girls than in boys to take advantage of the adolescent growth spurt. Because of the considerable individual variation, however, early-maturing boys will reach puberty ahead of slow-maturing girls, and it must be remembered that chronologic age is only a crude indicator of where an individual stands developmentally. The stage of development of secondary sexual characteristics provides a physiologic calendar of adolescence that correlates with the individual’s physical growth status. Not all the secondary sexual characteristics are readily visible, of course, but most can be evaluated in a normal fully clothed examination, such as would occur in a dental office.
Adolescence in girls can be divided into three stages, based on the extent of sexual development. The first stage, which occurs at about the beginning of the physical growth spurt, is the appearance of breast buds and early stages of the development of pubic hair. The peak velocity for physical growth occurs about 1 year after the initiation of stage I, and coincides with stage II of development of sexual characteristics (see Figure 4-3). At this time, there is noticeable breast development. Pubic hair is darker and more widespread, and hair appears in the armpits (axillary hair).
The stages of sexual development in boys are more difficult to specifically define. Puberty begins later and extends over a longer period—about 5 years compared with 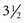 years for girls (see Figure 4-3). In boys, four stages in development can be correlated with the curve of general body growth at adolescence.
years for girls (see Figure 4-3). In boys, four stages in development can be correlated with the curve of general body growth at adolescence.
The timing of puberty seems to be affected by both genetic and environmental influences. There are early- and late-maturing families, and individuals in some racial and ethnic groups mature earlier than others. As Figure 4-4 shows, Dutch boys are about 5 cm taller than their American counterparts at age 10, and it is likely that both heredity and environment play a role in producing that considerable difference. In girls, it appears that the onset of menstruation requires the development of a certain amount of body fat. In girls of a slender body type, the onset of menstruation can be delayed until this level is reached. Athletic girls with low body fat often are slow to begin their menstrual periods, and highly trained female athletes whose body fat levels are quite low may stop menstruating, apparently in response to the low body fat levels.
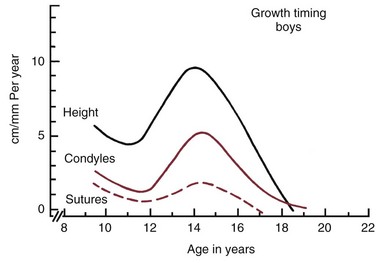
In the description above, the stages of adolescent development were correlated with growth in height. Fortunately, growth of the jaws usually correlates with the physiologic events of puberty in about the same way as growth in height (Figure 4-5). There is an adolescent growth spurt in the length of the mandible, though not nearly as dramatic a spurt as that in body height, and a modest though discernible increase in growth at the sutures of the maxilla. The cephalocaudal gradient of growth, which is part of the normal pattern, is dramatically evident at puberty. More growth occurs in the lower extremity than in the upper, and within the face, more growth takes place in the lower jaw than in the upper. This produces an acceleration in mandibular growth relative to the maxilla and results in the differential jaw growth referred to previously. The maturing face becomes less convex as the mandible and chin become more prominent as a result of the differential jaw growth.
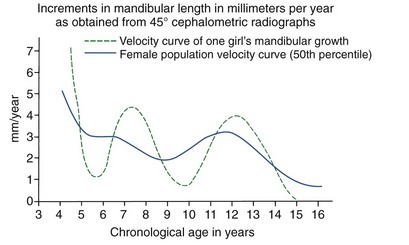
Although jaw growth follows the curve for general body growth, the correlation is not perfect. Longitudinal data from studies of craniofacial growth indicate that a significant number of individuals, especially among the girls, have a “juvenile acceleration” in jaw growth that occurs 1 to 2 years before the adolescent growth spurt (Figure 4-6).1 This juvenile acceleration can equal or even exceed the jaw growth that accompanies secondary sexual maturation. In boys, if a juvenile spurt occurs, it is nearly always less intense than the growth acceleration at puberty.
Recent research has shown that sexual development really begins much earlier than previously thought.2 Sex hormones produced by the adrenal glands first appear at age 6 in both sexes, primarily in the form of a weak androgen (dehydroepiandrosterone [DHEA]). This activation of the adrenal component of the system is referred to as adrenarche. DHEA reaches a critical level at about age 10 that correlates with the initiation of sexual attraction. It is likely that a juvenile acceleration in growth is related to the intensity of adrenarche and not surprising that a juvenile acceleration is more prominent in girls because of the greater adrenal component of their early sexual development.
Growth Patterns in the Dentofacial Complex
Growth of the Nasomaxillary Complex
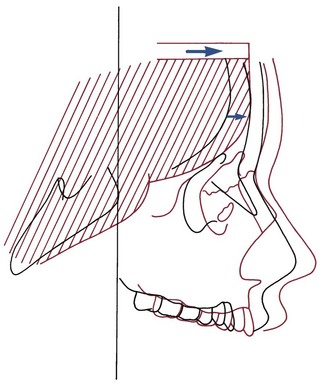
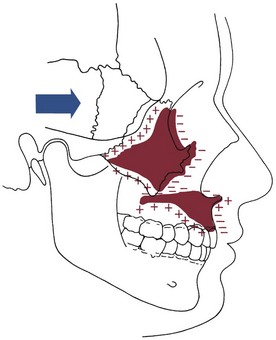
As we have noted in the preceding chapters, growth of the nasomaxillary area is produced by two basic mechanisms: (1) passive displacement, created by growth in the cranial base that pushes the maxilla forward, and (2) active growth of the maxillary structures and nose (Figure 4-7). Because the push from behind decreases greatly as the cranial base synchondroses close at about age 7, most of the growth after that time (i.e., during the time period when most orthodontic treatment is done) is due to active growth at the maxillary sutures and surfaces.
The effect of surface remodeling must be taken into account when active growth of the maxilla is considered. Surface changes can either add to or subtract from growth at the sutures by surface apposition or resorption, respectively. In fact, the maxilla grows downward and forward as bone is added in the tuberosity area posteriorly and at the posterior and superior sutures, but the anterior surfaces of the bone are resorbing at the same time (Figure 4-8). For this reason, the distance that the body of the maxilla and the maxillary teeth are carried downward and forward during growth is greater by about 25% than the forward movement of the anterior surface of the maxilla. This amount of surface remodeling that conceals the extent of relocation of the jaws is even more prominent when rotation of the maxilla during growth is considered (see the following sections).
Mandibular Growth
Growth of the mandible continues at a relatively steady rate before puberty. On the average, as Table 4-1 shows, ramus height increases 1 to 2 mm per year and body length increases 2 to 3 mm per year. These cross-sectional data tend to smooth out the juvenile and pubertal growth spurts, which do occur in growth of the mandible (see previous discussion).
TABLE 4-1
Data from Riolo ML, et al. An Atlas of Craniofacial Growth. Ann Arbor, Mich: University of Michigan Center for Human Growth and Development; 1974.
Stay updated, free dental videos. Join our Telegram channel

VIDEdental - Online dental courses


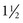 years after stage II and is marked by the onset of menstruation. By this time, the growth spurt is all but complete. At this stage, there is noticeable broadening of the hips with more adult fat distribution, and development of the breasts is complete.
years after stage II and is marked by the onset of menstruation. By this time, the growth spurt is all but complete. At this stage, there is noticeable broadening of the hips with more adult fat distribution, and development of the breasts is complete.