Occupational hazards
This chapter discusses the health of dental health workers (DHWs; dental professionals). Many occupations, especially the construction industry and armed services, are far more dangerous. The chapter also covers hazards to patients from some physical or chemical agents.
Health of DHWs
The standardized mortality ratio (SMR) – the ratio of observed deaths in an occupation or from any disease to the national average death rate for the same age and sex – is lower in dentists than in the general population. The main causes of death among dentists in UK, USA and other industrialized countries are cardiovascular disease, cancer and suicides, as in non-dental groups of similar socioeconomic status.
The risks in male dentists from liver cirrhosis and suicide are somewhat higher than in the general population, and dentists are also a risk group for malignant melanoma – presumably a consequence of sun exposure on holiday. Dentists, however, have a lower SMR for cancers, ischaemic heart disease, cerebrovascular disease and chronic lung diseases (bronchitis, emphysema and asthma).
Infections and physical, chemical and radiation hazards are the main occupational problems for DHWs. Nearly half of DHWs report sharp instrument injury at some time while treating patients. There are also risks from other accidents, and damage to eyes, hearing, mental health, musculoskeletal system, the fetus in pregnancy, respiratory systems and the skin (Table 31.1). Significantly greater numbers of female DHWs seem to experience musculoskeletal disorders and allergy to latex than males. The most prevalent preventive measure reported by DHWs is changing gloves between patients and using a face mask. Also, most DHWs have undergone hepatitis B virus and other vaccinations.
Table 31.1
Known occupational hazards in dentistry
< ?comst?>
| Hazard | Examples |
| Infections | |
| Bacteria | Legionellosis |
| Syphilis | |
| Tuberculosis | |
| Viruses | Common cold and other respiratory viruses |
| Hand, foot and mouth disease | |
| Hepatitis viruses | |
| Herpangina | |
| Herpesviruses | |
| HIV/AIDS and other retroviruses | |
| Human papillomavirus | |
| Parvoviruses | |
| Prions | |
| Physical and chemical | |
| Accidents and assaults | |
| Burns (chemical heat/cold/radiation) | |
| Chemical sterilizers | |
| Disinfectants | |
| Electrical accidents | |
| Fires and explosions | |
| Flammable or explosive agents | |
| Formaldehyde | |
| Inhalational anaesthetic agents | |
| Mercury | |
| Minerals | |
| Non-anaesthetic agents | |
| Poisons | |
| ‘Sick building syndrome’ | |
| Solvents | |
| Vibration injury | |
| X-ray processing chemicals | |
| Radiation | |
| Ionizing radiation | X-rays |
| Non-ionizing radiation | Electromagnetic radiation |
| Light (ultraviolet, blue, white) | |
| Lasers | |
| Video display units (VDUs or monitors) | |
| Pacemaker cardioverter and hearing aid interference | |
| To specific body systems | |
| Eyes | |
| Hearing | |
| Mental health | Reaction of staff to stress |
| Adapting to stress | |
| Musculoskeletal complaints | |
| Pregnancy | General anaesthetic agents |
| Infectious agents | |
| Ionizing radiation | |
| Mercury | |
| Respiratory system | |
| Skin | |
| To other systems | |
| Bone marrow | |
| Brain | |
| Cardiovascular system | |
| Gastrointestinal tract | |
| Kidneys | |
| Liver | |
< ?comen?>< ?comst1?>
< ?comst1?>
< ?comen1?>
In the UK, governmental concern and the defining of safe work practices have brought the question of occupational hazards into sharper focus, as have the measures for implementing safe practices enshrined in the Health and Safety at Work etc. Act (1974) and regulations such as the Ionizing Radiations Regulations (1988) and Control of Substances Hazardous to Health Regulations (1988). In the USA, the Department of Labor Occupational Safety and Health Administration (OSHA) has extensive legislation, which may be found at http://www.osha.gov/SLTC/dentistry/standards.html (accessed 30 September 2013).
Infections (See Also Ch. 21)
Overall, the major infectious hazards are respiratory and hepatitis B (HBV) and C virus (HCV) infections, and, to a very much lesser extent, human immunodeficiency virus (HIV), prions and health-care-associated infections (HCAIs), such as meticillin-resistant Staphylococcus aureus, Clostridium difficile, tuberculosis and Legionella.
Blood, serum and saliva are the sources most likely to transmit serious infection, as well as dental instruments that are often contaminated after use, and also bites. The transmission of blood-borne viruses is determined by the dose and viral concentration of an exposure. Whereas HBV transmission can be prevented by immunization of DHWs, controlling HCV and HIV requires efforts to reduce the incidence and dosage of exposures to blood and body fluids. Such strategies include the design and use of safe devices, targeted interventions, the use of gloves and other barriers, and ongoing surveillance and analysis of exposures.
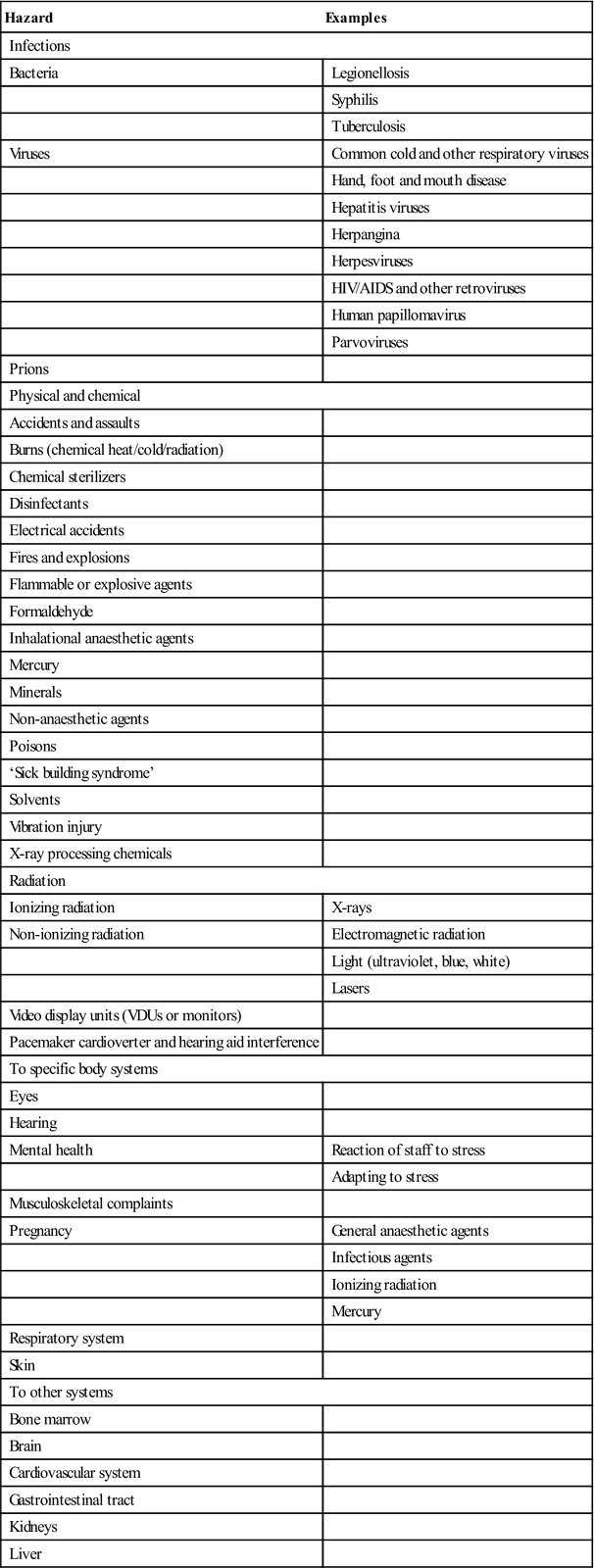
Since there is a potential for transmission of various infections, dental equipment should, where at all possible, be single-use and all reusable instruments must be sterilized or at least decontaminated (Fig. 31.1). DHWs should pay considerable attention to avoidance of needle-stick (percutaneous inoculation; sharps) infections/injuries and bites.
Standard (universal) infection control must be implemented; infection control guidelines can be found at http://www.bda.org/dentists/advice/ba/ic.aspx (accessed 30 September 2013).
The main points to remember in order to minimize cross-infection are shown in Box 31.1.
Needlestick/Occupational Exposure to Infections
Exposure-prone procedures (EPPs) are defined by the UK Department of Health as those where there is a risk that injury to the worker may result in exposure of the patient’s open tissues to the blood of the worker. These procedures include those where the worker’s gloved hands may be in contact with sharp instruments, needle tips or sharp tissues (spicules of bone or teeth) inside a patient’s open body cavity, wound or confined anatomical space where the hands or fingertips may not be completely visible at all times. Most procedures in dentistry are defined as EPPs, with the exception of:
■ examination using a mouth mirror only
■ visual and digital examination of the head and neck
■ visual and digital examination of the edentulous mouth
However, taking impressions from dentate or partially dentate patients would be considered exposure-prone, as would the fitting of partial dentures and fixed or removable orthodontic appliances, where clasps and other pieces of metal could result in injury to the dentist.
DHWs need not refrain from performing EPPs pending follow-up of occupational exposure to an HIV-infected source. The combined risks of contracting HIV infection from the source patient and then transmitting this to another patient during an EPP are so low as to be considered negligible.
Blood-borne microorganisms that can be transmitted via blood exposure include:
■ viruses:
Accidental exposure to blood caused by needle injuries or injuries following, cutting, biting or splashing incidents carries the risk of infection, particularly by blood-borne viruses though risks vary (Table 31.2).
Table 31.2
< ?comst?>
| Hepatitis C virus | Hepatitis B virus | Human immunodeficiency virus | |
| Risk of transmission by needlestick injury | 3% (3–10%) | 30% (5–40%) | 0.3% (0.2–0.5%) |
| Prevalence is higher than average in people who: | have had multiple blood transfusions; are dialysis patients or intravenous drug users | are intravenous drug users or men who have sex with men; are from developing countries | are men who have sex with men or intravenous drug users; are from areas where the condition is endemic |
< ?comen?>< ?comst1?>

< ?comst1?>
< ?comen1?>
Preventing needlestick injury and avoiding infection
The single most important measure that will help to prevent needle-stick injury is to avoid recapping and resheathing. Keep a rigid puncture-proof container close to hand for used needles to avoid the temptation of recapping. It is equally important to use proper protective clothing such as gloves, mouth mask and goggles.
Every DHW at risk from accidental exposure to blood should be trained in infection control and vaccinated against HBV (there are no preventive vaccines available for HCV or HIV).
Action after exposure to potentially contaminated material includes (Fig. 31.2) the following measures:
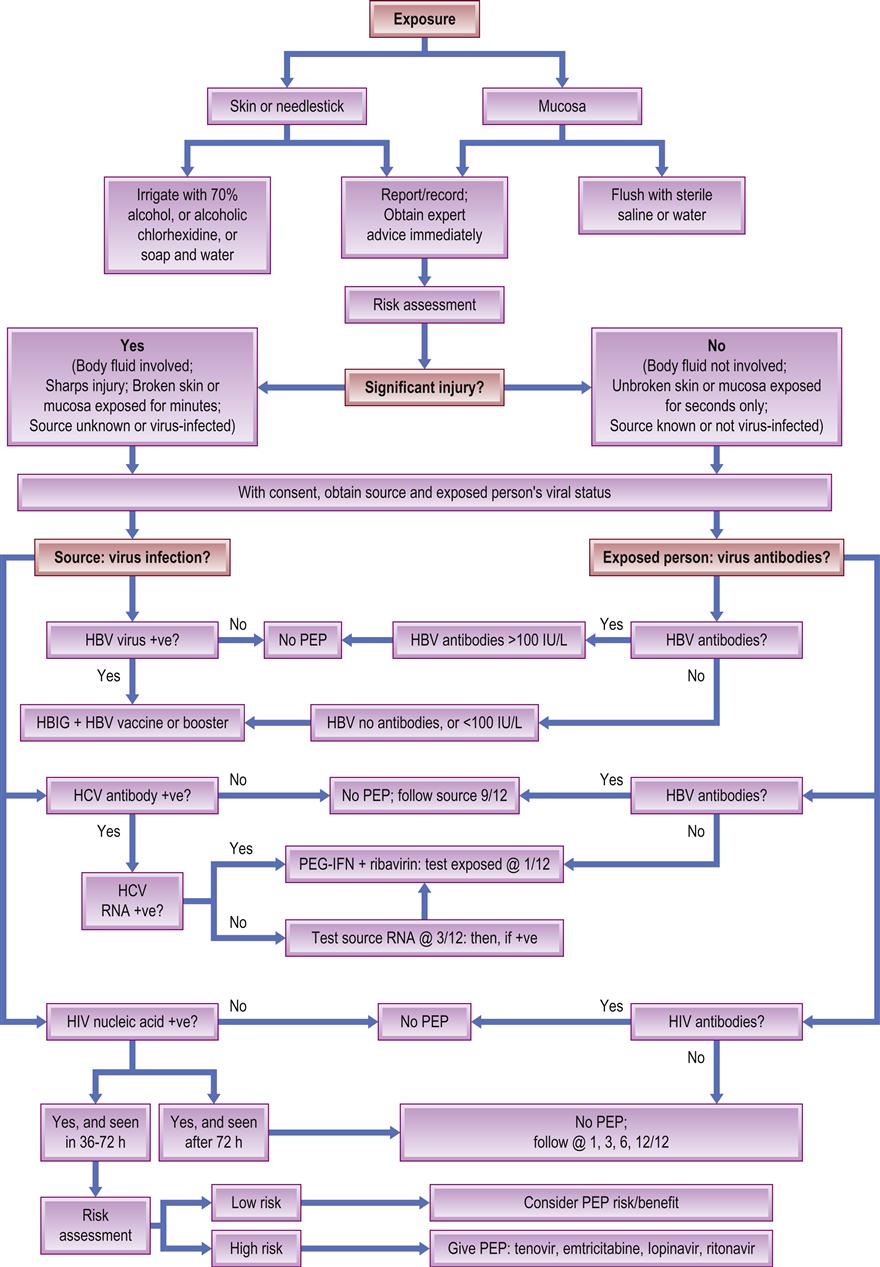
HBIG=hepatitis B immunoglobulin; HBV/HCV=hepatitis B/C virus; HIV=human immunodeficiency virus; PEG-IFN=pegylated interferon; RNA=ribonucleic acid. After Samaranayake and Scully 2013.
Under the Reporting of Injuries, Diseases and Dangerous Occurrences Regulations (1995), it is not usually necessary to tell the Health and Safety Executive (HSE) about an occupational exposure to blood or saliva; however, if the occupational exposure involves a known carrier of a blood-borne disease, this is classified as a dangerous occurrence and reporting then becomes mandatory – as it is when acute ill health has resulted from exposure to, or transference of, a biological pathogen. Recording of occupational exposure to blood or saliva in an accident report book is strongly advised and the incident should be reported immediately to the department dealing with occupational accidents.
Post-exposure prophylaxis (PEP) against HIV has been estimated to reduce the risk of transmission by 75%; it should be carried out within 1 hour for maximum effect, so an initial assessment must be performed as soon as possible. Even if there is a delay, it is still worth considering PEP within 24 hours of the exposure. A blood sample should be taken from the exposed person as soon as possible after the injury to act as a baseline value in case infection does arise. Further blood samples to test for HBV, HCV and HIV are collected after 1, 3, 6 and 12 months. If the source of the blood is known, the patient must be asked for permission to take a sample of blood for an HCV and HIV test. If the patient refuses, then they must be assumed to be a carrier. If the origin of the blood is unknown, then any blood present on the needle can be used for a serological examination.
Management is based on determining the level of risk of contracting HBV, HCV or HIV, depending on whether or not the injured person is non-immune, partially immune or fully immune for HBV (from vaccination or otherwise). If there is only limited immunity to HBV, then 5 mL intramuscular hepatitis B immunoglobulin (HBIG) should be given within 48 hours of the injury. After a potential HCV infection, combination treatment of pegylated interferon and ribavirin is the treatment of choice. A liver specialist should be consulted.
No PEP is currently available for hepatitis C. However, early treatment of acute hepatitis C infection may prevent chronic hepatitis C infection. Follow-up of exposed patients should be as that described in management for occupational exposure to hepatitis C.
A course of hepatitis B vaccination with or without immunoglobulin may be recommended as PEP following exposure to hepatitis B.
Post-exposure prophylaxis for HIV infection
After a potential HIV infection, the actual risk depends on the type of contact and on the amount of virus in the contaminated material. The risk of an HIV infection following exposure to blood is very small but factors associated with a higher risk are:
■ deep wounds (e.g. needlesticks, scalpel wounds)
■ visible blood on the instrument
■ needlestick injury from using hollow-bore needles containing blood
■ intravenous or intramuscular injection of contaminated blood
■ blood from a patient with a high virus level (e.g. untreated or terminal AIDS patients).
Decision 1: What is the level of risk of HIV transmission?
Level of risk is classed as low or negligible, which does not require PEP, or as a significant injury with a higher risk of transmission, which may require PEP. In the latter case, the source should be informed that the injury is significant enough to carry a risk of transmission and their permission should be obtained for a review of their social and medical histories. In the case of a splash, the volume of exposure should be assessed, as both HIV and HCV are reported to have been transmitted through only 0.5 mL of blood to the conjunctiva. In the unlikely event of a splash to a mucous membrane, then the exposure would be classed as a significant injury.
Decision 2: Is your patient at risk of carrying HIV?
A social and medical history can help determine whether HIV transmission is likely to have occurred and the associated degree of risk of that happening. Questions will relate to the patient’s sexuality and lifestyle; ideally, questioning should take place in a private room and the patient needs to be reassured that the discussion will remain confidential. The recipient of the injury should not ask the questions because he or she may be too anxious. When assessing the risk factors for acquiring HIV, the operator should concentrate on high-risk activities (Box 31.2) that increase the potential for transmission of the infection rather than concentrating on the main risk groups. The patient should be informed that guidelines are laid down by the UK health departments for the management of occupational exposures to blood or saliva, and that some questions need to be asked that relate to the risk of the patient having acquired HIV.
Low risk
If it is decided that the patient is in the low-risk category for HIV, then the procedure outlined under mild injury is followed.
High risk
If the patient admits to having participated in a high-risk activity, in an area with a high prevalence of HIV, then the specialist may recommend an HIV test. The test must not be carried out until trained health-care advisors have counselled the patient about the consequences of such a test. This takes more than an hour to carry out and the specialist may advise the recipient to attend a local hospital to be given initial doses of PEP drugs. Once the initial doses have been taken, a formal risk assessment for HIV may be carried out and a decision made as to whether to continue PEP until the HIV test result is known. If the injury occurs outside normal working hours, an alternative source of advice must be found and, if necessary, the first few doses of PEP obtained. The designated specialist can give guidance on this and the local accident and emergency department would probably be the first contact for advice and PEP.
What happens if the patient refuses to be tested?
A patient cannot be forced to give consent for the taking and testing of blood. The designated specialist can evaluate the history taken and give advice on whether the patient is at risk of a blood-borne disease. The patient can be asked to give permission for the practitioner to consult with the patient’s general medical practitioner for additional information. The incident should be recorded in detail in an accident report book.
What happens if the source patient is not known?
Exposure may occur while cleaning instruments from several patients. In this case, the patients should be identified and their medical histories reviewed. If the injury can be assessed as constituting a low risk for the transmission of HIV, the immediate administration of PEP against HIV is not necessary. The procedure outlined under mild injury should be followed, with the patients being contacted and asked to give blood for a hepatitis screen. If the injury is significant, then the designated specialist should be asked for advice as to whether PEP is necessary until the patients’ medical histories can be reviewed.
Immediate action after high-risk exposure
If PEP is advisable (Box 31.3), then it is important to discuss:
■ the advantages and disadvantages of PEP
■ the necessary follow-up examinations (of liver and kidneys) after 2 weeks and at 1, 3 and 6 months
■ follow-up examination for HIV infection (after 1, 3 and 6 months)
■ the importance of avoiding transmission to sexual partner(s) (use of condoms).
Start PEP as soon as possible after this. PEP for cases constituting a high risk of HIV infection is a three-drug combination consisting of two reverse transcriptase inhibitors and at least one protease inhibitor. The current regimen is:
The risk of acquiring HIV infection following occupational exposure is low – less than 3 per 1000 injuries.
Guidance on HIV PEP from the UK Chief Medical Officers’ Expert Advisory Group on AIDS (EAGA; 2008) is given in Appendix 31.1.
Current CDC guidance is similar. Nelfinavir, which was once used, is no longer recommended, since some preparations contain ethyl methane mesylate (EMS), a known animal carcinogen, mutagen and teratogen. This might be relevant to the choice of drugs included in an HIV PEP regimen because many health-care personnel are female and many are of child-bearing age.
Infection Control Measures
The four UK Health Departments’ decontamination policies are generally driven by the European Union’s Medical Devices Directive (MDD) 93/42/EEC and the Medical Device Regulations (MDR) 2005 (Box 31.4). ‘HTM 01-05’, published by the UK Department of Health in 2008 as ‘Health Technical Memorandum 01-05: Decontamination in primary care dental practices’ and superseded by a new edition published in April 2009 (http://www.dh.gov.uk/en/Publicationsandstatistics/Publications/PublicationsPolicyAndGuidance/DH_089245; accessed 30 September 2013), sets the standards for decontamination.
Infection control includes the safe disposal of sharps and disposable items, and the cleaning and sterilization of reusable dental instruments and devices. Care must be taken to ensure that all instruments are cleaned prior to sterilization, in a safe manner to avoid injury. Use of closed-system cassettes reduces the risk to the DHW during the execution of infection control. When using ultrasonic cleaners, washers and sterilizers, it is important always to follow the manufacturer’s instructions. Assurance of sterility of instruments and devices can be obtained through the use of one of several tests; these tests must be performed regularly to ensure that the sterilizer is cleaning all instruments and devices and that these are safe for use on patients. Disinfection after cleaning reduces the number of viable microorganisms on instruments, making them safer to handle; it is not acceptable to use chemical disinfectants (unless this is specifically recommended by the manufacturer), however, and therefore thermal disinfection is necessary.
Thermal disinfection is best achieved by using an automated washer–disinfector (WD), which precedes the use of autoclaves (sterilizers). The high temperature of the water and chemical additives in these devices clean and disinfect the instruments.
Two types of sterilizer are found in primary care dentistry:
Some instruments cannot be steam sterilized and must then be decontaminated according to the manufacturer’s instructions. If sending instruments for repair or disposal, decontaminate first.
Health-Care–Associated Infections
Health-care facilities, particularly hospitals, are well-recognized reservoirs of infection and may be responsible for a wide range of nosocomial (hospital) infections (Ch. 21).
Dental Unit Water Lines
The main routes of spread for prions, viruses, bacteria, fungi and protozoa in dental environments are via blood, infected equipment, saliva and water droplets, and direct patient contact. The dental unit structure favours the formation of aquatic microbial biofilms in dental unit waterlines (DUWLs), the small-bore flexible plastic tubing that conveys water to handpieces.
Biofilms are well-organized microorganism communities, and active biofilms are a source of microbial contamination of delivered water. Water delivered to a dental unit is by the so-called independent water system, the microbiological quality of which is extremely important for the water quality flowing from dental handpieces. In one study, over half of the water samples did not comply with the norms for potable water, though no Enterococcus or Legionella species were detected.
DHWs and patients can be exposed to direct contact with contaminated water both as splatter and as aerosol emitted by handpieces, including rotating and ultrasonic instruments. The most intensive aerosol and splatter emission occurs from an ultrasonic scaler tip or a high-speed handpiece.
Contamination of water in DUWLs is commonplace and the microbes isolated are various (Table 31.3). In one national survey (in Italy), for example, there was water microbial contamination in all dental surgeries examined, and 33% contained Legionella species.
Table 31.3
Microbes isolated in studies from dental unit water linesa
| Bacteria | Fungi |
| Achromobacter xylosoxidans | Alternaria alternata |
| Acinetobacter spp. | Aspergillus (=Eurotium herbariorum) repens |
| Actinomyces spp. | Aspergillus amstelodami |
| Alcaligenes denitificans | Aspergillus fumigatus |
| Bacillus spp. | Aspergillus glaucus group |
| Brevibacterium spp. | Aspergillus repens |
| Brevundimonas vesicularis | Aspergillus terreus |
| Burkholderiaceae | Candida albicans |
| Flavobacterium indologenes | Candida curvata |
| Klebsiella pneumoniae | Citromyces spp. |
| Legionella spp. | Cladosporium cladosporioides |
| Micrococcus luteus | Geotrichum candidum |
| Micrococcus lylae | Penicillium (glabrum) frequentans |
| Moraxella lacunata | Penicillium aspergilliforme |
| Nocardia spp. | Penicillium diversum |
| Ochromobacterum anthropi | Penicillium herquei |
| Pasteurella haemolytica | Penicillium pusillum |
| Pseudomonas acidovorans | Penicillium roseopurpureum |
| Pseudomonas aeruginosa | Penicillium turolense |
| Pseudomonas cepacia | Rhizopus nigricans |
| Pseudomonas fluorescens | Sclerotium sclerotiorum (Sclerotinia sclerotiorum) |
| Pseudomonas pickettii | |
| Pseudomonas paucimobilis | |
| Pseudomonas stutzeri | |
| Pseudomonas testosteroni | |
| Pseudomonas vesicularis | |
| Ralstonia pickettii | |
| Serratia marcescens | |
| Sphingomonas paucimobilis Stenotrophomonas maltophilia | |
| Staphylococcus capitis | |
| Staphylococcus cohnii | |
| Staphylococcus hominis ss. Novobiosepticus | |
| Staphylococcus saprophyticus | |
| Staphylococcus warneri | |
| Streptococcus spp. | |
| Streptomyces albus | |
| Xanthomonas maltophilia |
Legionella are common in DUWL, as are Legionella antibodies in DHWs. One 82-year-old patient appears to have contracted and died from Legionella infection emanating from her dental practice, and another case of fatal legionellosis in a dental surgeon concluded that the DUWL was the likely source of the infection. Although there are reports of unusual microbes, such as fungi (Exophiala mesophila) or non-tuberculous mycobacteria (NTM; Mycobacterium simiae and M. mucogenicum), in DUWL, other serious sequelae appear to be reported rarely. DUWL water in one study was only weakly contaminated by Legionella species and NTM, leading the authors to conclude that their aerosolization during dental treatment was not significant. The proportion of water samples with microbial levels above those recommended decreases during working or following attempts at disinfection.
Similar microbial contamination is also found in the operatory air, particularly close to the patient and DHW. Nevertheless, the number of published cases of infection resulting from exposure to water from conta/>
Stay updated, free dental videos. Join our Telegram channel

VIDEdental - Online dental courses


