Classification of Diseases and Conditions Affecting the Periodontium
Our understanding of the causes and pathogenesis of oral diseases and conditions is continually changing with increased scientific knowledge. In light of this, a classification can be most consistently defined by the differences in the clinical manifestations of diseases and conditions: they are clinically consistent and require little (if any) clarification by scientific laboratory testing. The classification presented in this chapter is based on the most recent internationally accepted consensus opinions of the diseases and conditions that affect the tissues of the periodontium; it was presented and discussed at the 1999 International Workshop for the Classification of the Periodontal Diseases organized by the American Academy of Periodontology (AAP).5 Box 3-1 presents the overall classification system, and each disease or condition is discussed where clarification is needed. In each case, the reader is referred to pertinent reviews on the subject and specific chapters within this book that discuss the topics in more detail.
Gingival Diseases
Dental-Plaque–Induced Gingival Diseases
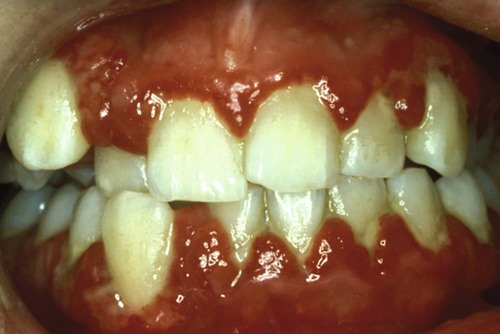
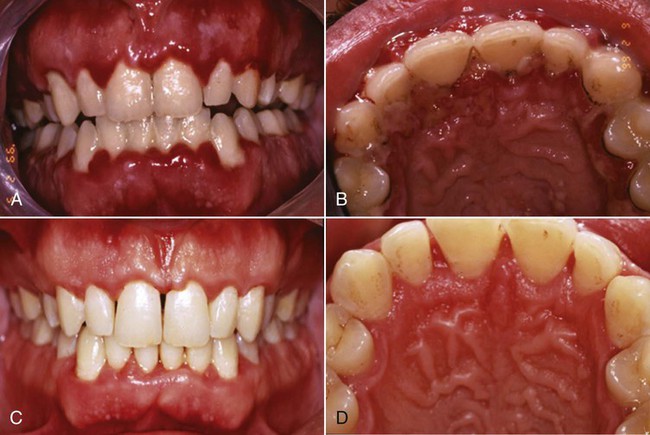
Gingivitis that is associated with dental plaque formation is the most common form of gingival disease (Figure 3-1). Box 3-2 outlines the classifications of gingival diseases. The epidemiology of gingival disease is reviewed in Chapter 4, and its causes are discussed in greater detail in Chapters 5 through 13 and elsewhere in this textbook.15,23,26,39,40 Gingivitis has been previously characterized by the presence of clinical signs of inflammation that are confined to the gingiva and associated with teeth showing no attachment loss. Gingivitis has also been observed to affect the gingiva of periodontitis-affected teeth that have previously lost attachment but that have received periodontal therapy to stabilize any further attachment loss. In these treated cases, plaque-induced gingival inflammation may recur but without any evidence of further attachment loss.
From this evidence, it has been concluded that plaque-induced gingivitis may occur on a periodontium with no attachment loss or on a periodontium with previous attachment loss that is stable and not progressing. This implies that gingivitis may be the diagnosis for inflamed gingival tissues associated with a tooth with no previous attachment loss or with a tooth that has previously undergone attachment and bone loss (i.e., with reduced periodontal support) but that is not currently losing attachment or bone, even though gingival inflammation is present (Figure 3-2). For this diagnosis to be made, longitudinal records of periodontal status, including clinical attachment levels, should be available.
Gingivitis Associated with Dental Plaque Only.
Plaque-induced gingival disease is the result of an interaction between the microorganisms found in the dental plaque biofilm and the tissues and inflammatory cells of the host. The plaque–host interaction can be altered by the effects of local factors, systemic factors, medications, and malnutrition, all of which can influence the severity and duration of the response. Local factors may contribute to gingivitis formation in addition to plaque-retentive calculus formation on crown and root surfaces (see Chapter 7). These factors are contributory because of their ability to retain plaque microorganisms and to inhibit the removal of those microorganisms via patient-initiated plaque-control techniques.
Gingival Diseases Modified by Systemic Factors.
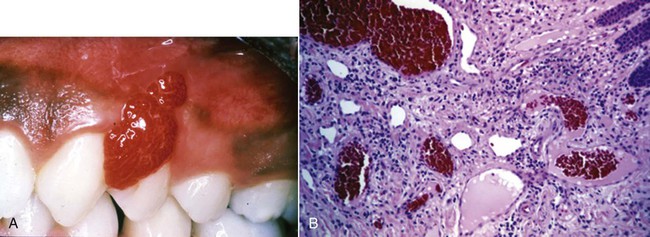
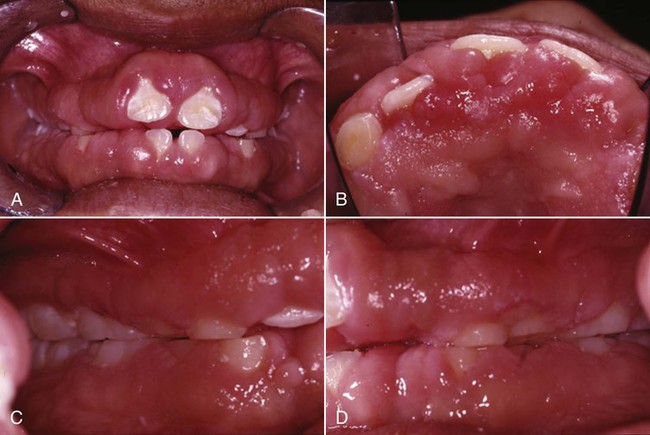
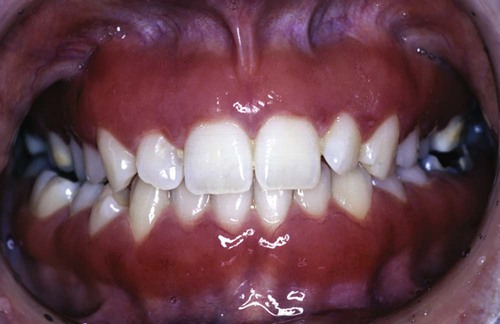
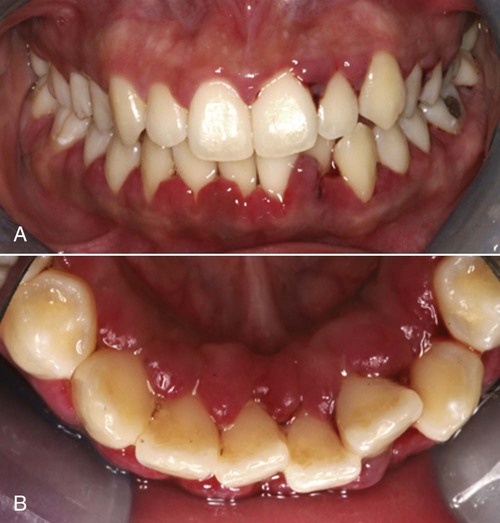
Systemic factors that contribute to gingivitis—such as the endocrine changes associated with puberty (Figure 3-3), the menstrual cycle, pregnancy (Figure 3-4, A), and diabetes—may be exacerbated as a result of alterations in the gingival inflammatory response to plaque.23,25,39 This altered response appears to result from the effects of systemic conditions on the host’s cellular and immunologic functions. These changes are most apparent during pregnancy, when the prevalence and severity of gingival inflammation may increase, even in the presence of low levels of plaque. Blood dyscrasias (e.g., leukemia) may alter immune function by disturbing the normal balance of the immunologically competent white blood cells that supply the periodontium. Gingival enlargement and bleeding are common findings that may be associated with the swollen, spongy gingival tissues caused by the excessive infiltration of blood cells (Figure 3-5).
Gingival Diseases Modified by Medications.
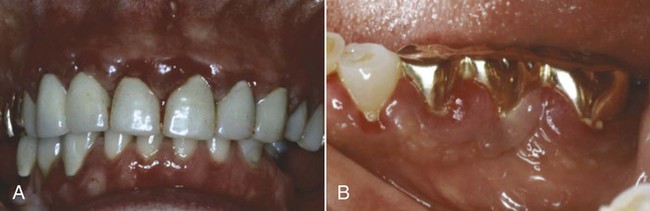
Gingival diseases that are modified by medications are increasingly prevalent as a result of the increased use of drugs known to induce gingival enlargement. These include anticonvulsant drugs such as phenytoin, immunosuppressive drugs such as cyclosporine (Figure 3-6), and calcium channel blockers such as nifedipine (Figure 3-7), verapamil, diltiazem, and sodium valproate.15,26,39 The development and severity of gingival enlargement in response to medications are patient specific and may be influenced by uncontrolled plaque accumulation as well as by elevated hormonal levels. The increased use of oral contraceptives by premenopausal women has been associated with a higher incidence of gingival inflammation and the development of gingival enlargement, which may be reversed by discontinuation of the oral contraceptive.
Gingival Diseases Modified by Malnutrition.
Gingival diseases modified by malnutrition have received attention because of clinical descriptions of bright red, swollen, and bleeding gingiva associated with severe ascorbic acid (vitamin C) deficiency or scurvy.26 Nutritional deficiencies are known to affect immune function, and they may alter the host’s ability to protect himself or herself against some of the detrimental effects of cellular products such as oxygen radicals. Unfortunately, little scientific evidence is available to support a role for specific nutritional deficiencies in the development or severity of gingival inflammation or periodontitis in humans.
Non–Plaque-Induced Gingival Lesions
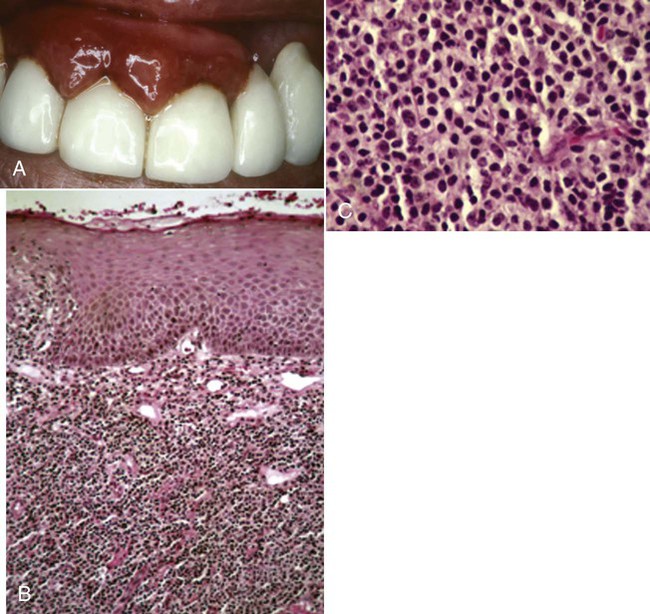
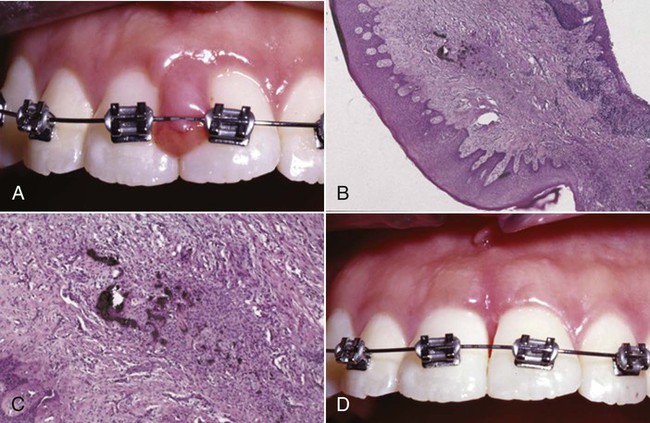
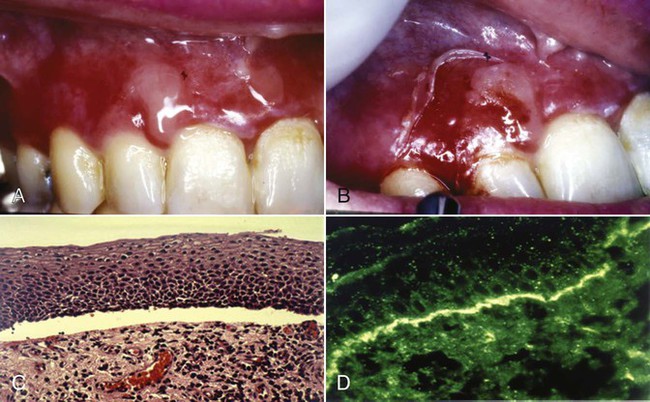
Oral manifestations of systemic conditions that produce lesions in the tissues of the periodontium are rare. These effects are observed more frequently among lower socioeconomic groups, in developing countries, and in immunocompromised individuals.20 Benign mucous membrane pemphigoid is an example of a non–plaque-induced lesion without socioeconomic implications. Sloughing gingival tissues leave painful ulcerations of the gingiva (Figure 3-8, A and B). Autoimmune antibodies are targeted at the basement membrane and cleave it from the underlying connective tissue (Figure 3-8, C and D).
Gingival Diseases of Specific Bacterial Origin.
Gingival diseases of specific bacterial origin are increasing in prevalence, especially as a result of sexually transmitted diseases such as gonorrhea (Neisseria gonorrhoeae) and, to a lesser degree, syphilis (Treponema pallidum).41,46 Oral lesions may be a result of systemic infection, or they may occur through direct infection. Streptococcal gingivitis or gingivostomatitis is a rare entity that may present as an acute condition with fever, malaise, and pain associated with acutely inflamed, diffuse, red, and swollen gingiva with increased bleeding and occasional gingival abscess formation. The gingival infections are usually preceded by tonsillitis, and they have been associated with group A β-hemolytic streptococcal infections.
Gingival Diseases of Viral Origin.
Gingival diseases of viral origin may be caused by a variety of deoxyribonucleic acid and ribonucleic acid viruses, with the most common being the herpesviruses (Figure 3-9, A and B). Lesions are frequently related to the reactivation of latent viruses, especially as a result of reduced immune function. The oral manifestations of viral infection have been comprehensively reviewed.20,45 Viral gingival diseases are treated with topical or systemic antiviral drugs (Figure 3-9, C and D).
Gingival Diseases of Fungal Origin.
Gingival diseases of fungal origin are relatively uncommon in immunocompetent individuals, but they occur more frequently in immunocompromised individuals and in those with normal oral flora that has been disturbed by the long-term use of broad-spectrum antibiotics.20,46,47 The most common oral fungal infection is candidiasis, which is caused by infection with Candida albicans; it can also be seen under prosthetic devices, in individuals using topical steroids, and in individuals with decreased salivary flow, increased salivary glucose, or decreased salivary pH. A generalized candidal infection may manifest as white patches on the gingiva, tongue, or oral mucous membrane that can be removed with gauze and that leave a red, bleeding surface. In individuals with human immunodeficiency virus (HIV), candidal infection may present as erythema of the attached gingiva; this has been referred to as linear gingival erythema or HIV-associated gingivitis (see Chapter 26).
The diagnosis of candidal infection can be made by culture, smear, or biopsy. Less common fungal infections have also been described.46,47
Gingival Diseases of Genetic Origin.
Gingival diseases of genetic origin may involve the tissues of the periodontium and have been described in detail.2 One of the most clinically evident conditions is hereditary gingival fibromatosis, which exhibits autosomal-dominant or (rarely) autosomal-recessive modes of inheritance. The gingival enlargement may completely cover the teeth, delay eruption, and present as an isolated finding; alternatively, it may be associated with several more generalized syndromes.
Gingival Manifestations of Systemic Conditions.
Gingival manifestations of systemic conditions may appear as desquamative lesions, ulcerations of the gingiva, or both.20,38,46 Allergic reactions that manifest with gingival changes are uncommon but have been observed in association with several restorative materials (Figure 3-10, A), toothpastes, mouthwashes, chewing gums (Figure 3-11), and foods (see Box 3-2). The diagnosis of these conditions may prove difficult and may require an extensive history and the selective elimination of potential causes. Histologic traits of biopsies from gingival allergic reactions include a dense infiltrate of eosinophilic cells (Figure 3-10, B and C).
Traumatic Lesions.
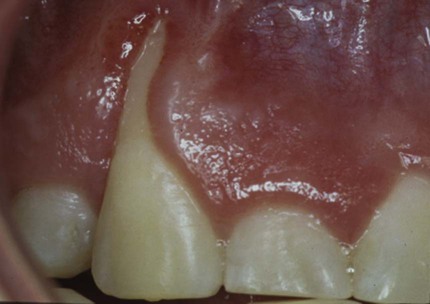
Traumatic lesions may be self-inflicted and factitious in origin, which means that they are produced by intentional or unintentional artificial means (Figure 3-12). Other examples of traumatic lesions include toothbrush trauma that results in gingival ulceration, recession, or both. Iatrogenic trauma (i.e., induced by the dentist or health professional) to the gingiva can be caused by the induction of orthodontic cement or preventive or restorative materials (Figure 3-13, A). Peripheral ossifying fibroma may develop in response to the embedment of a foreign body (Figure 3-13, B and C). Alternatively, accidental damage to the gingiva may occur as a result of minor burns from hot foods and drinks.20
Foreign-Body Reactions.
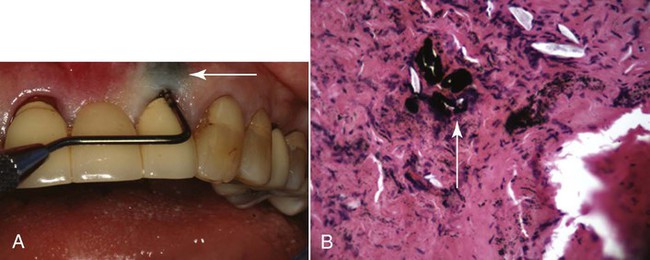
Foreign-body reactions lead to localized inflammatory conditions of the gingiva and are caused by the introduction of foreign material into the gingival connective tissues through breaks in the epithelium.20 Common examples are the introduction of amalgam into the gingiva during the placement of a restoration, the extraction of a tooth, or an endodontic apicoectomy with retrofill leaving an amalgam tattoo (Figure 3-14, A), with resultant metal fragments observed during biopsies (Figure 3-14, B); abrasives may also be introduced during polishing procedures.
Periodontitis
Although many classifications of the different clinical manifestations of periodontitis have been presented over the past 20 years, consensus workshops in North America in 19898 and in Europe in 19936 identified that periodontitis may present in early-onset, adult-onset, and necrotizing forms (Table 3-1). In addition, the AAP consensus concluded that periodontitis may be associated with systemic conditions (e.g., diabetes, HIV) and that some forms of periodontitis may be refractory to conventional therapy. Early-onset disease was distinguished from adult-onset disease by the age of onset (35 years of age was set as an arbitrary separation of diseases), the rate of disease progression, and the presence of alterations in host defenses. The early-onset diseases were more aggressive, occurred in individuals younger than 35 years old, and were associated with defects in host defenses; alternatively, adult forms of disease were slowly progressive, became clinically more evident during the fourth decade of life, and were not associated with defects in host defenses. In addition, early-onset periodontitis was subclassified into prepubertal, juvenile, and rapidly progressive forms with localized or generalized disease distributions.
TABLE 3-1
Classification of the Various Forms of Periodontitis
| Classification | Forms of Periodontitis | Disease Characteristics |
| American Academy of Periodontology World Workshop in Clinical Periodontics, 19895 | Adult periodontitis | Age of onset >35 years Slow rate of disease progression No defects in host defenses |
Stay updated, free dental videos. Join our Telegram channel

VIDEdental - Online dental courses