Anesthesia
• Perioperative Considerations of the Pregnant Patient
Laryngospasm
Discussion
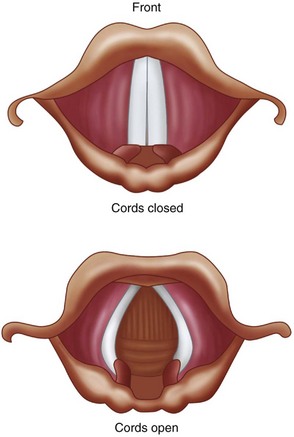
Laryngospasm results in tight approximation of the true vocal cords (Figure 3-1). It is a protective reflex that is most commonly caused by a noxious stimulus to the airway during a light plane of anesthesia. The structural and functional bases of the laryngospasm reflex were described by Rex. Secretions, vomitus, blood, pungent volatile anesthetics, painful stimuli, and oral and nasal airways may elicit this protective reflex. Mediated by the vagus nerve, this reflex is designed to prevent foreign materials from entering the tracheobronchial tree. During laryngospasm, the false vocal cords and supraglottic tissues act as a ball valve and obstruct the laryngeal inlet during inspiration. Laryngospasm has a reported occurrence of 8.7 per 1,000 patients receiving general anesthesia. It is 19 times more frequent than bronchospasm.
Baraka, A. Intravenous lidocaine controls extubation laryngospasm in children. Anesth Analg. 1978; 57:506–507.
Ciavarro, C, Kelly, JP. Postobstructive pulmonary edema in an obese child after an oral surgery procedure under general anesthesia: a case report. J Oral Maxillofac Surg. 2002; 60(12):1503–1505.
Hartley, M, Vaughan, RS. Problems associated with tracheal extubation. Br J Anaesth. 1993; 71:561–568.
Hurford, WE, Bailin, MT, Davison, JK, et al. Clinical procedures of the Massachusetts General Hospital, ed 5. Philadelphia: Lippincott-Raven; 1998.
Larson, CP. Laryngospasm–the best treatment. Journal of the American Society of Anesthesiologists. 1998; 89(5):1293–1294.
Leicht, P, Wisborg, T, Chraemmer-Joorgensen, B. Does intravenous lidocaine prevent laryngospasm after extubation in children? Anesth Analg. 1985; 64:1193–1196.
Louis, PJ, Fernandes, R. Negative pressure pulmonary edema. Oral Surg Oral Med Oral Pathol Oral Radiol Endod. 2002; 93(1):4–6.
Rex, MAE. A review of the structural and functional basis of laryngospasm and a discussion of the nerve pathways involved in the reflex and its clinical significance in man and animals. Br J Anaesth. 1970; 42:891–898.
Stoelting, RK, Miller, RD. Basics of anesthesia, ed 3. New York: Churchill Livingstone; 1994.
Perioperative Considerations for the Pregnant Patient
Briggs, GG, Freeman, RK, Yaffe, SJ. Drugs in pregnancy and lactation, ed 5. Philadelphia: Williams & Wilkins; 2005.
Cromwell, C. Hematologic changes in pregnancy. In Hoffman R, Benz EJ, Jr., Silberstein L, et al, eds. : Hoffman hematology: basic principles and practice, ed 6, St Louis: Saunders, 2012.
Cumminham, FG, Leveno, KJ, Bloom, SL, et al. Williams obstetrics, ed 21. New York: McGraw-Hill Professional; 2005.
Dollard, DF. Radiation in pregnancy and clinical issues of radiocontrast agents. In Roberts JR, Hedges JR, eds. : Clinical procedures in emergency medicine, ed 5, St Louis: Saunders, 2009.
Hawkins, JL, Bucklin, BA. Obstetrical anesthesia. In Gabbe SG, Niebyl JR, Galan HL, et al, eds. : Obstetrics: Normal and problem pregnancies, ed 6, St Louis: Saunders, 2012.
Lawrenz, DR, Whitley, BD, Helfrick, JF. Considerations in the management of maxillofacial infections in the pregnant patient. J Oral Maxillofac Surg. 1996; 54:474–485.
Mozurkewich, EL, Pearlman, MD. Trauma and related surgery in pregnancy. In Gabbe SG, Niebyl JR, Galan HL, et al, eds. : Obstetrics: Normal and problem pregnancies, ed 6, St Louis: Saunders, 2012.
Schwartz, N, Adamczak, J, Ludmir, J. Surgery during pregnancy. In Gabbe SG, Niebyl JR, Galan HL, et al, eds. : Obstetrics: Normal and problem pregnancies, ed 6, St Louis: Saunders, 2012.
Turner, M, Aziz, SR. Management of the pregnant oral and maxillofacial surgery patient. J Oral Maxillofac Surg. 2002; 60:1470–1488.
Respiratory Depression Secondary to Oversedation
HPI
The patient is an otherwise healthy woman for whom treatment was planned for bilateral upper and lower eyelid blepharoplasties with intravenous sedation. After the incision lines had been marked in the usual manner, ECG, blood pressure, pulse oximeter and a sidestream capnograph monitors were applied. The patient was administered 4 L of oxygen and 2 L of nitrous oxide via nasal hood (nitrous oxide decreases the amount of intravenous sedatives needed). Sedation was achieved using 5 mg of midazolam, 100 µg of fentanyl, and a propofol drip titrated to effect. Verrill’s sign (50% upper eyelid ptosis, indicating adequate sedation) was observed. Prior to administration of local anesthesia, 40 mg of propofol was administered as a bolus (propofol may cause a 20% to 25% drop in systolic blood pressure when given as a bolus). Upon administration of local anesthesia, loss of the capnogram, with no chest wall movement, was observed. (This indicates the presence of central apnea. Capnography is considered to be more sensitive than clinical assessment of ventilation in the detection of apnea. In a study by Soto and colleagues (2004), 10 of 39 patients (26%) experienced 20-second periods of apnea during procedural sedation and analgesia. All 10 episodes of apnea were detected by capnography but not by the anesthesia providers.) The apnea was attributed to the propofol bolus (combined with the respiratory depressant effects of fentanyl), which was anticipated to resolve shortly. However, the patient continued to be apneic, and her oxygen saturation decreased from 99% to 80% (pulse oximeter readings are about 30 seconds behind the real-time oxygen saturation). Tracheal tug and paradoxical chest wall motion were not observed (these would be signs of upper airway obstruction and inspiratory efforts). The patient began to appear cyanotic (bluish hue to facial skin and lips due to prolonged hypoxemia).
PMHX/PDHX/Medications/Allergies/SH/FH
The past medical and surgical histories are noncontributory. This patient is categorized as American Society of Anesthesiologist (ASA) Class I (Table 3-1). She does not use any medications and has no known drug allergies. She denies previous problems with local anesthetics (e.g., methemoglobinemia), intravenous sedation, or general anesthetics (problems with previous anesthesia or adverse drug reactions should alert clinicians to possible complications that may require modification of anesthetic techniques). There is no family history of complications with general anesthetics (e.g., malignant hyperthermia). She denies a history of drug or alcohol use (patients with a previous drug history or alcohol abuse may require higher doses of sedative-hypnotic drugs), and she does not smoke (smoking decreases oxyhemoglobin concentrations and increases pulmonary secretions).
Table 3-1
American Society of Anesthesiologists (ASA) Classification System for Stratifying Patients Preoperatively by Risk

Modified from the American Society of Anesthesiologists: Relative value guide, 2003, Park Ridge, Ill, the American Society of Anesthesiologists.
Examination
Cardiovascular. Heart is regular rate and rhythm without murmurs, rubs, or gallops.
Intraoperative. During the course of intravenous sedation (conscious sedation, deep sedation, or general anesthesia), it is important to continuously monitor the patient’s level of sedation and anesthesia (to prevent oversedation and respiratory depression) and to survey the ABCs (airway, breathing, and circulation [Box 3-1]).
Stay updated, free dental videos. Join our Telegram channel

VIDEdental - Online dental courses