Other Bone Diseases
This chapter discusses disorders of bone that do not easily fit into well-defined categories of disease.
Bone Dysplasias
Disease Mechanism
Bone dysplasias constitute a group of conditions in which normal bone is replaced with fibrous tissue containing abnormal bone. These lesions must be differentiated from tumors because the treatment is very different. Fibro-osseous lesion, originally a histopathologic term, is a commonly used term that includes the following bone dysplasias as well as neoplasms and other lesions of bone.
Fibrous Dysplasia
Disease Mechanism
Fibrous dysplasia results from a localized change in normal bone metabolism that results in the replacement of all the components of cancellous bone by fibrous tissue containing varying amounts of abnormal-appearing bone. At the histologic level, the result is the appearance of numerous short, irregularly shaped trabeculae of woven bone. These trabeculae are not aligned in response to stress but rather have a random orientation. Compared with normal bone, there are more trabeculae per unit volume within the cancellous component of the involved bone. This histologic appearance is responsible for the abnormal internal trabecular pattern seen in diagnostic images. More recent research has found that mutations in the Gs alpha (GNAS1) gene on chromosome 20q13.32 can be detected in 93% of cases of fibrous dysplasia tested. Fibrous dysplasias may be solitary or multiple (Jaffe type) or may occur in another multiple form associated with McCune-Albright syndrome, which usually comprises polyostotic fibrous dysplasia, cutaneous pigmentation (café au lait spots), and hyperfunction of one or more of the endocrine glands.
Clinical Features
The solitary (monostotic) form of fibrous dysplasia, which accounts for 70% of all cases, is the type that most often involves the jaws. The most common sites (in order) are the ribs, femur, tibia, maxilla, and mandible. The multiple (polyostotic) form usually is found in children younger than 10 years, whereas monostotic disease typically is discovered in a slightly older age group. The lesions usually become static when skeletal growth stops, but proliferation may continue, particularly in the polyostotic form. The lesions may become active in pregnant women or with the use of oral contraceptives; abnormal growth may occur after surgical intervention in young patients. Studies of the sex distribution of fibrous dysplasia show no sexual predilection except for McCune-Albright syndrome, which affects females almost exclusively. Symptoms of the disease may be mild or absent. Monostotic fibrous dysplasia often is discovered as an incidental finding in diagnostic images. Patients with jaw involvement first may complain of unilateral facial swelling or an enlarging deformity of the alveolar process. Pain and pathologic fractures are rare. If extensive craniofacial lesions have impinged on nerve foramina, neurologic symptoms such as anosmia (loss of the sense of smell), deafness, or blindness may develop.
Imaging Features
Location.
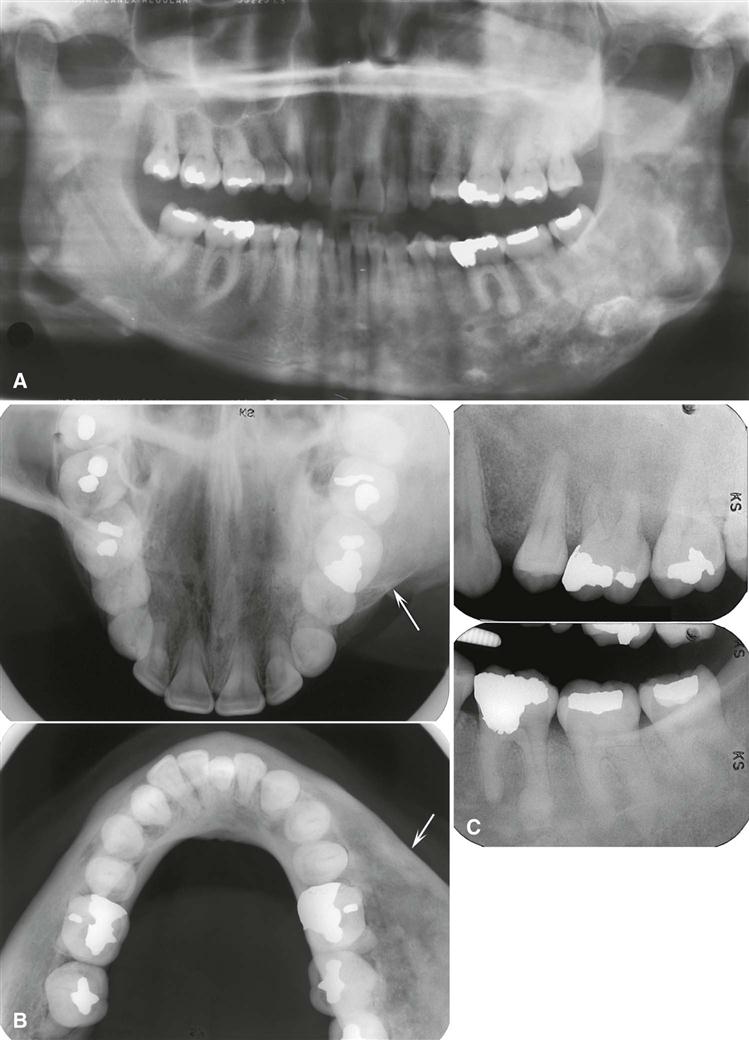
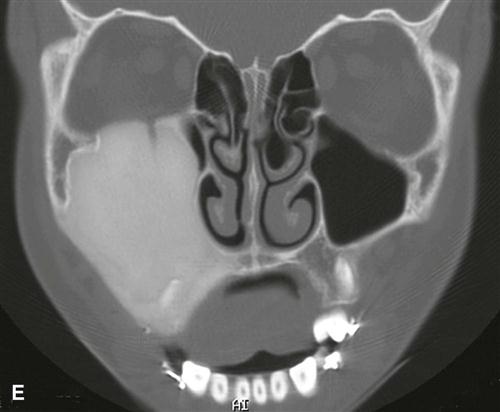
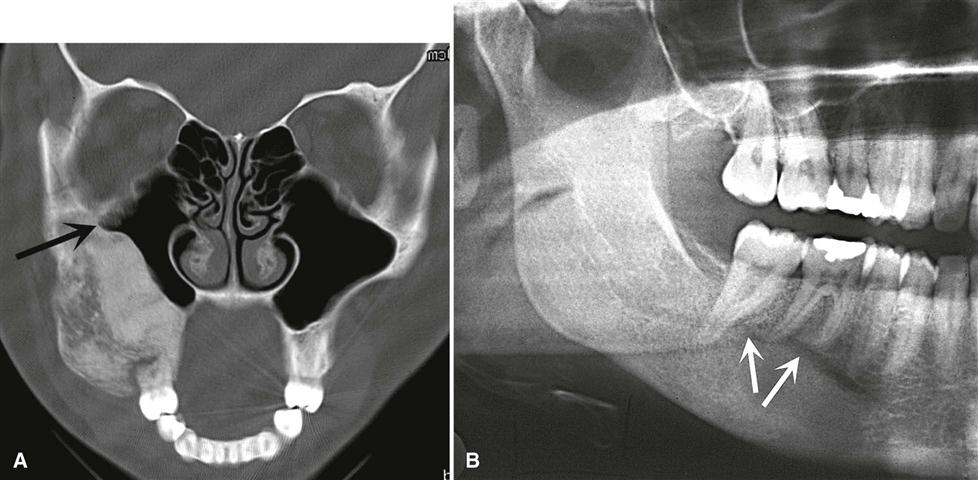
Fibrous dysplasia involves the maxilla almost twice as often as the mandible and occurs more frequently in the posterior aspect. Lesions more commonly are unilateral (Fig. 23-1) except for very rare extensive lesions of the maxillofacial region that are bilateral.
Periphery.
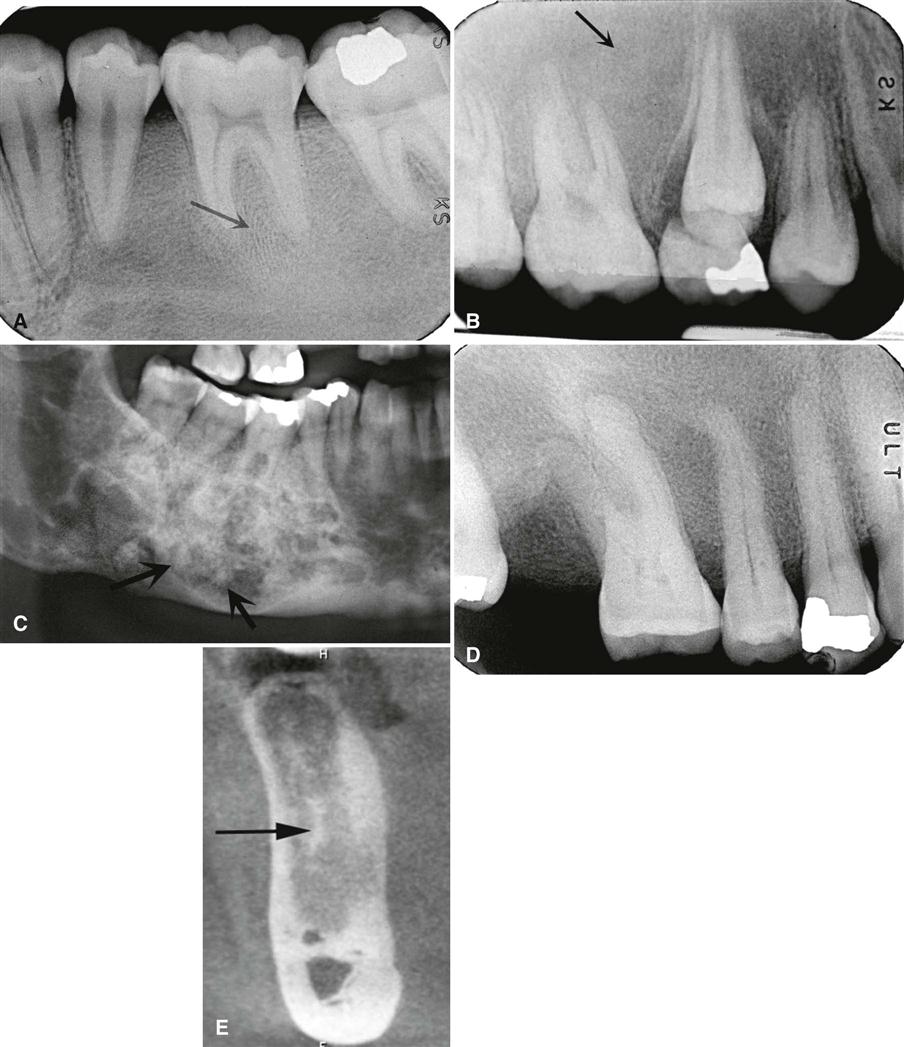
The periphery of fibrous dysplasia lesions most commonly is ill defined, with a gradual blending of normal trabecular bone into an abnormal trabecular pattern. Occasionally, the boundary between normal bone and the lesion can appear sharp and even corticated, especially in young lesions (Fig. 23-2).
Internal Structure.
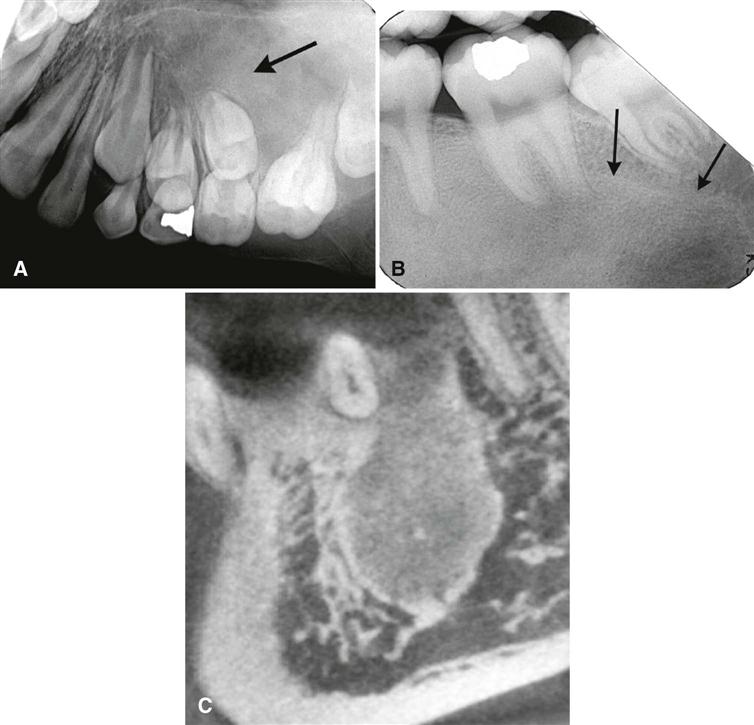
The density and trabecular pattern of fibrous dysplasia lesions vary considerably. The variation is more pronounced in the mandible and more homogeneous in the maxilla. The internal aspect of bone may be more radiolucent, more radiopaque, or a mixture of these two variations compared with normal bone (see Fig. 23-1). The internal density is more radiopaque in the maxilla and the base of the skull. Early lesions may be more radiolucent (Fig. 23-3) than mature lesions and in rare cases may appear to have granular internal septa, giving the internal aspect a multilocular appearance.
The abnormal trabeculae usually are shorter, thinner, irregularly shaped, and more numerous than normal trabeculae. This creates a radiopaque pattern that can vary; it may have a granular appearance (or ground-glass appearance, resembling the small fragments of a shattered windshield), a pattern resembling the surface of an orange (peau d’orange), a wispy arrangement (cotton wool), or an amorphous, dense pattern (Fig. 23-4). A distinctive characteristic is the organization of the abnormal trabeculae into a swirling pattern similar to a fingerprint (Fig. 23-5). Occasionally, radiolucent regions resembling cysts may occur in mature lesions of fibrous dysplasia. These are bone cavities that are analogous to simple bone cysts and occur more commonly in mandibular lesions (Fig. 23-6).
Effects on Surrounding Structures.
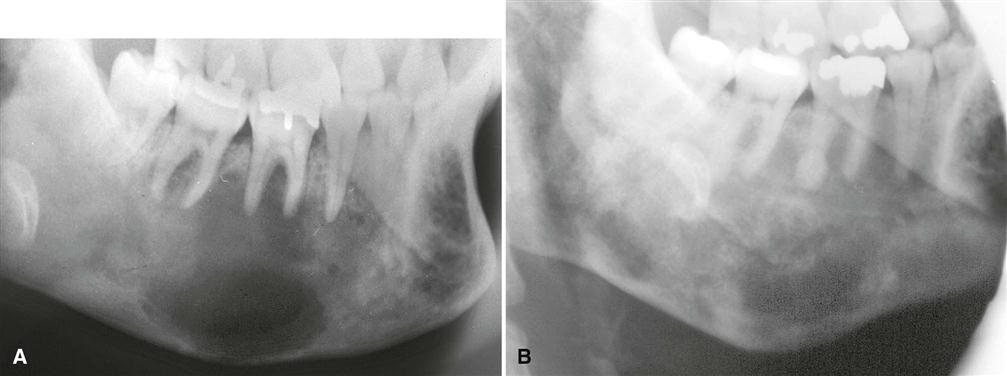
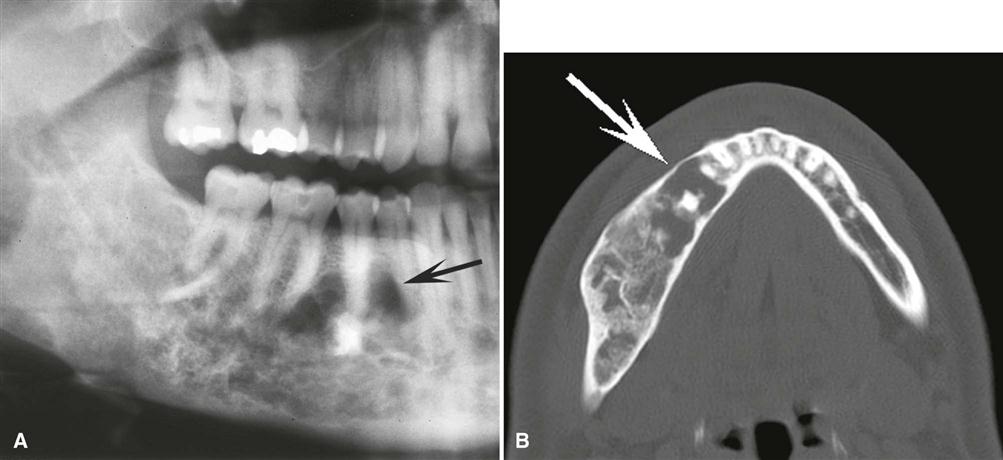
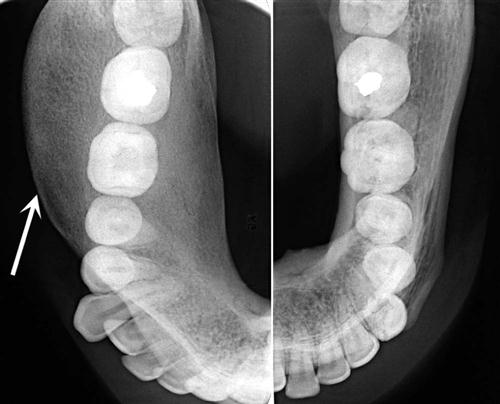
If the fibrous dysplasia lesion is small, it may have no effect on surrounding structures (subclinical variety). Effects on the involved bone may include expansion with maintenance of a thinned-outer cortex (Fig. 23-7). The expansion appears to affect the bone more evenly along its length rather than the more concentric expansion seen with benign tumors. Fibrous dysplasia may expand into the antrum by displacing its cortical boundary and subsequently occupying part or most of the maxillary sinus. Extension into the maxillary antrum usually occurs from the lateral wall, and the last section of the sinus to be involved usually is the most posterosuperior portion. Often the extension into the sinuses appears as a parallel thickening of the outer cortical border resulting in a residual antral air space, which still has approximately the normal anatomic shape of an antrum (Fig. 23-8). Cortical boundaries, such as the floor of the antrum, may be changed into the abnormal bone pattern. Often the bone surrounding the teeth is altered without affecting the dentition, and a distinct lamina dura disappears because this bone also is changed into the abnormal bone pattern (see Fig. 23-5). If the fibrous dysplasia increases the bone density, the periodontal ligament space may appear to be very narrow. Fibrous dysplasia can displace teeth or interfere with normal eruption, complicating orthodontic therapy. In rare cases, some root resorption may occur. Involved teeth may have hypercementosis. Fibrous dysplasia appears to be unique in its ability to displace the inferior alveolar nerve canal in a superior direction (see Fig. 23-8).
Differential Diagnosis
Other diseases can alter the bone pattern in a similar fashion. Metabolic bone diseases such as hyperparathyroidism may produce a similar pattern. However, these diseases are polyostotic, bilateral, and, in contrast to fibrous dysplasia, do not cause bone expansion. Paget’s disease may produce a similar pattern and may cause expansion, but it occurs in an older age group, and when it involves the mandible, the whole mandible is involved, in contrast to the unilateral tendency of fibrous dysplasia. Occasionally, periapical cemental dysplasia may show a similar bone pattern, but the distribution is different in that it often is bilateral, with an epicenter in the periapical region. Periapical cemental dysplasia also occurs in an older age group. With spontaneous healing of a simple bone cyst, the radiographic and histologic appearance of the new bone may be very similar to that of fibrous dysplasia.
Of paramount importance is the differentiation of osteomyelitis and osteogenic sarcoma because of both radiologic and histologic similarities. Osteomyelitis may result in enlargement of the jaws, but the additional bone is generated by the periosteum; therefore, the new bone is laid down on the surface of the outer cortex, and close examination may reveal evidence of the original cortex within the expanded portion of the jaw. Fibrous dysplasia, in contrast, expands the internal aspect of bone, displacing and thinning the outer cortex so that the remaining cortex maintains its position at the outer surface of the bone. The identification of sequestra aids in the identification of osteomyelitis. Osteogenic sarcoma may produce a similar pattern but should show malignant radiologic features (see Chapter 24).
Some difficulty may arise in differentiating ossifying fibroma of the maxilla, especially the juvenile ossifying fibroma type. If the bone pattern is altered around the teeth without displacement of the teeth from one specific epicenter, the lesion probably is fibrous dysplasia. The shape of the bone expansion of fibrous dysplasia into the antrum reflects the original outer contour of the antral wall, which is different from the more convex extension of a neoplasm.
Management
In most cases, the radiographic characteristics of fibrous dysplasia and the clinical information are sufficient to allow the practitioner to make a diagnosis without a biopsy. There are reports of exaggerated growth from stimulation of a lesion during surgical intervention in young patients. A consultation with a dental radiologist is advisable. The radiologist may supplement the examination with computed tomographic (CT) imaging, which can give a more accurate, three-dimensional representation of the extent of the lesion and can serve as a precise baseline study for future comparisons. It is reasonable to continue occasional monitoring of the lesion or ask the patient to report any changes. With most lesions, growth is complete at skeletal maturation; orthodontic treatment and cosmetic surgery may be delayed until this time. Sarcomatous changes are unusual but have been reported, especially if therapeutic radiation has been administered. In the case of female patients, hormonal changes from pregnancy or the use of oral contraceptives may stimulate growth or result in the development of lesions within the area of fibrous dysplasia, such as aneurysmal bone cysts (ABCs) or giant cell granulomas.
Periapical Osseous Dysplasias
It has been recognized more recently that it is impossible to identify cementum by either the histologic or the imaging appearance within these lesions and that this calcified material is likely amorphous bone. Therefore, the use of the term cementum should be discontinued. The term periapical osseous dysplasia (POD) has replaced periapical cemental dysplasia. This change in usage makes sense because it fits with the existing term florid osseous dysplasia (FOD), which is essentially the same disease but with a widespread involvement of the jaws.
Periapical Osseous Dysplasia
Synonyms.
Periapical cemental dysplasia, periapical cemento-osseous dysplasia, cementoma, fibrocementoma, sclerosing cementoma, periapical osteofibrosis, periapical fibrous dysplasia, and periapical fibro-osteoma are synonyms for POD.
Disease Mechanism.
POD is a localized change in normal bone metabolism that results in the resorption of normal cancellous bone and replacement with fibrous tissue and amorphous bone, abnormal bone trabeculae (similar to that seen in fibrous dysplasia), or a mixture of the two. Maturation phases correspond to the early phase, where the normal bone has been resorbed (radiolucent stage); developing phase, where abnormal bone is manufactured within the lesion (mixed radiolucent and radiopaque stage); and late or mature stage, where the internal structure is dominated by abnormal bone. By definition, the lesion is located near the apex of a tooth.
Clinical Features.
POD is a common bone dysplasia that typically occurs in middle age; mean age is 39 years. It occurs nine times more often in females than in males and almost three times more often in blacks than in whites. It also frequently is seen in Asians. The involved teeth are vital, and the patient usually has no history of pain or sensitivity. The lesions usually come to light as an incidental finding during a periapical or panoramic imaging examination performed for other purposes. The lesions can become quite large, causing a notable expansion of the alveolar process, and may continue to enlarge slowly.
Imaging Features
Location.
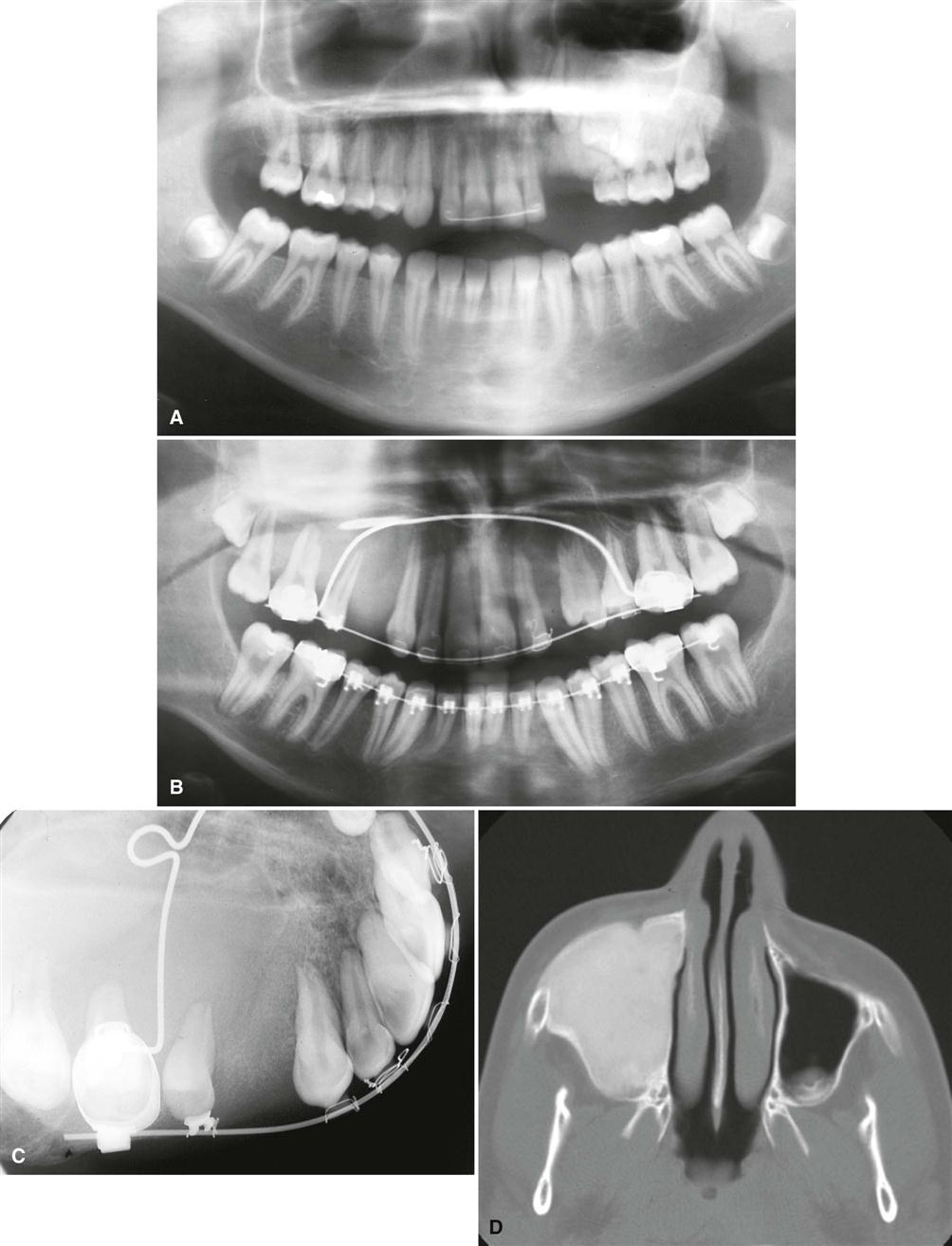
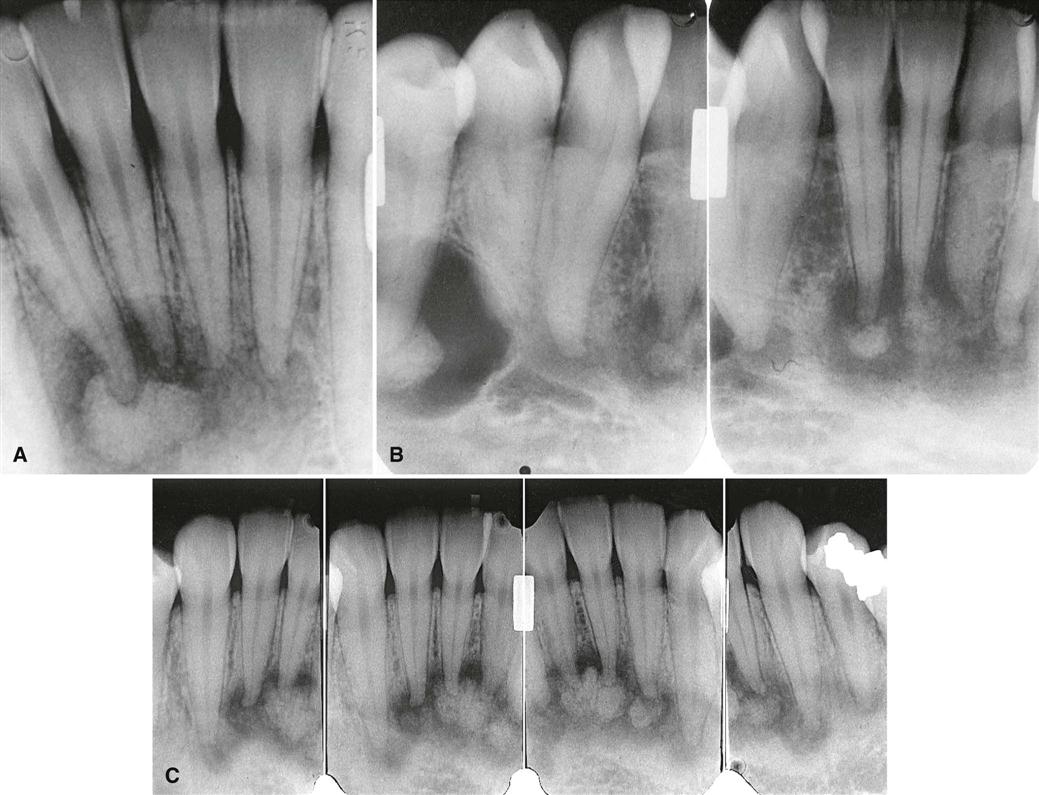
The epicenter of a POD lesion usually lies at the apex of a tooth (Fig. 23-9). In rare cases, the epicenter is slightly higher and over the apical third of the root. The condition has a predilection for the periapical bone of the mandibular anterior teeth, although any tooth can be involved, and in rare cases the maxillary teeth may be involved (Fig. 23-10). In most cases, the lesion is multiple and bilateral, but occasionally a solitary lesion arises. If the involved teeth have been extracted, this lesion can still develop, but the periapical location is less evident (Fig. 23-11). In these cases, the term osseous dysplasia may be more appropriate.
Periphery and Shape.
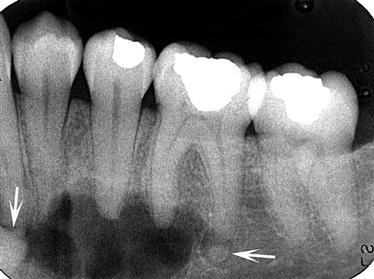
In most cases, the periphery of a POD lesion is well defined. Often a radiolucent border of varying width is present, surrounded by a band of sclerotic bone that also can vary in width (Fig. 23-12). The sclerotic bone represents a reaction of the immediate surrounding bone. The lesion may be irregularly shaped or may have an overall round or oval shape centered over the apex of the tooth.
Internal Structure.
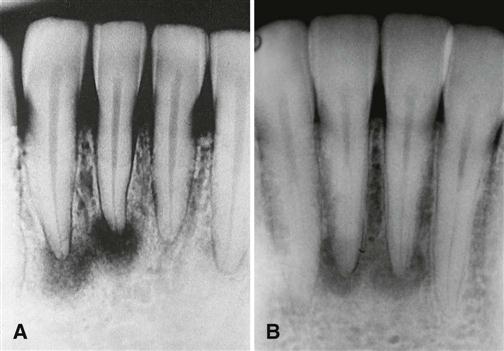
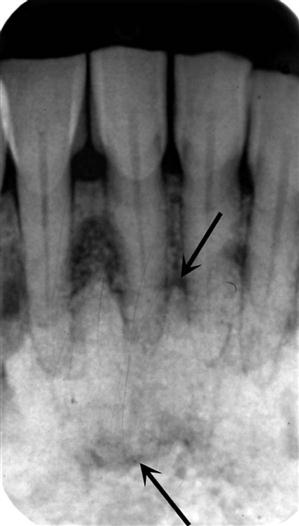
The internal structure varies, depending on the maturity of the lesion. In the early stage, normal bone is resorbed and replaced with fibrous tissue that usually is continuous with the periodontal ligament (causing loss of the lamina dura). This appears as a radiolucency at the apex of the involved tooth (see Fig. 23-9).
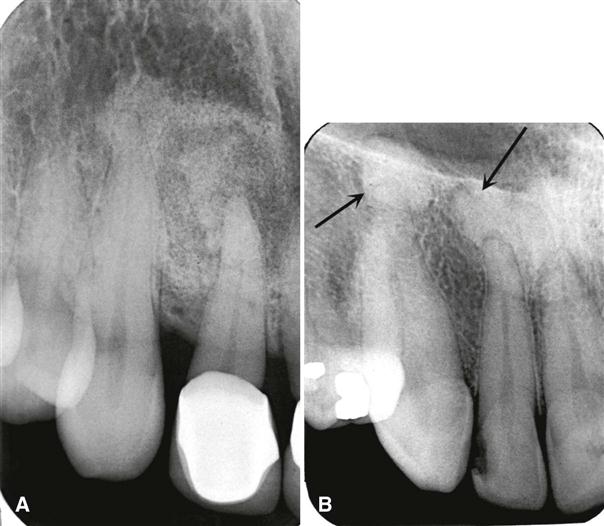
In the mixed stage, radiopaque tissue appears in the radiolucent structure. This material usually is amorphous; has a round, oval, or irregular shape; and is composed of abnormal bone (see Fig. 23-12). Sometimes the amorphous bone forms a swirling pattern (Fig. 23-13). In some cases, the radiopaque material resembles the abnormal trabecular patterns seen in fibrous dysplasia (see Fig. 23-13).
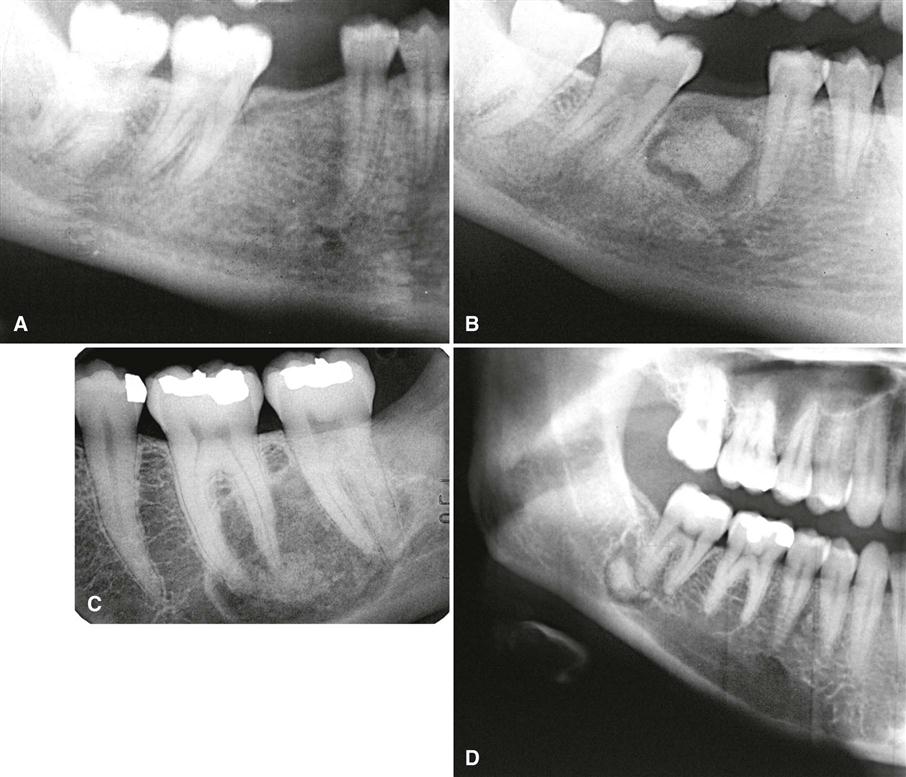
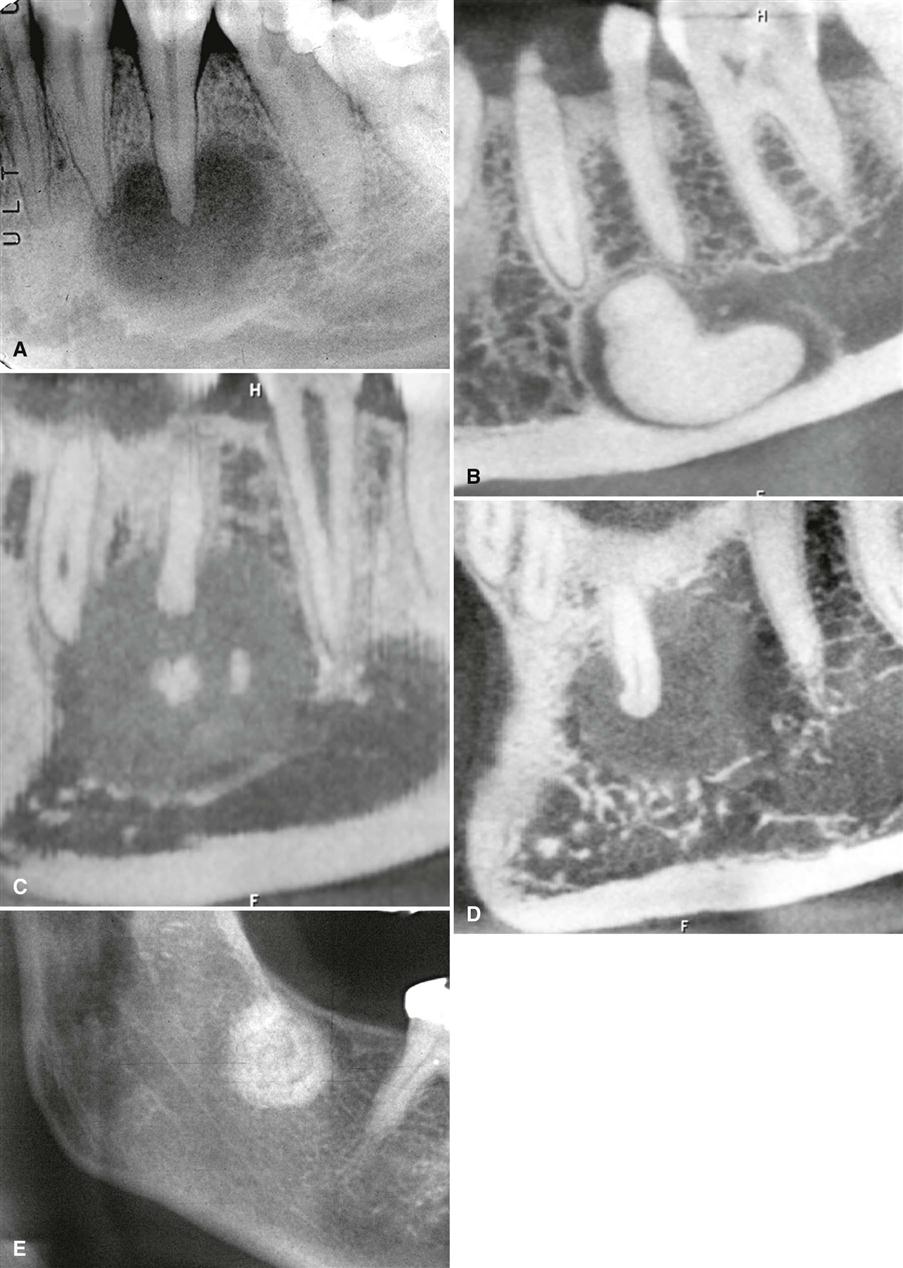
In the mature stage, the internal aspect may be totally radiopaque without any obvious pattern. A thin, radiolucent margin usually can be seen at the periphery because this lesion matures from the center outward (Fig. 23-14). Occasionally, this radiolucent margin is not apparent, which makes the differential diagnosis more difficult. The internal structure may appear dramatically radiolucent if cavities resembling simple bone cysts form within the lesion (Fig. 23-15). In some cases, the simple bone cyst extends beyond the original margin of the cemental lesion.
Effects on Surrounding Structures.
The normal lamina dura of the teeth involved with the lesion is lost, making the periodontal ligament space either less apparent or appear wider (see Fig. 23-9). The tooth structure usually is not affected, although in rare cases some root resorption may occur. Also, hypercementosis occasionally occurs on the root of a tooth positioned within the lesion. Some lesions stimulate a sclerotic bone reaction from the surrounding bone. Small lesions do not cause expansion of the involved jaw. However, larger lesions may cause expansion of the jaw, an area that is always bordered by a thin, intact outer cortex similar to that seen in fibrous dysplasia. The expansion is usually undulating in shape. This lesion may elevate the floor of the maxillary antrum.
Differential Diagnosis.
In early (radiolucent) POD lesions, the most important differential diagnosis is periapical rarefying osteitis. Occasionally, POD cannot be distinguished from this inflammatory lesion by radiographic characteristics alone. In these cases, the final diagnosis must rely on clinical information such as testing of the vitality of the involved tooth.
In the case of a solitary mature form of POD, the differential diagnosis may include a benign cementoblastoma, especially when periapical to the mandibular first molar. This tumor usually is attached to the surface of the root, which may be partly resorbed. Also, the peripheral soft tissue capsule is better defined, and there may be a unique pattern to the internal structure, such as a radiating pattern. Expansion caused by the tumor is more concentric and less undulating than in POD. The presence or absence of clinical symptoms may help distinguish POD from benign cementoblastoma. Another lesion to consider is an odontoma. Odontomas often start occlusal to a tooth and prevent its eruption, but some odontomas may have a periapical position. The organization of the internal aspect into toothlike structures and the identification of enamel (very radiopaque) can help in the differential diagnosis. Also, the peripheral cortex and soft tissue capsule of an odontoma are more uniform in width and better defined than the periphery of POD. In mature POD lesions, the appearance may resemble a dense bone island. The fi/>
Stay updated, free dental videos. Join our Telegram channel

VIDEdental - Online dental courses