2
Third Molar Surgery
INTRODUCTION
Any tooth that fails to erupt into the dental arch within the expected time frame and is no longer expected to do so is, by definition, an impacted tooth. Failure of a tooth to erupt into the arch in a timely fashion can be due to several factors such as crowding from inadequate arch length (Bolton discrepancy), retarded maturation of the third molar, malpositioned adjacent teeth, associated pathology (odontogenic cysts and tumors), trauma, previous surgery, dense overlying bone (lateral positioning) or soft tissue and systemic conditions (syndromes). The mandibular and maxillary third molars are the most common impacted teeth, followed by the maxillary canines and mandibular premolars. It is of no surprise that extraction of third molars, usually impacted, is the procedure performed with the highest frequency on daily basis by oral and maxillofacial surgeons.
The indications and timing for removal of impacted teeth and specifically third molars are set forth by the American Association of Oral and Maxillofacial Surgeons (AAOMS) parameters of care and are not discussed further here. Complication rates from the removal of impacted third molars range from 4.6% to 30.9% with an average of approximately 10%.1–6 The incidence of these complications varies with surgeon experience, patient age, and depth of impaction. Several factors are known to increase the risk of complications and these include increased age, female gender, presence of pericoronitis, poor oral hygiene, smoking, depth of impaction, and surgeon inexperience.2,5,6 The aim of this chapter is to provide a comprehensive review of the common as well as rare peri- and postoperative complications associated with impacted third molar surgery and their management.
ALVEOLAR OSTEITIS
Alveolar osteitis (AO) or “dry socket” is a clinical diagnosis with an incidence of between 1% and 37%.1,4–6,7 This wide range can best be explained by varied definitions of AO. Some studies define AO as pain that requires the patient to return to the surgeon’s office, while others define it as simply based on a clinical diagnosis of AO. In addition, some studies report only those teeth that required surgical extraction or use varied surgical protocols.5–8 The average incidence of AO in a private practice setting based on a survey of AAOMS members was 6.5%.6 Contributing factors to the development of AO include the use of oral contraceptives, smoking, increasing age, female gender, presence of pericoronitis, surgical time, surgical trauma, and compromised medical status.6,7,8
AO is often described as the loss, lysis, or breakdown of a fully formed blood clot prior to its maturation into granulation tissue. Patients will present with a myriad of symptoms and signs for approximately 3 to 5 days following extraction. The most common complaints are pain, malodor, and foul taste that do not respond well to oral analgesics and often keep a patient up at night. Clinically, a gray-brown clot or the complete absence of an organized clot may be present in the extraction socket. Food debris may or may not be present and the surrounding tissue may be erythematous and edematous. The site is exquisitely tender and often patients will have referred pain to other areas of the head and neck including the ear, eye, or temporal and frontal regions.
The incidence of AO can effectively be decreased through a variety of interventions, all of which focus on decreasing bacterial counts at the surgical site. Chlorhexidine gluconate 0.12% presurgical irrigation either with or without postoperative rinses has shown to be beneficial in decreasing the incidence of AO.7–9 Copious irrigation and lavage of the surgical site with normal saline has been reported to decrease AO. In one study, it was as effective as pre- and postoperative rinses with chlorhexidine, “Cepacol,” and normal saline. Others have demonstrated no significant difference between pulse lavage and hand syringe irrigation. Intra-alveolar antibiotics, specifically tetracycline, lincomycin, or clindamycin may also decrease the incidence of AO.8 Postoperative antibiotics have not consistently shown an ability to influence the development of AO and the evidence to support preoperative or intraoperative systemic antibiotics is controversial.3,7,8 Most studies do not demonstrate a significant difference. Overall, good surgical technique with minimal trauma, copious irrigation, and the use of chlorhexidine rinses or topical antibiotics have shown promise in decreasing the incidence of AO.
The goal in treatment of AO is to relieve pain until adequate maturation of the healing socket has occurred. Most treatment regimens focus on gentle irrigation with or without mechanical debridement and placement of obtundant dressings. Interestingly, there is very little evidence to support the use of a certain dressing or medicament over another. Commonly, iodoform gauze and eugenol are used to “pack” the socket and this packing is changed QD or QOD.4,8 Eugenol is a member of the phenylpropanoid class of chemical compounds and is beneficial due to its inhibition of neural transmission and neurotoxicity. Iodoform is an organoiodine compound that has antibacterial properties and has been used since the early 20th century as an antiseptic wound dressing. Most commercially available dry socket pastes or dressings include eugenol in combination with various other medicaments such as guaiacol, chlorobutanol, balsam peru, and butamben. The use of gelfoam as a carrier and obtundant dressing has also been reported. Patients should be seen regularly for follow up to ensure elimination of symptoms and if iodoform packing is used, to change or remove the packing. It is important to avoid the use of eugenol and other neurotoxic chemicals in the presence of an exposed inferior alveolar or lingual nerve. The use of systemic antibiotics is not recommended for treatment of AO.8 Typically, patients will have resolution of symptoms within 3 to 5 days; however, in certain patients it may take up to 14 days for complete resolution.4,8 In summary, AO is one of the more common complications of third molar surgery. Its incidence can be decreased though a combination approach of preoperative rinses, irrigation, and/or local antibiotic application, and its treatment is straightforward.
INFECTION
Surgical wound infection rates as a result of third molar extraction range from 0.8% to 4.2%, and almost exclusively involve the mandibular third molar sites.1–6,10,11 According to most general surgery and infectious disease literature, any surgical procedure within the oropharynx is considered a clean-contaminated wound, a class II wound, and carries a less than 10% risk of surgical site infection (SSI). If inflammation without purulence is noted, such as that with pericoronitis, the wound is then classified as contaminated, class III, and carries an SSI rate of 20%. The presence of purulence or necrotic tissue at the time of surgery results in a 40% risk of SSI. Class I data are available to support the use of preoperative antibiotic prophylaxis for clean-contaminated wounds; however, there are no data to support continued antibiotic administration beyond the first 24 hours after surgery.12,13,14 In relation to third molar surgery, 50% of infections are localized subperiosteal abscess-type infections occurring approximately 2–4 weeks after surgery.10 This type of infection is attributed to debris left under the surgically created mucoperiosteal flap and would likely not be prevented with the use of antibiotic prophylaxis. The remainder of third molar SSI cases are rarely severe enough to necessitate further surgery or antibiotics. SSI within the first postoperative week occurs only 0.5–1.0% of the time.10,11,15
The risk of developing an SSI associated with the removal of third molars increases with degree of impaction, need for bone removal or sectioning of the tooth, the presence of gingivitis, periodontal disease and/or pericoronitis, surgeon experience, increasing age, and antibiotic use. The benefit of systemic antibiotic administration on the incidence of SSI in relation to third molar extractions is questionable and is not currently recommended since the incidence of complications from antibiotic administration is higher than the incidence of SSI: 11% and 0.8% to 4.2%, respectively.10,11,15 It is also unlikely that perioperative systemic antibiotics are of any benefit in delayed, subperiosteal-type infections due to the nature of these infections as described previously.10
Signs of SSI can vary from localized swelling and erythema to fluctuance and trismus or systemic manifestations with fevers, dehydration, etc.10 The treatment of SSI due to third molar surgery involves surgical incision and drainage in addition to the administration of systemic antibiotics. Penicillin is often used, as the vast majority of infections are caused by a mixed flora of micro-organisms with anaerobic and gram positive streptococci being the most common. Amoxicillin has a slightly wider spectrum of activity and metronidazole can be added to cover anaerobic organisms. For the penicillin-allergic patients, clindamycin is a good choice of antibiotic and can also be used when aerobic and anaerobic coverage is desired. Most often, patients will present with a vestibular, body of the mandible, or localized subperiosteal abscess. A rare occurrence is the spread of infection along fascial tissue planes and involvement of multiple potential spaces. This situation requires surgical drainage, IV antibiotics, and close follow-up as progression to parapharyngeal, submandibular, and retropharyngeal spaces can lead to airway embarrassment and even mediastinal abscess formation with potentially fatal result.10,15
BLEEDING/HEMORRHAGE
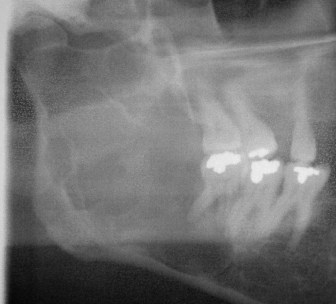
The incidence of clinically significant bleeding as a result of third molar surgery ranges from 0.2% to 5.8%.4–6 According to the AAOMS Age-Related Third Molar Study, approximately 0.7% occur intraoperatively and 0.1% postoperatively.1 Significant bleeding or hemorrhage is most often associated with mandibular third molar surgery (80%) when compared with maxillary third molar surgery (20%).16 Specific risk factors include advanced age, distoangular impactions, and deep impactions.6 Massive intraoperative bleeding is a rare occurrence and is often attributed to the presence of an arteriovenous malformation (AVM).16 As such, examination of the surgical site for gingival discoloration, palpable thrill, or bruit is necessary. Imaging may demonstrate a multilocular radiolucency in the area of AVM in proximity to the third molar tooth (Fig. 2.1). In these patients, angiography is essential to confirm diagnosis and treatment with embolization is often necessary.
Fig. 2.1. Panoramic radiographic demonstrating a multilocular radiolucency arteriovenous malformation (AVM) associated with a third molar tooth.
The most common inherited bleeding disorder, von Willebrand disease, affects an estimated 1% of individuals. Hemophilia A or B is present in 1 in 5,000 live births. Depending on patient age and sex, the first surgical procedure a patient undergoes may be third molar extraction as patients with mild to moderate forms of certain coagulopathies may be undiagnosed. Patients with an acquired or congenital coagulopathy will require further workup prior to surgery. Depending on the specific condition, recent laboratory values, factor replacement, hematology consultation, or inpatient surgery may be necessary.
Antithrombotic treatment with medications such as warfarin, thienopyridines, or aspirin is commonly encountered among patients requiring extractions. Warfarin and clopidogrel rank among the top 100 prescribed medications in the United States, an estimated 25% of individuals over age 75 currently take warfarin, and, according to the FDA, over 100 billion aspirins are taken each year. Most current literature does not recommend withholding these medications for tooth extraction. The risk of a thrombotic event outweighs any benefit of holding the medication. In patients taking warfarin, a preoperative INR (international normalized ratio) may be of value. According to Potoski, a value of 4.0 is acceptable for minor surgical procedures, 3.0 if also taking clopidogrel, aspirin, or other antiplatelet medications, and 2.5 for more involved surgery.16
The treatment of bleeding or hemorrhage begins with local measurements, pressure with gauze, and packing. Intraoperative bleeding from soft tissues can usually be controlled with cautery, taking care to avoid any neurovascular structures. Bone bleeding or bleeding from extraction sockets can be controlled through a variety of measures. Intra-alveolar hemostatic agents such as gelfoam, surgicel, avitene, collaplug, collatape, thrombin, tiseel, or bone wax may be used alone or in various combinations. Over suturing and primary closure of the wound can also assist in hemostasis and containment of the various hemostatic agents. Oral rinsing with an antifibrinolytic such as Amicar® (epsilon-aminocaproic acid) or Cyclokapron® (tranexamic acid) can aid in maintenance of an organized clot.16
In the case of prolonged postoperative bleeding, the patient should be instructed to remove loose clots and bite firmly and continuously on a moist gauze pack for 30 minutes. If this is unsuccessful, exploration and debridement of the wound should be completed under local anesthesia without vasoconstrictor to allow for diagnosis of the cause of bleeding. Granulation tissue should be debrided, irregular sharp bony edges removed, and hemostatic agents used within the alveolus to assist in bleeding control. As with intraoperative bleeding, over suturing and primary wound closure can assist in hemostasis and maintenance of the various hemostatic agents.
MANDIBULAR FRACTURE
Mandibular fracture following third molar surgery is a rare occurrence and most often associated with deeply impacted third molars in patients over 40 years of age.17 The reported incidence of mandibular fracture both intraoperative and postoperative ranges from 0.00490% to 0.00003% with a mean time to fracture ranging from 6.6 to 14 days following surgery according to studies by Iizuka and Krimmel respectively.18,19 Fractures up to 28 days following surgery have been reported and no fractures reported beyond 6 weeks after surgery.18,19 This time period correlates with increased masticatory forces due to decreased trismus, pain, and edema. Libersa, in his review of 37 fractures from 750,000 extractions, found that 8 of 10 late fractures occurred in men with 6 occurring during mastication.17 Risk factors for fracture include age over 40, male, advanced atrophy, associated pathology such as cysts or tumors, osteoporosis, full dentition and bruxism.17–19 The angle region of the mandible is of particular risk for fracture due to its relatively decreased cross-sectional area. The presence of a deeply impacted, fully developed third molar can occupy a significant portion of this cross-sectional area leaving little support following surgical extraction.18 Intraoperative mandibular fracture is almost exclusively due to the application of excessive force during third molar surgery. Often, it occurs during the use of dental elevators combined with the application of heavy pressure beyond that required to extract the tooth.4 In patients over 40 years of age presenting with partial bone impactions (loss of external oblique ridge) and risk factors, even light force can cause fracture.17
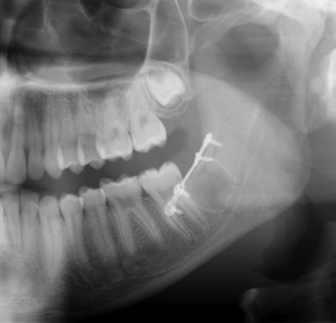
Should a fracture occur during or after third molar surgery, it should be treated immediately. Open reduction and internal fixation can be easily accomplished in this region using the Champy technique with tension band plating (Fig. 2.2). Alternatively, closed reduction with intermaxillary fixation may be appropriate in certain cases. Regardless of the technique, the complication should be communicated to the patient and/or guardian and treatment initiated.
Fig. 2.2. Panoramic radiograph demonstrating open reduction and fixation of mandibular fracture after extraction of third molar.
ORAL-ANTRAL COMMUNICATION
The extraction of maxillary molars can lead to a communication between the maxillary sinus and oral cavity. If this communication does not heal, or is treated inappropriately, it can lead to development of an oral-antral fistula (OAF). The incidence of oral-antral communication (OAC) from third molar extraction ranges from 0.08% to 0.25%; however, documented cases may under represent the actual number of cases due to the self-limiting nature of most communications and flap closure following impacted third molar removal.1–6 It should be noted that OAC is more common at the first molar site followed by the second molar site and all patients should be alerted to the possibility of OAC and even OAF following removal of maxillary molars.3,4
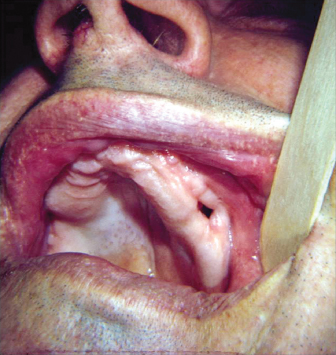
When extracting maxillary molars in close approximation to the sinus, especially when sinus pneumatization and widely divergent roots are noted radiographically, excessive force should be avoided and consideration for sectioning of roots made. Predisposing factors include pneumatization around the tooth roots, periapical infection, acute/chronic sinusitis, adjacent edentulous spaces, and traumatic extraction (Fig. 2.3).3,4,6 The assessment of an OAC should include the etiology, location, and size of the defect. Identification of an OAC can be assisted by having the patient perform the nose-blow test. The patient should pinch the nostrils together preventing air flow out of the nares. Next, have the patient attempt to blow gently though the nose while observing the extraction site. If an OAC exists, air will pass through it and bubbling of blood/fluid in the socket is observed. Another method also uses the nose-blow test but instead of observing the site directly, a mirror is placed near the site and is observed for fogging of the mirror. This test can be of particular use in third molar OAC due to the inability to visualize the depth of the socket in certain patients. Additionally, if upon inspection of the extracted tooth a segment of bone remains attached to the tooth toward the root apices, it is likely a communication exists. However, if no bone is present, this does not rule out the possibility of an OAC. The surgeon must avoid the temptation to probe or explore the extraction site as this can perforate an intact membrane and introduce foreign material, including bacteria, into the sinus cavity.
Fig. 2.3. Oral-antral communication after extraction.
Once the surgeon has determined an OAC exists, the size of the defect should be appreciated. Defects less than 2 mm in diameter will close spontaneously. If desired, measures can be taken to ensure stability of a quality blood clot in the extraction site. A collagen plug, gelfoam, and/or sutures can be placed to assist in clot formation and maintenance. A moderately sized defect of 2–6 mm in diameter will require additional measures to aid closure of the OAC. A figure-of-eight stitch should be placed over the tooth socket to assist in clot maintenance, and gelfoam or a collagen plug can be placed within the socket to assist in formation of a stable clot. In addition, multiple medications should be prescribed to prevent congestion and development of maxillary sinusitis. Amoxicillin, cephalosporins, augmentin, or clindamycin can be prescribed. Nasal decongestants such as oxymetazo/>
Stay updated, free dental videos. Join our Telegram channel

VIDEdental - Online dental courses


